Development and Clinical Validation of a Sensor-Fusion-Based Exoskeleton Robot for Elbow Spasticity Relief
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Exoskeleton systems developed thus far have demonstrated limitations in clinical applicability and objective assessment of spasticity. To address these gaps, a sensor-fusion-based elbow exoskeleton robot designed for reliable therapeutic intervention and quantitative evaluation was developed in this study. Its efficacy in quantifying and relieving spasticity in stroke patients with hemiparesis was evaluated. The robotic device integrates high-precision torque sensors and kinematic sensors to ensure accurate, sufficient, and safe spasticity management. To validate the gaugeability of the system, patients with varying Modified Ashworth Scale (MAS) scores were assessed using the robot's embedded sensor system. A total of 22 stroke patients with severe elbow spasticity were recruited and randomly assigned to either an intervention or control group. The intervention group underwent a static stretching protocol (20 min/session, 5 days/week for 2 weeks). Spasticity was quantitatively measured three times—once before and twice after the intervention—using both MAS and sensor-derived metrics, including maximum torque and gradient slope. The results demonstrated a strong correlation between therapist-assessed MAS scores and torque sensor readings (r = 0.75, p < 0.05), as well as between the MAS and gradient A (r = 0.71, p < 0.05). Statistically significant improvements were observed in both MAS (Pre: 3.29±0.55; Post-2: 2.25±0.89) and torque values (Pre: 11.22±3.62 Nm; Post-2: 9.27±3.92 Nm), confirming both the therapeutic effect and measurement precision of the system. These findings suggest that the proposed exoskeleton robot not only reduces the spasticity but also provides a reliable, sensor-based quantitative assessment of muscle tone, supporting its application as both a therapeutic and diagnostic device in neurorehabilitation.
Keywords:
Spasticity, Exoskeleton robot, Torque sensor, Modified Ashworth Scale (MAS), Elbow rehabilitation1. INTRODUCTION
Spasticity is a common and problematic sequela in hemiparetic stroke patients [1,2]. It is defined as a velocity-dependent increase in muscle tone that is characterized by a hyperactive stretch reflex following upper motor neuron injury [3]. Flexor-type elbow spasticity is frequently observed in hemiparetic stroke patients [4].
Without proper management, elbow spasticity may lead to disturbances when performing activities of daily living by limiting the range of motion or contracture of the elbow joint. Many studies have reported that spasticity is closely related to poor motor function, and a previous study reported that a 50% reduction in the range of motion of the elbow joint could lead to a loss of up to 80% of upper extremity function [1,5-7]. Consequently, aggressive management of elbow spasticity is required for successful rehabilitation of patients with hemiparetic stroke.
Many modalities, including medication, stretching exercises, braces, stretching devices, and surgery, have been used to manage spasticity in patients [8-13]. Stretching is a basic and common modality because it has the advantages of easy application, low cost, and few side effects [14-16]. Hemiplegic stroke patients are required to perform stretching exercises several times a day to relieve spasticity to a satisfactory state which they have difficulty performing without assistance. Thus, professional therapists may consume a significant portion of the available treatment time relieving spasticity in patients with hemiparetic stroke in hospitals, especially in those with severe spasticity. Therefore, we developed a robotic device to relieve elbow spasticity in a safe, easy, and repeatable manner.
Many rehabilitation robots have been developed with many advantages and unique features that our robot does not possess. To our knowledge, however, there are not many that have been applied to clinical practices. Among the exoskeletal-type robots for the upper extremities that have been either used for clinical purposes or commercialized, there are ARMIN [17] and KINARM [18]. ARMIN, developed by HOCOMA and commercialized under the brand name ARMEO, is a general-purpose six-degree-of-freedom rehabilitation robot for the upper extremities that can be applied to spasticity treatment. Its elbow part has the maximum torque capability of 27 N·m, which was determined by measuring the maximum voluntary contraction in healthy people [19]. KINARM was designed to provide horizontal flexion and extension movements at both the shoulder and elbow joints. The robot was primarily used for spasticity assessment, and the maximum torque at the elbow joint was rated at 16.5 N·m.
