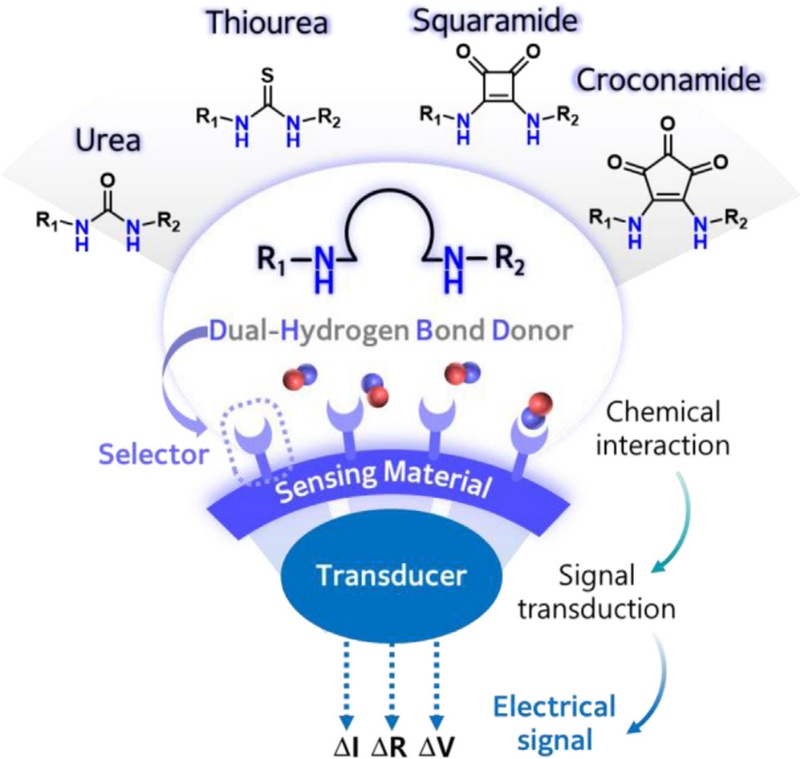
Dual-Hydrogen Bond Donors for Chemical Sensors: Transducing Chemical Interactions into Electrical Signals
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Dual-hydrogen bond donors are molecular selectors capable of forming directional hydrogen bonds with various chemical species. Structural tunability of these selectors enables the precise control of binding affinity and selectivity, making them highly attractive for chemical sensing applications. As a result, considerable research has focused on developing chemical sensors utilizing dual-hydrogen bond donors as selectors. The selectivity and sensitivity of such sensors can be enhanced by tailoring the molecular structures of dual-hydrogen bond donors and modulating their hydrogen bonding ability. These features offer significant potential for developing diverse chemical sensors that utilize various signal transduction mechanisms. As electrical signals can be readily acquired, processed, stored, and transmitted, sensors that convert chemical interactions into electrical signals are considered highly suitable for real-time monitoring and device integration. In this review, we provide an overview of dual-hydrogen bond donors, focusing on their structural characteristics, acidity, and hydrogen bonding properties. We then highlight their applications in chemical sensors designed for gas- and liquid-phase analyte detection. In conclusion, this review offers comprehensive insight into the correlation between the chemical structure of dual-hydrogen bond donors and their sensing performances.
Keywords:
Dual-hydrogen bond donor, Selectors, Chemical sensors, Gas sensors, Liquid sensors1. INTRODUCTION
The importance of chemical sensors is increasing across various fields, ranging from environmental monitoring to clinical diagnosis [1]. The interest in artificial sensory systems that mimic human olfactory and gustatory perception is increasing, emphasizing the need for advanced chemical sensors [2-4]. In this context, miniaturized electronic sensors have attracted significant attention because electronic signals can be efficiently acquired, processed, and stored [5-7].
The performance of chemical sensors is determined by the sensing material, which facilitates the chemical interaction and generates the signal output. The sensing materials consist of two main components: a selector and transducer [8]. The selector induces chemical interaction toward target analytes through intermolecular interactions, including hydrogen bonding, electrostatic forces, and π–π interactions, while the transducer converts these chemical interactions into measurable electrical signals. Among various intermolecular interactions, hydrogen bonding is particularly attractive due to its directionality and tunability [9,10].
Dual-hydrogen bond donors are representative molecule selectors that form strong and geometrically defined hydrogen bonds. They typically consist of two amide –NH groups that are symmetrically arranged and spatially separated, enabling directional and cooperative hydrogen bond donation. By introducing asymmetric N,N’-substituents on the two amide – NH groups of dual-hydrogen bond donors, additional functionalities, such as supplementary hydrogen bonding, π–π interactions, and electrostatic interactions, can be incorporated by tailoring the molecular structures of these donors. The hydrogen bond donating ability and binding properties of dual-hydrogen bond donors are primarily governed by the acidity of the NH protons [11]. This acidity can be systematically modulated by introducing electron-withdrawing or electron-donating substituents, enabling precise control over hydrogen bond strength. This structural tunability facilitates the rational design of selectors tailored for selective interaction with analytes.
In this context, extensive efforts have been made to develop chemical sensors that exploit the chemical interaction properties of dual-hydrogen bond donors. In general, dual-hydrogen bond donors can be directly employed as optical (i.e., colorimetric and fluorescent) sensors [12-14]. The concept of optical sensing using dual-hydrogen bond donors is well established and has been comprehensively reviewed in the literature [15-17]. In addition, dual-hydrogen bond donors have attracted significant interest for the synthesis of composite materials for chemical sensors that transduce chemical interactions into distinguishable electrical signals. Although dual-hydrogen bond donor-based chemical sensors that utilize electrical signal transduction have been actively investigated, no review on this topic has been published in recent years.
In this review, we discuss dual-hydrogen bond donors and their composite derivatives for application in chemical sensors. In particular, we focus on sensors that transduce the chemical interaction between sensing materials and analytes into electrical signals (Fig. 1). We first introduce the structural and chemical properties of dual-hydrogen bond donors, followed by a discussion of their application in chemical sensors in the gaseous and liquid phases. This review comprehensively discusses the correlation between the chemical binding affinities of dual-hydrogen bond donors toward specific analytes and their sensing performances, providing design strategies for the development of novel sensing materials with improved selectivity and sensitivity.
2. DUAL-HYDROGEN BOND DONOR
2.1 Dual-hydrogen bond donors as molecular selectors
As the importance of molecular recognition continues to increase, molecular selectors that facilitate specific and reversible interactions with target molecules have gained considerable attention. Consequently, there have been extensive efforts to develop various molecular selectors that enable diverse intermolecular interactions, including hydrogen bonding, π–π stacking, electrostatic attraction, and coordination bonding, to achieve selective molecular recognition [18,19]. In particular, hydrogen bonding has attracted significant interest because of its directionality and chemical tunability [20].
Notably, dual-hydrogen bond donors, which provide two-directional hydrogen bonding through –NH groups, have emerged as highly effective selector motifs. The strength of hydrogen bonding is closely correlated to the acidity of NH protons. Therefore, the pKa values of dual-hydrogen bond donors are often used as indicators of hydrogen bond strength. This relationship has been well-established through computational studies, which have quantitatively correlated pKa values with hydrogen bond strengths [21].
Urea and thiourea are common dual-hydrogen bond donors characterized by planar geometries. It is known that thiourea exhibits a lower pKa value than its urea analogs, indicating that thiourea exhibits stronger hydrogen bond strength. Notably, the NH proton of thiourea can be deprotonated upon chemical interaction with basic anions such as F– and AcO–, while urea exhibits hydrogen bonding [22].
In addition to urea and thiourea, other dual-hydrogen bond donors have been developed to enhance hydrogen bond donating ability and improve binding selectivity. Among these, squaramide is one of the most widely studied motifs, possessing two amide –NH groups connected to the four-membered squaric ring [23]. The –NH groups in squaramides are spatially more separated than those in the urea and thiourea analogs. In addition, its pseudo-aromatic nature enhances the acidity of –NH groups, which differentiates them from (thio)urea [24]. Therefore, squaramides generally exhibit stronger hydrogen bond donating ability compared to (thio)urea.
Furthermore, croconamides have been introduced as novel dual-hydrogen bond donors derived from cyclic oxocarbon structures [25]. Notably, croconamides exhibit significantly higher acidity than their squaramide analogs, further enhancing their hydrogen bond donating ability [26]. These developments highlight the structural diversity and tunability of dual-hydrogen bond donors, enabling the rational design of molecular selectors with tailored hydrogen bonding interactions suited for specific analyte recognition. Owing to these features, dual-hydrogen bond donors have gained increasing attention in the design of chemical sensors with enhanced sensing performance.
3. GAS SENSORS
3.1 General features of gas sensors
Gas sensors have been developed to monitor air quality and ensure safety in both daily life and occupational environments [27-30]. In severe cases, the early detection of toxic gases is critical for protecting human health. More recently, the detection of volatile organic compounds (VOCs) in human breath has gained considerable attention as a promising method for non-invasive disease diagnosis [31-35]. Consequently, considerable efforts have been made to develop various types of gas sensors capable of precisely discriminating gaseous analytes.
Various transduction techniques have been applied to develop gas sensors, including potentiometric, capacitive, optical, and chemiresistive [36-40]. Among these, chemiresistive-type transduction techniques are widely applied because of their simple working principles and ability for continuous monitoring [41]. Chemiresistive sensors exhibit changes in electrical resistance in response to chemical interactions occurring within the sensing layer.
However, chemiresistive gas sensors commonly suffer from issues such as cross-sensitivity and the need for elevated operating temperatures, which increase energy consumption [42]. To overcome these limitations, novel sensing materials that enable room-temperature operation while exhibiting high selectivity toward target gas analytes have been developed. One effective strategy to further improve selectivity involves functionalizing these sensing materials with selectors, which can facilitate specific chemical interactions with target analytes [43].
3.2 Dual-hydrogen bond donor-based gas sensors
Dual-hydrogen bond donors, which serve as active binding sites for small gas molecules, have been employed as functional sensing materials in gas sensor development [44,45]. Both small molecules and polymeric derivatives containing dual-hydrogen bond donors have been utilized to interact directly with target gas analytes through hydrogen bonding. For example, ethynylated-thiourea derivatives coated onto interdigitated electrodes exhibited a chemiresistive response to CO2 at room temperature [44].
Moreover, Zhou et al. developed a chemiresistive gas sensor that uses squaramide to detect ammonia (NH3) [46]. Specifically, poly(4,4′-azodianiline-squaramide) (PADS) was synthe-sized as a sensing layer (Fig. 2 (a)). The polymer contained squaramide, facilitating effective hydrogen bonding interactions with the nitrogen atoms of NH3. SEM and TEM images revealed that the PADS microspheres featured a hierarchical porous structure decorated with surface nanoplates (Fig. 2 (b)).
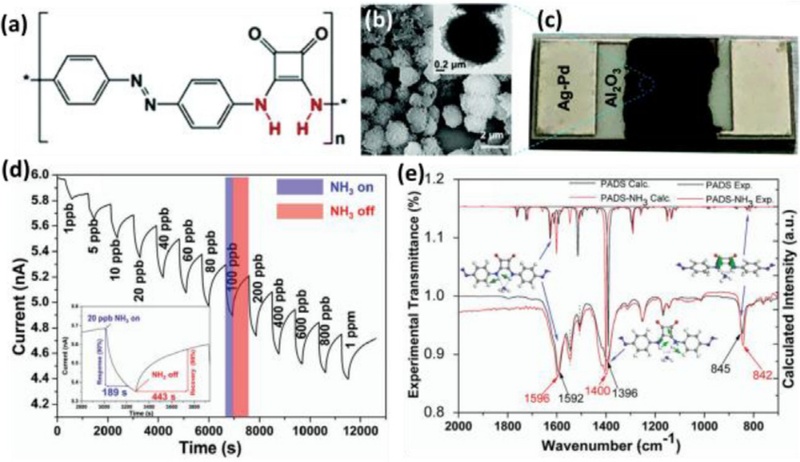
Chemiresistive gas sensor using squaramide. (a) Chemical structure of PADS. (b) SEM and TEM images of PADS microspheres. (c) Camera image of chemiresistive gas sensor. (d) Dynamic current transition toward 1 ppb–1 ppm of NH3. (e) Experimental and theoretical IR spectra of PADS before and after NH3 exposure. Reprinted with permission from Ref. [46], Copyright (2019) Royal Society of Chemistry.
The chemiresistive gas sensor was fabricated using PADS powder and a ceramic substrate with Ag-Pd interdigital electrodes to characterize the NH3 sensing properties (Fig. 2 (c)). The real-time current transitions of the PADS exhibited reversible and rapid reactions toward NH3 at concentrations ranging from 1 ppb to 1 ppm (Fig. 2 (d)). The PADS current decreased upon exposure to NH3 and recovered upon N2 purging. In addition, the sensor exhibited excellent long-term stability over 240 days. The humidity effect on the sensing performances of PADS was investigated, and the results revealed that enhanced sensor responses were observed with increasing relative humidity (25−80%). The authors suggested that the formation of a molecularly thin water layer on the sensor surface prompted the adsorption of NH3.
To validate the sensing mechanism, in situ IR analysis and theoretical calculations were conducted (Fig. 2 (e)). Upon NH3 adsorption, the IR spectra of PADS exhibited a blue shift in the N–H in-plane bending mode (from 1592 to 1596 cm−1) and C–N stretching (from 1396 to 1400 cm−1). Additionally, a red shift was observed in the amide N–H wagging (from 842 to 845 cm−1). These results were consistent with theoretical predictions, indicating a strong interaction between NH3 and PADS. This interaction involves electron donation from NH3 to PADS, which disrupts hole transport within PADS. Because charge transport in PADS is dominated by holes, the adsorption of NH3 decreased hole conduction, inducing a decrease in current after exposure to NH3.
In terms of sensing VOCs, detection remains particularly challenging due to the low chemical reactivity and trace-level concentrations of these compounds under ambient conditions [47]. To address this issue, the induction of specific intermolecular interactions between VOCs and selector molecules has emerged as a promising approach [48]. In this regard, dual-hydrogen bond donors have been shown to enhance the sensing performance of gas sensors by facilitating hydrogen bonding with VOCs, thereby improving both sensitivity and selectivity.
Therefore, efforts have been made to transduce chemical interactions into electrical signals by functionalizing dual-hydrogen bond donors onto conductive materials, such as carbon nanotubes (CNTs). Saetia et al. reported that hydrogen bond interactions between thiourea-based selectors and dimethyl methylphosphonate (DMMP), a nerve agent simulant, were successfully transduced by multi-walled carbon nanotubes (MWCNTs), resulting in a chemiresistive response [49]. In addition, CNTs functionalized with dual-hydrogen bond donors can effectively detect VOC markers for explosive compounds, such as cyclohexanone [50-52].
Yoon et al. developed a chemiresistive sensor for the detection of cyclohexanone using thiourea-based selector functionalized single-walled carbon nanotubes (SWCNTs) (Fig. 3 (a)) [50]. The sensor was fabricated by immobilizing a thiourea-functionalized polymer−SWCNT composite on a flexible poly(ethylene terephthalate) (PET) substrate. The thiourea-based selectors enabled selective recognition of cyclohexanone through hydrogen bonding. The binding properties were elucidated by 1H NMR titrations, which revealed a downfield shift in the α- and β-NH protons of the thiourea group upon the addition of cyclohexanone (Fig. 3 (b)). This result indicates the formation of collective hydrogen bonding of NH protons with cyclohexanone.
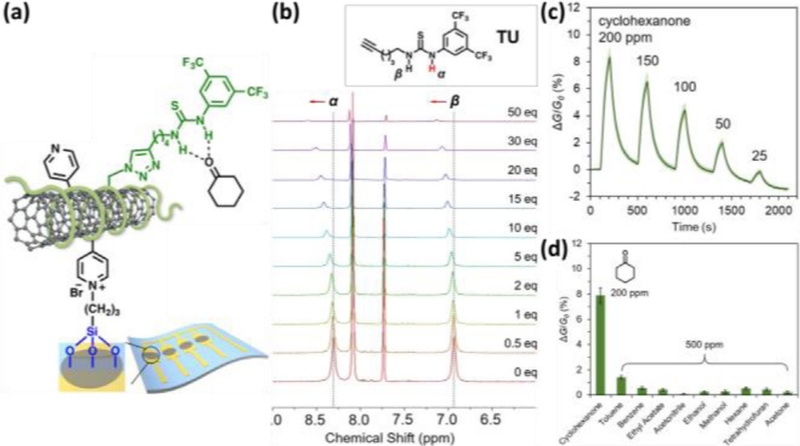