After the features of those two robots had been examined, the aim was to develop a robot dedicated to both the measurement and treatment of elbow spasticity; does reflect, at the same time, the clinical needs and practices more closely; and thus is readily applicable for clinical purposes. Specifically, we aim to develop a robot with the following attributes and features. First, we sought to develop a robot that could provide accurate exercise levels for spasticity treatment and/or measurements in post-stroke patients. Patients with spasticity may have varying ranges of motiondepending on their condition and may have a greater risk of injury than patients with normal joints, particularly if the range of motion during treatment is exceeded. Therefore, the robot should be able to operate accurately in accordance with the condition of the individual patient. In addition, it is proposed to develop the robot not only for treatment but also for measurement purposes. In order to consider the data recorded by the robot as meeting reliable measurement criteria, it is necessary for the intervention to be uniform. Therefore, an important aspect of robot development is the accuracy of the device.
Second, a robot that could provide sufficient torque or force was developed. Because patients with a high degree of muscle and joint stiffness may impose a physical burden on therapists, potentially leading to ineffective treatment, consideration was given to the amount of force required to treat such patients during the development stage. Accordingly, the robot was designed to reduce the burden on the therapist and ensure that effective treatment is easily accessible to patients with spasticity.
The third objective was to develop a robot that could be operated at the maximum safety level. Safety for both the patients and therapists is of paramount importance in instances where the robot is in close contact with patients. And as was demonstrated in the survey results, there is always anxiety about the stability of these robots [20]. To solve this problem, the robot system must be equipped with multiple, overlapping protection mechanisms. In addition, the robot was expected to be able to provide patients with compliant movement in order to deal with situations where a patient suddenly became stiff or convulsive; if the robot continued to operate without regard to such occurrences, the patient’s muscles and/or joints could be seriously injured.
For this study, a robot device was developed with the above described features and was applied in actual clinical practice to determine its effectiveness. Previously, many rehabilitation robots have been developed and reported on, but they have rarely been used in hospitals except for the aforementioned robots. There is also a lack of reports validating their performance. especially when they were applied to spasticity. Therefore, we aimed to provide robot based treatment for patients with elbow spasticity who were undergoing actual rehabilitation in a hospital. The study’s experimental design was formulated to confirm the robot’s therapeutic effect in a clinical setting.
In summary, a sensor fusion-based exoskeleton robot was developed to address the clinical need for quantitative spasticity assessment and therapeutic intervention.
2. DEVELOPMENT OF SYSTEM
2.1 Overall System Design
During the design process, the most important step is to establish design targets. Our design targets were determined based on an understanding of the patients and insights into their needs. To understand patients and properly address their needs, a lot of time and effort went into collaborating with medical doctors and physical therapists. As a result of these collaborations, we defined the design targets in terms of the target group and tasks.
2.2 System Specification
To meet the aforementioned design target, and with clinical relevance in mind, we derived the corresponding design specifications. Consideration of the target group and target task enabled the specification of the maximum torque, degrees of freedom (DOF), and range of motion, all of which are required by the patients and must be supplied by the robot. These specifications determine the detailed specifications of the kinematic structure of the robot, its actuators, sensors, and other details.
Because the MAS 3 patients in the target group required the highest torques owing to the severest muscle tone [21,22], these torques were used to represent the maximum torque. In practice, there was a scarcity of MAS 3 patients, and only two male patients were available for torque measurement. For these patients, the required torques were measured using a digital dynamometer (MicroFet 2; Hoggan Scientific, USA), as 38.16 N·m and 30.70 N·m. These torques were measured by a male therapist according to the standard procedures for MAS score assessment. While he exerted forces on the patients by grasping their hands, the forces were measured using a digital dynamometer attached between their hands. The maximum force was then converted to the maximum torque. We set the maximum torque to 45.99 N·m, so that the robot would be able to provide sufficient stretching torques for all patients in the target group.
In addition, the target task of providing elbow flexion and extension movement made it straightforward to determine the following kinematic structure: the robot was required to be of an exoskeleton type with one DOF revolute joint rotating from 140o (flexion) to -10o (hyperextension). Stretching exercises were to be performed at a sufficiently slow rate, especially in patients with spasticity, similar to that provided by conventional physical therapy.
The control was conducted in the presence of resisting torques originating from the muscle tones. The control objective is the concurrent control of both position and compliance in the presence of substantial resisting torques.
As feedback controller, we employed time-delay control(TDC) [23,24], whose control law is described as
| (1) |
where τ denotes control input caused by a motor; q, , the angular position, velocity, and acceleration, respectively ; ㆍd the desired value of ㆍ; e = qd - q the tracking error. L is a very small time delay (L = 0.002 s), which is intentionally introduced to estimate and compensate for the robot dynamics and uncertainties, such as the resisting torques. Here, M represents the control gain for the TDC, and KD and KP denote feedback gains. These were set to M = 0.3 kg·m2, KD = 20, and KP = 100. In (1), any desired dynamics can be inserted in the second bracket, including the desired compliance (or impedance) for patient safety and comfort [25-27]. As such, we employed typical compliance dynamics as
| (2) |
where Md, Bd, and Kd denote the desired mass, damping, and stiffness, respectively; τext the interaction torque on the human limb exerted by the robot, which is to be measured by the torque sensor.
As a result of these compliance dynamics, the TDC controls the dynamic interaction between the robot and the environment – the patient’s elbow in our case – such that the robot behaves as if it were a virtual mass, combined with a spring and damper interacting with the environment. This allows the robot’s actions to be somewhat adaptive or compliant when the patient exhibits sudden spasms or when there is a sudden increase in stiffness. Consequently, the compliance control equation becomes
| (3) |
where Md = 2.0 Nm·s2, Bd = 89.44 N·m·s, and Kd = 10.0 N·m. The q is measured by the encoder, whereas and are determined by numerical differentiation using low-pass filtering, and τext is measured by the torque sensor.
For safety, excessive torque and motion beyond the range of motion (ROM) are prevented by a triple-protection mechanism consisting of a software-based element, an electrical element, and a mechanical element.
Specifically, the software-based element features a set of control codes that use the encoder signal to monitor the robot’s position with respect to the ROM. If the robot moves beyond this, then the control code is to halt that movement. As the electrical means for protection, the input to the motor driver is to be restricted to ± 10V, which corresponds to the torque limit of ± 59.4 N·m. Note that this torque limit is the sum of the maximum resisting torque, 45.99 N·m, and the extra torque to drive the mechanism itself. Any torque exceeding this limit is to be truncated to prevent excessive torque. Mechanical stops were installed in cases where all the aforementioned means failed. These adjustable stops were designed to be placed at both the maximum and minimum ROM so that no motion was allowed beyond the ROM. In addition to these three methods, an emergency stop button is designed.
The aforementioned compliance control is used to ensure security by making the robot comply with patients when they exhibit sudden, abnormal behavior.
excessive physical strain, enabling clinicians to promptly adjust the intensity of the therapy [12]. The joint angle sensor utilizes an encoder to accurately measure the joint angles in real time, providing essential feedback regarding the patient's ROM, which is a critical metric for evaluating rehabilitation progress [13]. A flexible pressure sensor embedded within the wearable gloves was used to measure the pressure distribution. This sensor captures data on grip strength, weight distribution, and postural balance and offers valuable insights into motor recovery [14].
2.3 Prototype
Fig. 1 shows the fabricated robot, where Figs. 1 (a), (b), and (c) display its anterior, lateral, and superior views, respectively. Its parts are as follows.