Chemiresistive gas sensor using thiourea. (a) Schematic illustration of chemiresistive cyclohexanone sensor on a flexible PET substrate. (b) 1H NMR chemical shifts of the thiourea NH protons (α and β) upon addition of increasing concentrations of cyclohexanone (0−50 equiv). (c) Dynamic response transitions of the sensor toward cyclohexanone in the concentration range of 25−200 ppm. (d) Average responses of the sensors to 200 ppm cyclohexanone and VOCs at a concentration of 500 ppm in N2. Reprinted with permission from Ref. [50], Copyright (2021) American Chemical Society.
The chemiresistive sensing properties of cyclohexanone were investigated over a concentration range of 25–200 ppm in dry N2 (Fig. 3 (c)). The sensor responses were calculated as ΔG/G0 (%) = (I−I0)/I0, where I and I0 are the current values upon exposures to gas and the baseline current, respectively. Upon exposure to 200 ppm of cyclohexanone vapor, the sensor exhibited an increase of 7.9 ± 0.6% in conductance. This increase in conductance indicates enhanced hole transport in SWCNTs, which is attributed to hydrogen bonding between the thiourea-based selectors and cyclohexanone. Moreover, the sensor exhibited high selectivity toward cyclohexanone, while negligible responses were observed to other VOCs, even at higher concentrations (Fig. 3 (d)). Under humid conditions (relative humidity of 55%), the sensors exhibited decreased conductance upon exposure to cyclohexanone. This is attributed to a hydrogen bonding complex of cyclohexanone and thiourea-based selectors, resulting in water adsorption on the SWCNT surface.
4. LIQUID SENSORS
4.1 General features of liquid sensors
Liquid sensors have primarily been developed to detect a broad range of analytes in aqueous media, with an increasing focus on biomedical and environmental applications [53,54]. As various chemical species in biological fluids have been analyzed to enable human health monitoring, wearable sensors have emerged as promising tools for healthcare applications, including continuous detection of biomarkers such as glucose, lactate, and electrolytes in biofluids like sweat and interstitial fluid [55,56]. Similarly, the rapid and on-site monitoring of toxic pollutants in water, including heavy metals and organic compounds, has also been extensively investigated as a strategy to protect human health from environmental contamination [57,58]. Subsequently, various liquid sensors have been developed to precisely and effectively recognize chemical analytes in fluidic environments.
The transduction of chemical changes into electrical signals is the fundamental operating principle of liquid sensors. The specific types of liquid sensors are determined by the transduction mechanism and nature of the electrical signals they generate, both of which are influenced by the chemical interaction between the analytes and sensing material [59]. Among these, chemiresistive and electrochemical sensors have been widely utilized for portable and on-site monitoring due to their simple and rapid operation [60,61]. Chemiresistive sensors measure changes in electrical resistance induced by interaction between target analytes and the sensing material. In the case of electrochemical sensors, redox reactions with the sensing material generate electrical signals such as current, voltage, or impedance, which are detected via electrode [62-64].
Despite these advantages, liquid sensors often exhibit limited selectivity, stability, or sensitivity under real-world conditions. To address these challenges, considerable efforts have been directed toward the rational design of sensing materials that can enhance sensor performance through tailored chemical interactions with target analytes [65,66]. In particular, the functionalization of molecular selectors has emerged as an effective strategy for governing selectivity toward specific analytes and improving the sensing capabilities of liquid sensors [67,68].
4.2 Dual-hydrogen bond donor-based liquid sensors
Several studies have reported the fabrication of sensor electrodes using dual-hydrogen bond donor-functionalized carbon nanomaterials to characterize the sensing performances of electrochemical sensors. Among these, a wide range of voltammetric sensors have been developed to detect various analytes, including heavy metal ions, dopamine, and phenolic compounds, by measuring oxidation or reduction currents [69-71].
Additionally, dual-hydrogen bond donors have been incorporated with redox-active units, allowing chemical interactions between dual-hydrogen bond donors and analytes to be monitored through shifts in the oxidation and reduction potentials. Zaleskaya et al. developed a squaramide-based ion pair sensor for anion detection using electrochemical measurements (Fig. 4 (a)) [72]. The sensor consists of squaramide integrated with benzo-18-crown-6 ether for cation recognition and a ferrocene unit for electrochemical signaling, enabling the simultaneous binding of both cations and anions. Notably, cation binding plays a crucial role in enhancing anion affinity by modulating the electronic properties of squaramide. To investigate the anion binding properties, UV-vis titrations were performed using a squaramide-based ion pair sensor upon the addition of tetrabutylammonium nitrate (TBANO3) in acetonitrile (Fig. 4 (b)). The results exhibited a gradual bathochromic shift of approximately 330–350 nm upon the addition of TBANO3. In the presence of cations such as Na+ or K+, this shift became more pronounced, indicating enhanced complex formation owing to the cation-induced electron-withdrawing effect. Furthermore, cyclic voltammetry measurements demonstrated that the sensor can effectively detect NO3−, with significantly enhanced signal responses in the presence of Na+ or K+ (Fig. 4 (c)). This improvement is attributed to a cation-induced electron-withdrawing effect that increases squaramide acidity and enhances anion binding affinity.
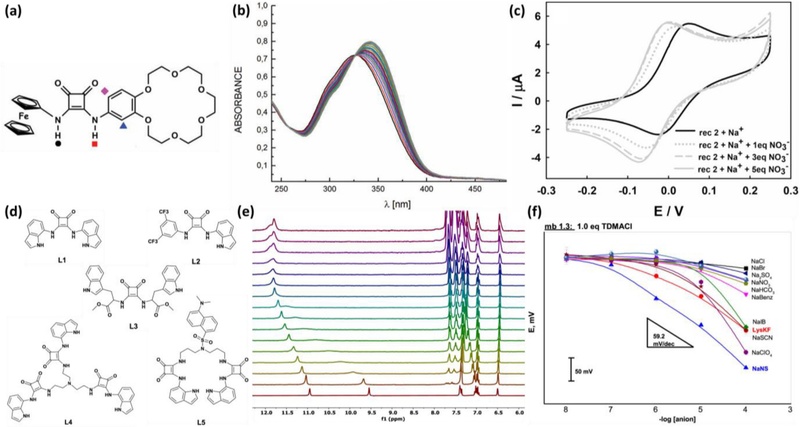
Electrochemical liquid sensors using squaramide. (a) Chemical structures of squaramide-based ion pair sensor. (b) UV-vis titrations of squaramide-based sensors upon additions of TBANO3 in the concentration range of 0−10 equiv. (c) Cyclic voltammograms recorded for 0.5 mM solution of squaramide-based sensors after adding 1, 3, and 5 equivalents of TBANO3 in acetonitrile. Reprinted with permission from Ref. [72], Copyright (2020) Royal Society of Chemistry. (d) Chemical structures of selective ionophores (L1 – L5). (e) 1H NMR titrations of L4 toward KF−. (f) Potentiometric responses of ISE containing L4. Reprinted with permission from Ref. [76], Copyright (2023) American Chemical Society.
Potentiometric sensing is another promising electrochemical sensing method. In this regard, dual-hydrogen bond donors have been employed as ionophores in the fabrication of ion-selective electrodes (ISEs) for use in potentiometric detection [73-76]. Picci et al. employed squaramide-based selectors as selective ionophores for the detection of nonsteroidal anti-inflammatory drug ions, including ketoprofen (KF) and naproxen (NS) anions in aqueous solution [76]. A series of acyclic squaramide-based selectors (L1 – L5) were synthe-sized by modifying the indole moiety, which introduced an additional hydrogen bond site through its NH proton (Fig. 4 (d)). The binding properties of these squaramide-based selectors toward KF− and NS− were systematically investigated by 1H NMR titration. The results revealed that NH protons of L4 significantly downfield-shifted upon addition of KF− (Fig. 4 (e)). This result indicates that L4 exhibited strong binding affinity toward KF− with a 1:2 binding stoichiometry. In contrast, the L3 selector showed a low binding affinity toward KF− and NS−, which is mainly attributed to the different directions of NH groups of indoles and squaramide, leading to the absence of cooperative hydrogen bonding between indole and dual-hydrogen bond donor.