Elbow exoskeleton robot. (a) Anterior view of exoskeleton robot that mainly illustrates the driving part of the robot, (b) lateral view of exoskeleton robot showing splint locations, (c) superior view, (d) anterior view showing extension of the upper limb.
The exoskeleton robot developed for elbow rehabilitation consists of three major parts: an upper-arm support that holds the upper arm, a driving part that rotates the elbow joint, and a forearm support that holds the forearm and provides movement (Fig. 1 (d)).
The driving part of the robot system, used to provide elbow flexion and extension movement, is composed of a brushless DC motor (EC-4pole 30, Maxon Motor, Swizerland; nominal torque 136 mN·m), a planetary gear head (GP-42C; Maxon Motor, Switzerland; gear ratio 6:1), a harmonic drive (CSG-20B-100, THK, gear ratio 100:1), and a torque sensor (TRT-500; Transducer Techniques, USA; capacity range 56.49 N·m). Via this configuration, the final output torque is 59.49 N·m, similar to the value specifically required to control the increase in muscle tone owing to spasticity in patients with a MAS score of 3. In addition, the motor was equipped with a quadrature encoder with a resolution of 500 pulses/rev, equivalent to the resolution of 0.3×10-3o at the elbow joint.
The robot control system is composed of a hardware platform and a real-time operating system. The hardware platform comprises an industrial PC, Intel Core i3-3240 based, and a dedicated interface board (Sensoray s626 board), used to interface with the motor and sensors. On this platform, a real-time Linux OS (Ubuntu 14.04, 64 bit) and a real-time application interface (RTAI 4.1) were installed. The TDC was implemented in this environment [23] with a sampling time of 0.002 s (500 Hz).
We tested whether the safety software was functioning properly; it was confirmed that it successfully halted the robot movement upon detecting joint values exceeding the ROM. It was also verified that any input voltage to the motor drive exceeding ± 10 V was truncated to ± 10 V. In addition, we checked that all the mechanical stops shown in Figs. 2 (a) and (b) and the emergency button in Fig. 2 (c) functioned as they were supposed to be.
In addition, accessories were installed to provide patients with comfortable and stable arm postures. For example, there are two resting splints shown in Fig. 1 (b) that support the forearm and upper arm, which are specifically tailored to the human body shape to provide patient comfort. The forearm support part has a length adjuster that can meet the lower 5% of length of a female forearm and the upper 5% of that of a male forearm derived from previous Korean anthropometric data [28]. The handgrip portion of the forearm prevents the hand from shaking or moving.
3. EXPERIMENTAL SETUP AND RESULTS
3.1 Experimental Setup
Twenty-two hemiparetic stroke patients (mean age 55.64 years ± 8.36, range 42-77; 14 males and 8 females) were recruited to this study. The following inclusion criteria were applied: (1) first ever stroke; (2) over 20 years old; (3) severe spasticity of the affected elbow flexors MAS score ≥ 2); (4) no contracture of the affected elbow; (5) no history of peripheral nerve injury or musculoskeletal disease (e.g., arthritis, musculotendinous injury, and bone fractures ); (6) patients without severe cognitive problems (MMSE>25); (7) ability to sit independently; and (8) no history of any invasive procedure for the treatment of spasticity (botulinum toxin injection, neurolysis, or surgery) within six months of the start of this study. Drugs capable of affecting spasticity were administered