ISEs consisting of squaramide-based selectors were prepared to characterize the potentiometric sensing responses and correlate them with the binding properties of the selectors. ISE containing L4 exhibited response slopes of −55.6 ± 2.0 mV/dec and −71.2 ± 2.0 mV/dec toward KF− and NS−, respectively (Fig. 4 (f)). The L3 containing ion-selective membrane exhibited low potentiometric responses toward both KF− and NS−. These results correlate with the binding affinity of squaramidebased selectors.
Choi et al. reported a novel sensing platform utilizing a chemiresistive transduction method to convert chemical interactions between selectors and anions into electrical signals [77]. Various dual-hydrogen bond donors, including thiourea, squaramide, and croconamide, have been used as anion selectors to facilitate hydrogen bonding with anions. To fabricate the chemiresistive sensor, dual-hydrogen bond donors were functionalized onto poly(4-vinylpyridine)-wrapped single-walled carbon nanotubes (P4VP-SWCNTs). This platform enabled the transduction of chemical interactions into electrical signals.
A systematic study was conducted to correlate anion binding affinities with chemiresistive sensing properties by comparing two squaramide-based selectors, (1) and (2), which differ in their electron-withdrawing substituents (Fig. 5 (a)). UV-vis titration analysis demonstrated that selector (2) shows a stronger binding affinity for acetate (AcO−) compared to selector (1), primarily due to the increased acidity of the NH protons induced by the directly-attached electron-withdrawing substituents (Fig. 5 (b)). Selective binding properties were further investigated, and the results revealed a weak binding affinity toward Cl−, Br−, and NO3−.
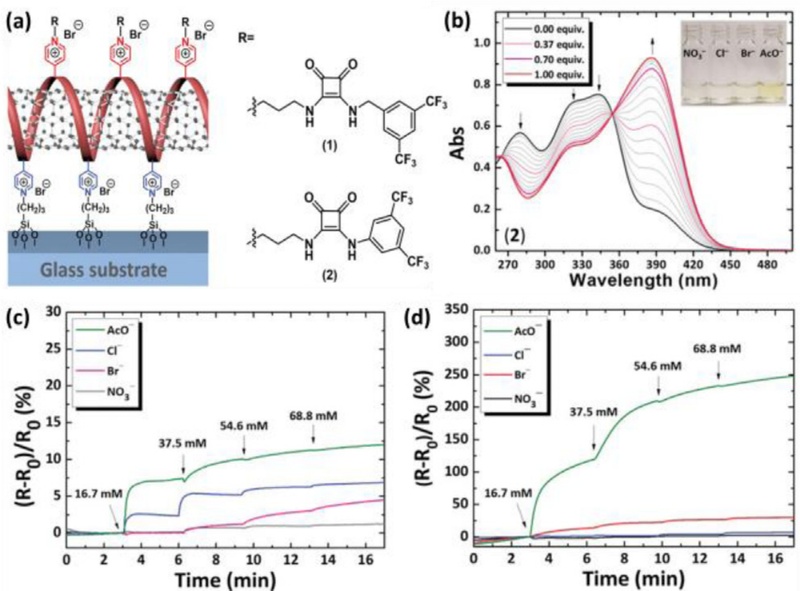
Chemiresistive liquid sensors using squaramide. (a) Schematic illustration of squaramide-based selector-functionalized P4VP-SWCNT. (b) UV-vis titrations of (2) toward AcO–. Dynamic response curves of (c) (1) functionalized P4VP-SWCNT and (d) (2) functionalized P4VP-SWCNT. Reprinted with permission from Ref. [77], Copyright (2020) Wiley-VCH.
To characterize the chemiresistive anion sensing properties, anion sensors were fabricated by functionalizing P4VP-SWCNTs with squaramide-based selectors. The sensor responses were evaluated using the normalized resistance transition, i.e., (R–R0)/R0 × 100 (%), where R0 and R are the baseline resistance and resistance after injecting anion solutions, respectively. The observed sensor responses correlated well with the anion binding affinities of the corresponding selectors. In particular, the anion sensor functionalized with selector (2) exhibited enhanced response and selectivity toward AcO– compared to the sensor consisting of selector (1) (Fig. 5 (c) and (d)).
Similarly, thiourea- and croconamide-based selectors were functionalized onto P4VP-SWCNTs for developing chemiresistive anion sensors [78,79]. These sensors demonstrated high selectivity toward AcO−, confirming the effect of dual-hydrogen bond donors on chemiresistive transduction of the selective anion binding into electrical signals.
Furthermore, Choi et al. developed chemiresistive anion sensors using dual-hydrogen bond donor-functionalized carbon nanotube fibers (CNTFs) (Fig. 6 (a)) [80,81]. Squaramide-based selectors were functionalized onto the CNTFs through either covalent or non-covalent approaches to investigate the influence of functionalization methods on the sensing properties (Fig. 6 (b)). The results showed that non-covalent functionalization enhanced sensing responses toward AcO− (Fig. 6 (c) and (d)). Subsequent studies using thiourea- and croconamide-based selectors revealed that squaramide-functionalized CNTFs exhibit improved sensing responses compared to other dual-hydrogen bond donors.
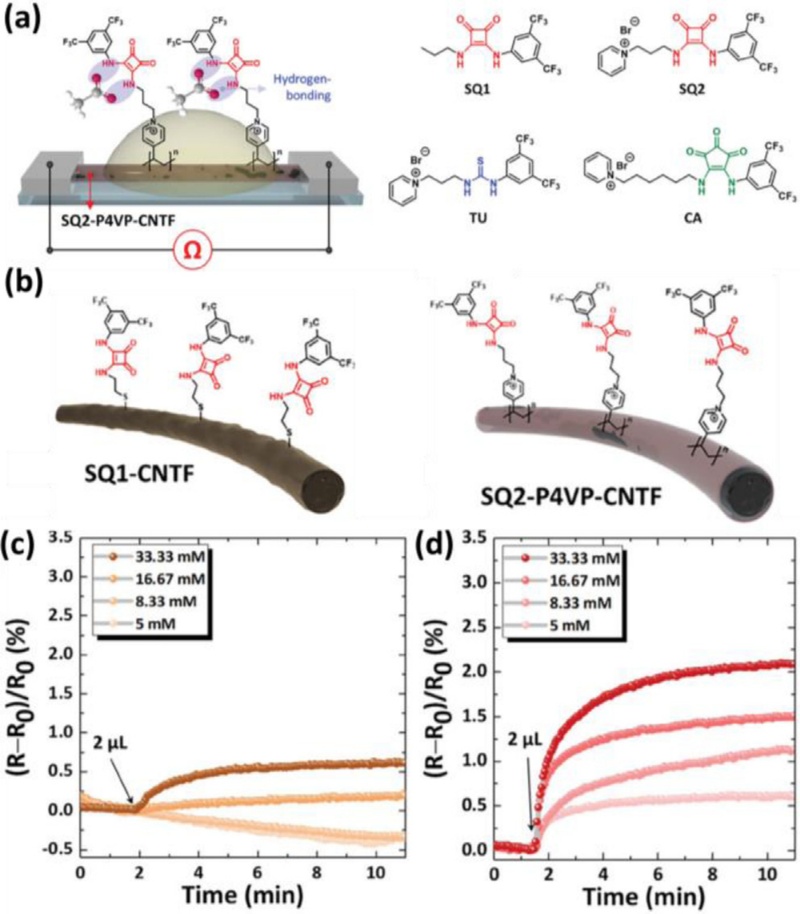
Chemiresistive liquid sensors using dual-hydrogen bond donors. (a) Schematic illustration of chemiresistive anion sensor and molecule structures of dual-hydrogen bond donor-based selectors. (b) Schematic illustrations of CNTFs functionalized with squaramide-based selectors through covalent (left) and non-covalent (right) approaches. Dynamic response transitions of (c) covalently functionalized CNTF and (d) non-covalently functionalized CNTF toward 5–33.33 mM AcO–. Reprinted with permission from Ref. [80], Copyright (2025) Wiley-VCH.
5. CONCLUSIONS AND PERSPECTIVES
This review focused on advanced chemical sensors that incorporate dual-hydrogen bond donor-based selectors, highlighting their design principles and sensing mechanisms in both gas- and liquid-phase sensing environments. By providing a mechanistic understanding of the transduction of chemical interactions into electrical signals, this study provides valuable insights into the rational design of dual-hydrogen bond donor-based selectors, thereby contributing toward improving sensing performance across diverse chemical environments.
The molecular-level structural tunability of dual-hydrogen bond donors provides tailored chemical interaction properties, leading to optimized selectivity and sensitivity toward various chemical analytes. This high structural flexibility facilitates the development of next-generation chemical sensors and provides a foundation for the construction of advanced sensor arrays that can be applied in IoT-integrated devices and wearable monitoring systems. These sensor systems enable rapid and continuous data acquisition, generating large volumes of multidimensional data in real time. By leveraging artificial intelligence-based data processing, such systems can be extended to mimic artificial olfaction and gustation.
Future research could focus on the systematic correlation between the selector structure and analyte selectivity, the development of multi-analyte sensor arrays, and the integration of dual-hydrogen bond donor-based sensors into wireless or implantable platforms for environmental and healthcare monitoring in real time. The ultimate goal of developing high-performance chemical sensors is to rationalize the artificial senses of smell and taste, enabling their application in futuristic humanoid robots.
Acknowledgments
This work was supported by the U.S. Army Combat Capabilities Development Command Soldier Center (DEVCOM SC) and International Technology Center Pacific (ITC-PAC) Global Research Project under contract FA520922C0008, and was conducted at Hanyang University. This work was also supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. RS-2024-00407282) and by the BK21 FOUR (Fostering Outstanding Universities for Research) project of the National Research Foundation of Korea Grant.
References
-
J.R. Sempionatto, J.A. Lasalde-Ramírez, K. Mahato, J. Wang, W. Gao, Wearable chemical sensors for biomarker discovery in the omics era, Nat. Rev. Chem. 6 (2022) 899–915.
[https://doi.org/10.1038/s41570-022-00439-w]