throughout the study. All patients were randomly assigned to either the intervention group (11 patients; 7 males; mean age, 55.10±7.06 years) or the control group (11 patients; 7 males; mean age, 56.18±9.80 years). Three patients in the intervention group dropped out of the study due to personal reasons. As a result, eight patients (8 patients; 6 males; mean age, 54.50 ± 7.33 years) finished the intervention program. No significant differences in demographic and baseline clinical data (MAS scores) were detected between the intervention and control groups (p > 0.05) (Table 1).
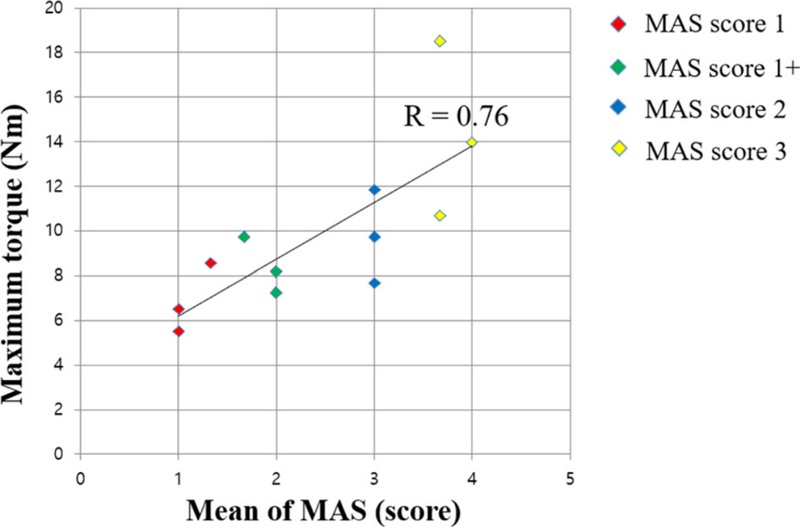
To investigate the correlation between the MAS score measured by the therapist and the values measured by the robot, three patients at each MAS score level (MAS score 1, 1+, 2, 3) were recruited. The MAS score for each patient was remeasured by three blinded therapists, and the average of those MAS scores was used as the representative score. The correlation between the MAS scores measured by the therapists and the maximum torque value among the torque values measured by the robot is presented in Fig. 3. The value of the correlation coefficient (R) for that relationship was 0.76, indicating a strong relationship.
3.2 System performance evaluation
The spasticity of the affected elbow was assessed three times over 2 weeks. The first assessment (Pre) was performed before the start of the stretching program, the second (Post-1) after the first week of therapy, and the third (Post-2) immediately after the end of the program. Assessments were conducted by determining each participant’s MAS score, which was used to evaluate the severity of spasticity in the flexors of the elbow joint. The MAS scores were as follows: 0 = no increase in muscle tone; 1 = slight increase in muscle tone, manifested by catch and release or by minimal resistance at the end of the ROM when the affected parts were moved in flexion or extension; 1+ = slight increase in muscle tone, manifested by a catch followed by minimal resistance in less than half of the ROM; 2 = a more marked increase in muscle tone throughout most of the ROM, but with passive movement difficulty; and 4 = affected part rigid in flexion or extension [3]. MAS score categories 1+ to 4 were modified to fall within the range of 2–5 for the statistical analysis.
The intervention group patients placed the affected forearm and upper-arm on the respective resting splints, and the forearm and upper-arm were fixed in the splints by using Velcro® straps. Subsequently, the elbow joint axis of the robot must be adjusted such that it aligns with the lateral and medial epicondyles of the patient’s elbow. All interventions were carried out by stretching from a starting angle of 120o (flexion) to -10o (hyper extension). For assessment, three therapists entered the room separately (to maintain objectivity), and each therapist measured and recorded the patient’s MAS scores. The robot was then attached to the patient and it started to stretch the patient’s elbow under the supervision of a therapist. When −10o hyperextension was reached, the robot maintained that stretched position for 10 s and then returned to the initial flexion position of 120o. This was repeated twice, and the robot was stopped. We then performed stretching exercises at rates of 5 o/s, 15 o/s, and 25 o/s and collected data for each patient. These stretching speeds were selected based on clinical practice and previous studies that demonstrated the velocity-dependent characteristics of spasticity, as defined by the Modified Ashworth Scale.
The patients in the intervention group received robotic exercise treatment for approximately 20 min, five days/week for two weeks at a stretching speed of 5 o/s and a stretching hyperextension hold time of 30 s [29].
Statistical analysis was performed with SPSS (SPSS Inc. Released 2006. SPSS for Windows, version 15.0. SPSS Inc., Chicago, IL, USA). Demographic data, MAS scores, and sensor values between the intervention and control groups were compared by using the Mann-Whitney U test. Statistical significance was accepted for p values of < 0.05.
Eight patients completed the intervention program without any side effects or complications. The upper graph in Fig. 4 shows the change in the mean elbow MAS scores in the intervention and control groups over the two-week treatment period. A significant difference was observed in the mean elbow MAS scores between Pre (MAS = 3.29) and Post-2 (MAS = 2.25) assessments in the intervention group (p < 0.05). There was no significant difference in the control group between Pre (MAS = 3.12) and Post-2 (MAS = 3.06) assessments (p > 0.05).
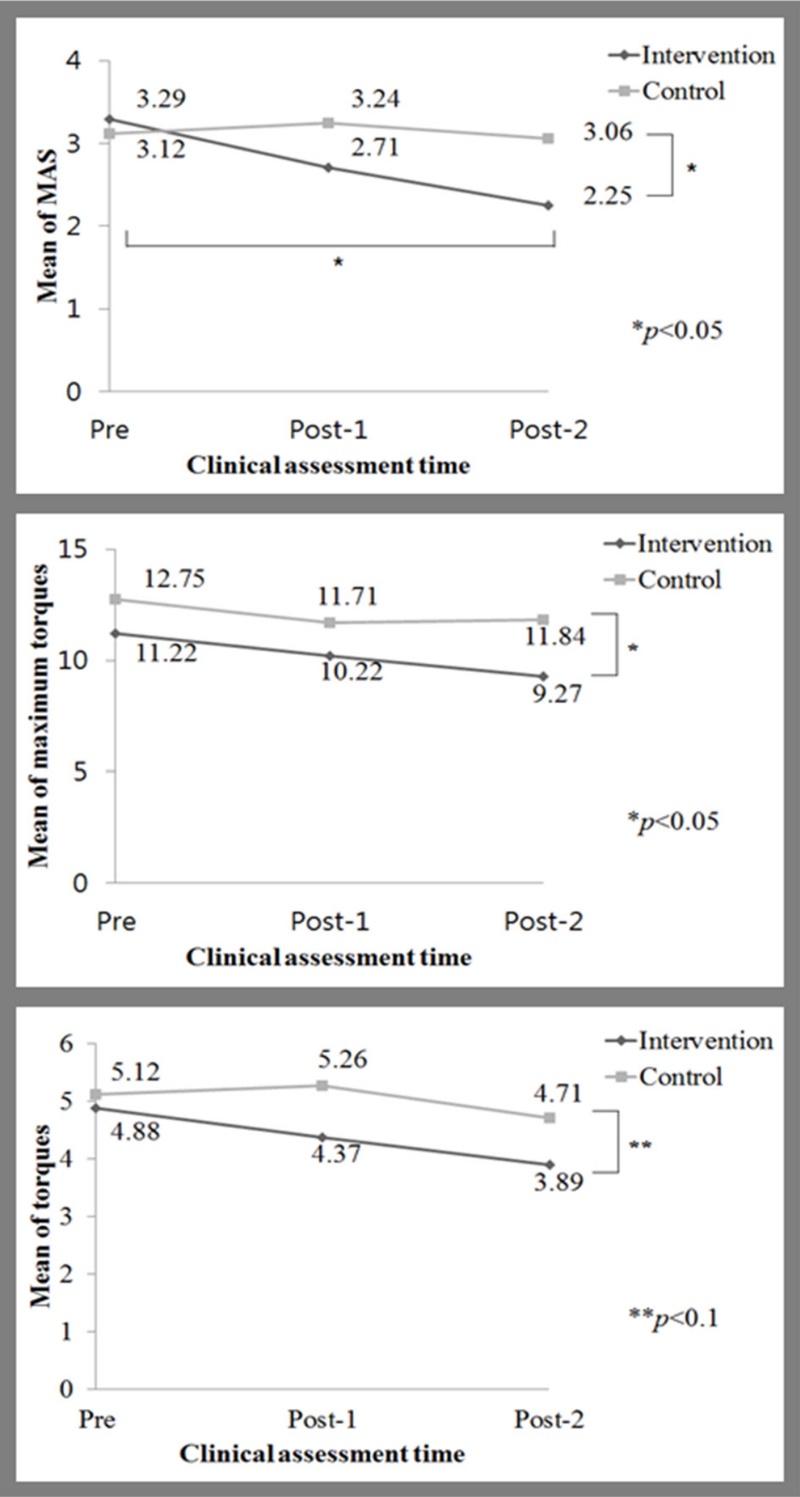
Changes in mean values in the intervention and control groups over the two week study period: (top) changes in mean MAS scores, (middle) changes in mean maximum torque values, and (bottom) changes in mean of torque values.
A significant difference in the mean maximum torque was detected between the intervention group (9.27) and the control group (11.84) in the Post-2 assessment (p < 0.05). A significant difference was detected in the Post-2 period (p < 0.1) between the intervention group (3.89) and the control group (4.71).
4. CONCLUSIONS
In this study, we developed and clinically validated a sensor fusion-based elbow exoskeleton robot designed for the treatment and measurement of elbow spasticity in hemiparetic stroke patients. The proposed system demonstrated high accuracy, sufficient torque output, and a high level of safety through the integration of torque, position, and compliance control mechanisms.
The clinical results confirmed the effectiveness of the robot in reducing spasticity, as evidenced by significant improvements in the Modified Ashworth Scale (MAS) scores and torque measurements in the intervention group. Moreover, the strong correlations between the therapist-assessed MAS scores and robot-measured torque values support the utility of this system as an objective assessment tool for spasticity.
Future studies should focus on refining the adaptive control algorithms of the system to enable fully personalized treatment protocols. Additionally, we plan to integrate the measurement and treatment functionalities in real-time, allowing on-the-fly adjustment of therapeutic parameters based on the patient’s condition. Expanding the clinical trials to larger and more diverse populations will further verify the applicability of robots in real-world healthcare settings. We aim to enhance the adaptability of robots by integrating real-time physiological feedback (e.g., EMG signals) and machine-learning algorithms to personalize therapy protocols. Furthermore, wireless connectivity and cloud-based data analytics should be explored to facilitate remote monitoring and longitudinal patient tracking in home-based rehabilitation settings.
Acknowledgments
This research was supported by Changwon National University from 2023 to 2024.
References
-
M. Schinwelski, J. Sławek, Prevalence of spasticity following stroke and its impact on quality of life with emphasis on disability in activities of daily living. Systematic review, Neurol. Neurochir. Pol. 44 (2010) 404–411.
[https://doi.org/10.1016/S0028-3843(14)60300-5]