-
C. Wang, Z. Chen, C.L.J. Chan, Z. Wan, W. Ye, W. Tang, et al., Biomimetic olfactory chips based on large-scale monolithically integrated nanotube sensor arrays, Nat. Electron. 7 (2022) 157–167.
[https://doi.org/10.1038/s41928-023-01107-7]

-
E.K. Boahen, H. Kweon, H. Oh, J.H. Kim, H. Lim, D.H. Kim, Bio-Inspired Neuromorphic Sensory Systems from Intelligent Perception to Nervetronics, Adv. Sci. 12 (2025) 2409568.
[https://doi.org/10.1002/advs.202409568]

-
B. Lee, J.-O. Lee, J. Lee, I. Park, D.-S. Lee, Breath Gas Sensors for Diabetes and Lung Cancer Diagnosis, J. Sens. Sci. Technol. 32 (2023) 1–9.
[https://doi.org/10.46670/JSST.2023.32.1.1]

-
S.-H. Choi, J.-S. Lee, W.-J. Choi, J.-W. Seo, S.-J. Choi, Nanomaterials for IoT Sensing Platforms and Point-of-Care Applications in South Korea. Sensors 22 (2022) 610.
[https://doi.org/10.3390/s22020610]

-
I. Kim, E.D. Park, H.-S. Kim, S.-D. Han, Fabrication of Au-In2O3 Thin/Thick-Film Gas Sensors and Their Sensing Characteristics for Toxic Gases, J. Sens. Sci. Technol. 33 (2024) 516–524.
[https://doi.org/10.46670/JSST.2024.33.6.516]

-
K. Bae, E. Jo, Y. Kang, Sensitivity Enhancement of Benzene, Toluene, and Xylene Gas Sensors using Nanostructured TiO2: a Mini-Review, J. Sens. Sci. Technol. 34 (2025) 89–96.
[https://doi.org/10.46670/JSST.2025.34.2.89]

-
R. Paolesse, S. Nardis, D. Monti, M. Stefanelli, C. Di Natale, Porphyrinoids for Chemical Sensor Applications. Chem. Rev. 117 (2017) 2517–2583.
[https://doi.org/10.1021/acs.chemrev.6b00361]

-
D. Santos-Martins, S. Forli, Charting Hydrogen Bond Anisotropy, J. Chem. Theory Comput. 16 (2020) 2846–2856.
[https://doi.org/10.1021/acs.jctc.9b01248]