-
J.R. McGuire, R.L. Harvey, The prevention and management of complications after stroke, Phys. Med. Rehabil. Clin. N. Am. 10 (1999) 857–874.
[https://doi.org/10.1016/S1047-9651(18)30167-0]

-
R.W. Bohannon, M.B. Smith, Interrater reliability of a modified Ashworth scale of muscle spasticity, Phys. Ther. 67 (1987) 206–207.
[https://doi.org/10.1093/ptj/67.2.206]

-
S. Viscuso, S. Pittaccio, M. Caimmi, G. Gasperini, S. Pirovano, E. Villa, et al., Pseudoelastic nitinol-based device for relaxation of spastic elbow in stroke patients, J. Mater. Eng. Perform. 18 (2009) 805–813.
[https://doi.org/10.1007/s11665-009-9418-6]

-
J.S. Ryu, J.W. Lee, S.I. Lee, M.H. Chun, Factors predictive of spasticity and their effects on motor recovery and functional outcomes in stroke patients, Top. Stroke Rehabil. 17 (2010) 380–388.
[https://doi.org/10.1310/tsr1705-380]

-
A.-K. Welmer, M. Von Arbin, L.W. Holmqvist, D.K. Sommerfeld, Spasticity and its association with functioning and health-related quality of life 18 months after stroke, Cerebrovasc. Dis. 21 (2006) 247–253.
[https://doi.org/10.1159/000091222]

-
J.O. Sojbjerg, The stiff elbow: How I do it, Acta Orthop. Scand. 67 (1996) 626–631.
[https://doi.org/10.3109/17453679608997771]

-
S.H. Jang, S.H. Ahn, S.M. Park, S.H. Kim, K.H. Lee, Z. I. Lee, Alcohol neurolysis of tibial nerve motor branches to the gastrocnemius muscle to treat ankle spasticity in patients with hemiplegic stroke, Arch. Phys. Med. Rehabil. 85 (2004) 506–508.
[https://doi.org/10.1016/S0003-9993(03)00468-4]

-
R. Turk, J.H. Burridge, R. Davis, G. Cosendai, O. Sparrow, H.C. Roberts, et al., Therapeutic effectiveness of electric stimulation of the upper-limb poststroke using implanted microstimulators, Arch. Phys. Med. Rehabil. 89 (2008) 1913–1922.
[https://doi.org/10.1016/j.apmr.2008.01.030]

-
E. Cardoso, B. Rodrigues, R. Lucena, I.R. de Oliveira, G. Pedreira, A. Melo, Botulinum toxin type A for the treatment of the upper limb spasticity after stroke: a meta-analysis, Arq. Neuropsiquiatr. 63 (2005) 30–33.
[https://doi.org/10.1590/S0004-282X2005000100006]

-
N. A. Lannin, I. Novak, A. Cusick, A systematic review of upper extremity casting for children and adults with central nervous system motor disorders, Clin. Rehabil. 21 (2007) 963–976.
[https://doi.org/10.1177/0269215507079141]

-
S.H. Jang, S.M. Park, S.H. Kim, S.H. Ahn, Y.W. Cho, M.O. Ahn, The effect of selective tibial neurotomy and rehabilitation in a quadriplegic patient with ankle spasticity following traumatic brain injury, Yonsei Med. J. 45 (2004) 743–747.
[https://doi.org/10.3349/ymj.2004.45.4.743]

- K.-H. Tsai, C.-Y. Yeh, H. Y. Chang, J.J. Chen, Effects of a single session of prolonged muscle stretch on spastic muscle of stroke patients, Proc. Natl. Sci. Counc. Repub. China B 25 (2001) 76–81.
-
J. Gracies, Pathophysiology of impairment in patients with spasticity and use of stretch as a treatment of spastic hypertonia, Phys. Med. Rehabil. Clin. N. Am. 12 (2001) 747–768.
[https://doi.org/10.1016/S1047-9651(18)30031-7]

-
T. Pin, P. Dyke, M. Chan, The effectiveness of passive stretching in children with cerebral palsy, Dev. Med. Child. Neurol. 48 (2006) 855–862.
[https://doi.org/10.1111/j.1469-8749.2006.tb01236.x]

- M. Stokes, Physical management in neurological rehabilitation, 2nd ed., Elsevier Mosby, Edinburgh, 2004.
-
T. Nef, R. Riener, ARMin-design of a novel arm rehabilitation robot, Proceedings of 9th Int. Conf. Rehabil. Robot. (ICORR 2005), Chicago, USA, 2005, pp. 57–60.
[https://doi.org/10.1109/ICORR.2005.1501051]

-
S.H. Scott, Apparatus for measuring and perturbing shoulder and elbow joint positions and torques during reaching, J. Neurosci. Methods 89 (1999) 119–127.
[https://doi.org/10.1016/S0165-0270(99)00053-9]

-
T. Nef, M. Mihelj, R. Riener, ARMin: a robot for patient-cooperative arm therapy, Med. Biol. Eng. Comput. 45 (2007) 887–900.
[https://doi.org/10.1007/s11517-007-0226-6]

- National Rehabilitation Center, Status and Development Strategy of Rehabilitation Robot Intermediary Research, National Rehabilitation Center, Republic of Korea, 2012.
-
J. Mehrholz, K. Wagner, D. Meissner, K. Grundmann, C. Zange, R. Koch, et al., Reliability of the Modified Tardieu Scale and the Modified Ashworth Scale in adult patients with severe brain injury: a comparison study, Clin. Rehabil. 19 (2005) 751–759.
[https://doi.org/10.1191/0269215505cr889oa]

- J.J. Craig, Introduction to robotics: mechanics and control, 3rd Ed., Pearson/Prentice Hall, Upper Saddle River, New Jersey, 2005.
-
K. Youcef-Toumi, O. Ito, A time delay controller for systems with unknown dynamics, J. Dyn. Sys., Meas., Control. 112 (1990) 133–142.
[https://doi.org/10.1115/1.2894130]

-
T.S. Hsia, T. Lasky, Z. Guo, Robust independent joint controller design for industrial robot manipulators, IEEE Trans. Ind. Electron. 38 (1991) 21–25.
[https://doi.org/10.1109/41.103479]

-
S. Jung, T.C. Hsia, R.G. Bonitz, Force tracking impedance control of robot manipulators under unknown environment, IEEE Trans. Control Syst. Technol. 12 (2004) 474–483.
[https://doi.org/10.1109/TCST.2004.824320]

-
M. Jin, S.H. Kang, P.H. Chang, Robust compliant motion control of robot with nonlinear friction using time-delay estimation, IEEE Trans. Ind. Electron. 55 (2008) 258–269.
[https://doi.org/10.1109/TIE.2007.906132]

-
S.H. Kang, M. Jin, P.-H. Chang, A solution to the accuracy/robustness dilemma in impedance control, IEEE/ASME Trans. Mechatron. 14 (2009) 282–294.
[https://doi.org/10.1109/TMECH.2008.2005524]

- Size Korea, Korean Human Body Measurement Data. https://sizekorea.kr, , 2024 (accessed 25 September 2024).
-
Y.J. Jung, J.H. Hong, H.G. Kwon, J.-C. Song, C. Kim, S. Park, et al., The effect of a stretching device on hand spasticity in chronic hemiparetic stroke patients, NeuroRehabilitation 29 (2011) 53–59.
[https://doi.org/10.3233/NRE-2011-0677]