-
R.-B. Lin, B. Chen, Hydrogen-bonded organic frameworks: Chemistry and functions. Chem 8 (2022) 2114–2135.
[https://doi.org/10.1016/j.chempr.2022.06.015]

-
T. Lu, S.E. Wheeler, Origin of the Superior Performance of (Thio)Squaramides over (Thio)Ureas in Organocatalysis, Chem. Eur. J. 19 (2013) 15141–15147.
[https://doi.org/10.1002/chem.201302990]

-
J. Mishra, R. Kaur, A.K. Ganguli, N. Kaur, Urea/thiourea based dipodal nanoreceptors: Aqueous medium fluorescent chemosensor for Zn(II) and Hg(II) ions by photoinduced electron transfer, Microchem. J. 206 (2024) 111501.
[https://doi.org/10.1016/j.microc.2024.111501]

-
J. Gomez-Vega, J.M. Soto-Cruz, O. Juárez-Sánchez, H. Santacruz-Ortega, J.C. Gálvez-Ruiz, D.O. Corona-Martínez, et al., Tritopic Bis-Urea Receptors for Anion and Ion-Pair Recognition, ACS Omega 7 (2022) 22244–22255.
[https://doi.org/10.1021/acsomega.2c00935]

-
J.D. Lane, K.A. Jolliffe, Squaramides for colorimetric and fluorescent anion sensing, Org. Biomol. Chem. 21 (2023) 3226–3234.
[https://doi.org/10.1039/D3OB00069A]

-
V.B. Bregović, N. Basarić, Anion binding with urea and thiourea derivatives, Coord. Chem. Rev. 295 (2015) 80–124.
[https://doi.org/10.1016/j.ccr.2015.03.011]

-
G. Picci, R. Montis, V. Lippolis, C. Caltagirone, Squaramide-based receptors in anion supramolecular chemistry: insights into anion binding, sensing, transport and extraction, Chem. Soc. Rev. 53 (2024) 3952–3975.
[https://doi.org/10.1039/D3CS01165H]

-
G. Picci, R. Montis, A.M. Gilchrist, P.A. Gale, C. Caltagirone, Fluorescent and colorimetric sensors for anions: Highlights from 2020 to 2022, Coord. Chem. Rev. 501 (2024) 215561.
[https://doi.org/10.1016/j.ccr.2023.215561]

-
L. Escobar, P. Ballester, Molecular Recognition in Water Using Macrocyclic Synthetic Receptors, Chem. Rev. 121 (2021) 2445–2514.
[https://doi.org/10.1021/acs.chemrev.0c00522]

-
G.T. Williams, C.J. Haynes, M. Fares, C. Caltagirone, J.R. Hiscock, P.A. Gale, Advances in applied supramolecular technologies, Chem. Soc. Rev. 50 (2021) 2737–2763.
[https://doi.org/10.1039/D0CS00948B]

-
P. Gilli, G. Gilli, Hydrogen bond models and theories: The dual hydrogen bond model and its consequences, J. Mol. Struct. 972 (2010) 2–10.
[https://doi.org/10.1016/j.molstruc.2010.01.073]

-
J. Ho, V.E. Zwicker, K.K. Yuen, K.A. Jolliffe, Quantum Chemical Prediction of Equilibrium Acidities of Ureas, Deltamides, Squaramides, and Croconamides, J. Org. Chem. 82 (2017) 10732–10736.
[https://doi.org/10.1021/acs.joc.7b02083]

-
D.E. Gómez, L. Fabbrizzi, M. Licchelli, E. Monzani, Urea vs. thiourea in anion recognition, Org. Biomol. Chem. 3 (2005) 1495–1500.
[https://doi.org/10.1039/B500123D]

-
R.I. Storer, C. Aciro, L.H. Jones, Squaramides: physical properties, synthesis and applications, Chem. Soc. Rev. 40 (2011) 2330–2346.
[https://doi.org/10.1039/c0cs00200c]

-
T.J. Auvil, A.G. Schafer, A.E. Mattson, Design Strategies for Enhanced Hydrogen-Bond Donor Catalysts, Eur. J. Org. Chem. 2014 (2014) 2633–2646.
[https://doi.org/10.1002/ejoc.201400035]

-
A. Jeppesen, B.E. Nielsen, D. Larsen, O.M. Akselsen, T.I. Sølling, T. Brock-Nannestad, et al., Croconamides: a new dual hydrogen bond donating motif for anion recognition and organocatalysis, Org. Biomol. Chem. 15 (2017) 2784–2790.
[https://doi.org/10.1039/C7OB00441A]

-
V.E. Zwicker, K.K. Yuen, D.G. Smith, J. Ho, L. Qin, P. Turner, et al., Deltamides and Croconamides: Expanding the Range of Dual H-bond Donors for Selective Anion Recognition, Chem. Eur. J. 24 (2018) 1140–1150.
[https://doi.org/10.1002/chem.201704388]

-
H. Lee, J.-S. Lee, G.-W. Kwak, J. Kim, K.-M. Kim, D.G. Kang, et al., Carbide-Induced Thermal Shock Synthesis of High-Entropy Alloy Nanoparticles Anchored on WO3 Nanofibers for High-Performance Gas Sensors, ACS Nano 19 (2025) 18095–18107.
[https://doi.org/10.1021/acsnano.4c11149]

-
J.-W. Seo, J.-H. Park, J.-W. Jung, S.-J. Choi, Dual-catalytic activation of Pt and MoSₓOᵧ on carbon nanofibers for NO2 sensors, Sens. Actuators B Chem. 412 (2024) 135750.
[https://doi.org/10.1016/j.snb.2024.135750]

-
D.-H. Kim, J.-S. Lee, H.J. Park, I.-D. Kim, S.-J. Choi, Temperature-Dependent n-to-p-Type Transition of 2D Mn Oxide Nanosheets toward NO2 for Flexible Gas Sensor Application, ACS Sens. 8 (2022) 280–288.
[https://doi.org/10.1021/acssensors.2c02181]

-
Y. Ha, D.G. Jung, J. Lee, Y. Yang, U. Han, D. Jung, Highly Sensitive Platinum-Decorated Tungsten Oxide for Ultra-Low-Concentration Hydrogen Detection, J. Sens. Sci. Technol. 33 (2024) 504–509.
[https://doi.org/10.46670/JSST.2024.33.6.504]

-
J.W. Baek, E. Shin, J. Lee, D.-H. Kim, S.-J. Choi, I.-D. Kim, Present and Future of Emerging Catalysts in Gas Sensors for Breath Analysis, ACS Sens. 10 (2025) 33–53.
[https://doi.org/10.1021/acssensors.4c02464]

-
M. Rodríguez-Aguilar, L.D. de León-Martínez, P. Gorocica-Rosete, R. Pérez-Padilla, C.A. Domínguez-Reyes, J.A. Tenorio-Torres, et al., Application of chemoresistive gas sensors and chemometric analysis to differentiate the fingerprints of global volatile organic compounds from diseases. Preliminary results of COPD, lung cancer and breast cancer, Clin. Chim. Acta 518 (2021) 83–92.
[https://doi.org/10.1016/j.cca.2021.03.016]

-
S.-Y. Jeong, Highly Sensitive and Fast-Responding Ethanol Sensor Using Au Doped-In2O3 Hollow Spheres, J. Sens. Sci. Technol. 33 (2024) 242–247.
[https://doi.org/10.46670/JSST.2024.33.5.242]

-
D.G. Jung, J. Lee, D.H. Jung, W.O. Lee, B.S. Park, D. Jung, Development of a MEMS-Based H2S Sensor with a High Detection Performance and Fast Response Time, J. Sens. Sci. Technol. 32 (2023) 207–212.
[https://doi.org/10.46670/JSST.2023.32.4.207]

-
D.H. Jeong, D.G. Jung, D. Jung, Fabrication and Evaluation of Single Layer Graphene/SnO2 Based Gas Sensor for NO2 Detection, J. Sens. Sci. Technol. 33 (2024) 493–498.
[https://doi.org/10.46670/JSST.2024.33.6.493]

-
H. Zhang, Z. Zhang, Z. Li, H. Han, W. Song, J. Yi, A chemiresistive-potentiometric multivariate sensor for discriminative gas detection, Nat. Commun. 14 (2023) 3495.
[https://doi.org/10.1038/s41467-023-39213-x]

-
Z. Zhai, X. Zhang, X. Hao, B. Niu, C. Li, Metal–Organic Frameworks Materials for Capacitive Gas Sensors, Adv. Mater. Technol. 6 (2021) 2100127.
[https://doi.org/10.1002/admt.202100127]

-
R. Mesin, C.-S. Chu, Optical Dual Gas Sensor for Simultaneous Detection of Nitric Oxide and Oxygen, Chemosensors 11 (2023) 454.
[https://doi.org/10.3390/chemosensors11080454]

-
H. Shin, J. Ahn, D.-H. Kim, J. Ko, S.-J. Choi, R.M. Penner, et al., Rational design approaches of two-dimensional metal oxides for chemiresistive gas sensors: A comprehensive review, MRS Bull. 46 (2021) 1080–1094.
[https://doi.org/10.1557/s43577-021-00218-w]

-
T.W. Ha, D.-Y. Lim, C.-H. Lee, High-Temperature Stable NOx Sensor for Exhaust Gas Monitoring in Automobiles, J. Sens. Sci. Technol. 33 (2024) 439–447.
[https://doi.org/10.46670/JSST.2024.33.6.439]

-
G.G. Yang, D.-H. Kim, S. Samal, J. Choi, H. Roh, C.E. Cunin, et al., Polymer-Based Thermally Stable Chemiresistive Sensor for Real-Time Monitoring of NO2 Gas Emission, ACS Sens. 8 (2023) 3687–3692.
[https://doi.org/10.1021/acssensors.3c01530]

-
H. Mei, J. Peng, T. Wang, T. Zhou, H. Zhao, T. Zhang, et al., Overcoming the Limits of Cross-Sensitivity: Pattern Recognition Methods for Chemiresistive Gas Sensor Array, Nano Micro Lett. 16 (2024) 269.
[https://doi.org/10.1007/s40820-024-01489-z]

-
P.M. Bulemo, D.-H. Kim, H. Shin, H.-J. Cho, W.-T. Koo, S.-J. Choi, et al., Selectivity in Chemiresistive Gas Sensors: Strategies and Challenges, Chem. Rev. 125 (2025) 4111–4183.
[https://doi.org/10.1021/acs.chemrev.4c00592]

-
A.I. Daud, K.A.A. Wahid, W.M. Khairul, Room-temperature operated cyano-terminated ethynylated-thiourea as a resistive-type carbon dioxide (CO2) gas sensor, Org. Electron. 70 (2019) 32–41.
[https://doi.org/10.1016/j.orgel.2019.04.001]

-
D. Han, W. Yang, F. Gu, Z. Wang, A low temperature NO2 gas sensor based metal-thiourea complex nanowires, Sens. Actuators B Chem. 255 (2018) 1139–1146.
[https://doi.org/10.1016/j.snb.2017.08.083]

-
J. Zhou, H. Lin, X.-F. Cheng, J. Shu, J.-H. He, H. Li, et al., Ultrasensitive and robust organic gas sensors through dual hydrogen bonding, Mater. Horiz. 6 (2019) 554–562.
[https://doi.org/10.1039/C8MH01098F]

-
M. Khatib, H. Haick, Sensors for Volatile Organic Compounds, ACS Nano 16 (2022) 7080–7115.
[https://doi.org/10.1021/acsnano.1c10827]

-
R.J. Rath, S. Talebian, J. Giaretta, S. Naficy, F. Dehghani, Organic-Based Chemiresistive Sensors for Detection of Water-Soluble Gases: Strategies and Roadmap for Enhancing Sensing Performance, Adv. Funct. Mater. 35 (2025) 2417323.
[https://doi.org/10.1002/adfm.202417323]

-
K. Saetia, J.M. Schnorr, M.M. Mannarino, S.Y. Kim, G.C. Rutledge, T.M. Swager, et al., Spray-Layer-by-Layer Carbon Nanotube/Electrospun Fiber electrodes for Flexible Chemiresistive Sensor Applications, Adv. Funct. Mater. 24 (2014) 492–502.
[https://doi.org/10.1002/adfm.201302344]

-
B. Yoon, S.-J. Choi, T.M. Swager, G.F. Walsh, Flexible Chemiresistive Cyclohexanone Sensors Based on Single-Walled Carbon Nanotube–Polymer Composites, ACS Sens. 6 (2021) 3056–3062.
[https://doi.org/10.1021/acssensors.1c01076]

-
K.M. Frazier, T.M. Swager, Robust Cyclohexanone Selective Chemiresistors Based on Single-Walled Carbon Nanotubes, Anal. Chem. 85 (2013) 7154–7158.
[https://doi.org/10.1021/ac400808h]

-
J.M. Schnorr, D. van der Zwaag, J.J. Walish, Y. Weizmann, T.M. Swager, Sensory Arrays of Covalently Functionalized Single-Walled Carbon Nanotubes for Explosive Detection, Adv. Funct. Mater. 23 (2013) 5285–5291.
[https://doi.org/10.1002/adfm.201300131]

-
Y.-J. Kim, S.-H. Choi, S.-J. Choi, Enhanced Electrochemical Detection of Heavy Metal Ions via Post-Synthetic Schiff Base Modification of MWCNT-MOF Composites, J. Sens. Sci. Technol. 33 (2024) 366–372.
[https://doi.org/10.46670/JSST.2024.33.5.366]

-
S.-J. Choi, S. Savagatrup, Y. Kim, J.H. Lang, T.M. Swager, Precision pH Sensor Based on WO3 Nanofiber-Polymer Composites and Differential Amplification, ACS Sens. 4 (2019) 2593–2598.
[https://doi.org/10.1021/acssensors.9b01579]

-
P. Mehrotra, Biosensors and their applications – A review, J. Oral Biol. Craniofac. Res. 6 (2016) 153–159.
[https://doi.org/10.1016/j.jobcr.2015.12.002]

-
H. Choi, S.-W. Lee, Metal Oxide-Based Electrochemical Non-Enzymatic Glucose Biosensors: A Mini-Review, J. Sens. Sci. Technol. 34 (2025) 105–115.
[https://doi.org/10.46670/JSST.2025.34.2.105]

-
T. Hu, Q. Lai, W. Fan, Y. Zhang, Z. Liu, Advances in Portable Heavy Metal Ion Sensors, Sensors 23 (2023) 4125.
[https://doi.org/10.3390/s23084125]

-
P. Samanta, A.V. Desai, S. Let, S.K. Ghosh, Advanced Porous Materials for Sensing, Capture and Detoxification of Organic Pollutants toward Water Remediation, ACS Sustain. Chem. Eng. 7 (2019) 7456–7478.
[https://doi.org/10.1021/acssuschemeng.9b00155]

-
Z. Meng, R.M. Stolz, L. Mendecki, K.A. Mirica, Electrically-Transduced Chemical Sensors Based on Two-Dimensional Nanomaterials, Chem. Rev. 119 (2019) 478–598.
[https://doi.org/10.1021/acs.chemrev.8b00311]

-
H. Sohrabi, A. Hemmati, M.R. Majidi, S. Eyvazi, A. Jahanban-Esfahlan, B. Baradaran, et al., Recent advances on portable sensing and biosensing assays applied for detection of main chemical and biological pollutant agents in water samples: A critical review, TrAC Trends Anal. Chem. 143 (2021) 116344.
[https://doi.org/10.1016/j.trac.2021.116344]

-
M. Kilani, G. Mao, Nanomaterials-Enabled Sensors for Detecting and Monitoring Chemical Warfare Agents, Small 21 (2025) 2409984.
[https://doi.org/10.1002/smll.202409984]

-
H.A. Saputra, Electrochemical sensors: basic principles, engineering, and state of the art, Monatsh. Chem. 154 (2023) 1083–1100.
[https://doi.org/10.1007/s00706-023-03113-z]

-
C. Bae, M. Han, Y. Song, H. Lee, M. Lee, J.-J. Park, et al., Research on an Electrochemical Salinity Sensor with Indium-Tin-Oxide Conductive Ceramic Electrodes, J. Sens. Sci. Technol. 34 (2025) 37–43.
[https://doi.org/10.46670/JSST.2025.34.1.37]

-
P.M. Rajaitha, R. Jana, S. Hajra, S. Panda, H.J. Kim, Review on CNT-Based Electrode Materials for Electrochemical Sensing of Ascorbic Acid, J. Sens. Sci. Technol. 32 (2023) 131–139.
[https://doi.org/10.46670/JSST.2023.32.3.131]

-
O.S. Kwon, H.S. Song, T.H. Park, J. Jang, Conducting Nanomaterial Sensor Using Natural Receptors, Chem. Rev. 119 (2018) 36–93.
[https://doi.org/10.1021/acs.chemrev.8b00159]

-
R. Kubota, Y. Sasaki, T. Minamiki, T. Minami, Chemical Sensing Platforms Based on Organic Thin-Film Transistors Functionalized with Artificial Receptors, ACS Sens. 4 (2019) 2571–2587.
[https://doi.org/10.1021/acssensors.9b01114]

-
S. Hasanova, A. Ait Lahcen, E. Zor, Recent advances in molecular imprinting techniques for the electrochemical analysis of chiral compounds, J. Pharm. Biomed. Anal. Open 4 (2024) 100046.
[https://doi.org/10.1016/j.jpbao.2024.100046]

-
J.J. Lavigne, E.V. Anslyn, Sensing A Paradigm Shift in the Field of Molecular Recognition: From Selective to Differential Receptors, Angew. Chem. Int. Ed. 40 (2001) 3118–3130.
[https://doi.org/10.1002/1521-3773(20010903)40:17<3118::AID-ANIE3118>3.0.CO;2-Y]

-
N.F. Atta, A. Galal, A.R.M. El-Gohary, Novel method of one pot preparation of thiourea self-assembled monolayers over gold nanoparticles-carbon nanotubes composite for sensing application of phenolic compounds, J. Electroanal. Chem. 902 (2021) 115795.
[https://doi.org/10.1016/j.jelechem.2021.115795]

-
V.B. Jaryal, S. Kumar, D. Singh, N. Gupta, Thiourea-Modified Multiwalled Carbon Nanotubes as Electrochemical Biosensor for Ultra-Precise Detection of Dopamine, Chem-NanoMat 10 (2024) e202300637.
[https://doi.org/10.1002/cnma.202300637]

-
S. Sapari, N.H.A. Razak, S.A. Hasbullah, L.Y. Heng, K.F. Chong, L.L. Tan, A regenerable screen-printed voltammetric Hg(II) ion sensor based on tris-thiourea organic chelating ligand grafted graphene nanomaterial, J. Electroanal. Chem. 878 (2020) 114670.
[https://doi.org/10.1016/j.jelechem.2020.114670]

-
M. Zaleskaya, D. Jagleniec, M. Karbarz, Ł. Dobrzycki, J. Romański, Squaramide based ion pair receptors possessing ferrocene as a signaling unit, Inorg. Chem. Front. 7 (2020) 972–983.
[https://doi.org/10.1039/C9QI01452G]

-
L. Li, P. Du, Y. Duan, Y. Zhang, Y. Li, Y. Qian, et al., Hydrogen Bond-Based Macrocyclic and Tripodal Neutral Ionophores for Highly Selective Polymeric Membrane Sulfate-Selective Electrodes, ACS Sens. 6 (2020) 245–251.
[https://doi.org/10.1021/acssensors.0c02231]

-
Y. Liu, Y. Qin, D. Jiang, Squaramide-based tripodal ionophores for potentiometric sulfate-selective sensors with high selectivity, Analyst 140 (2015) 5317–5323.
[https://doi.org/10.1039/C5AN00259A]

-
L. Fan, T. Xu, J. Feng, Z. Ji, L. Li, X. Shi, et al., Tripodal squaramide Derivative as a Neutral Chloride Ionophore for Whole Blood and Sweat Chloride Measurement, Electroanalysis 32 (2020) 805–811.
[https://doi.org/10.1002/elan.201900693]

-
G. Picci, S. Farotto, J. Milia, C. Caltagirone, V. Lippolis, M.C. Aragoni, et al., Potentiometric Sensing of Nonsteroidal Painkillers by Acyclic Squaramide Ionophores, ACS Sens. 8 (2023) 3225–3239.
[https://doi.org/10.1021/acssensors.3c00981]

-
S.-J. Choi, B. Yoon, J.D. Ray, A. Netchaev, L.C. Moores, T.M. Swager, Chemiresistors for the Real-Time Wireless Detection of Anions, Adv. Funct. Mater. 30 (2020) 1907087.
[https://doi.org/10.1002/adfm.201907087]

-
S.-J. Choi, B. Yoon, S. Lin, T.M. Swager, Functional Single-Walled Carbon Nanotubes for Anion Sensing, ACS Appl. Mater. Interfaces 12 (2020) 28375–28382.
[https://doi.org/10.1021/acsami.0c03813]

-
B. Yoon, S.-J. Choi, Selective acetate recognition and sensing using SWCNTs functionalized with croconamides, Sens. Actuators B Chem. 346 (2021) 130461.
[https://doi.org/10.1016/j.snb.2021.130461]

-
S.-H. Choi, J.-S. Lee, S. Lee, H.S. Jeong, S.-J. Choi, Dual-Hydrogen Bond Donor-Functionalized Carbon Nanotube Fibers: Enhancing Anion-Sensing Performance Through Functionalization Approaches, Small 21 (2025) 2405070.
[https://doi.org/10.1002/smll.202405070]

-
S.-H. Choi, J.-S. Lee, W.-J. Choi, S. Lee, H.S. Jeong, S.-J. Choi, Selective acetate detection using functional carbon nanotube fiber, J. Sens. Sci. Technol. 30 (2021) 357–363.
[https://doi.org/10.46670/JSST.2021.30.6.357]


Seung-Ho Choi is currently pursuing a Ph.D. in the Division of Materials Science and Engineering at Hanyang University (HYU), under the supervision of Prof. Seon-Jin Choi. He received his B.S. degree in Materials Science and Engineering from HYU in 2020. His research focuses on organic selectors, selector-functionalized nanomaterials, and their applications in electronic devices.

Seon-Jin Choi is an Associate Professor in the Division of Materials Science and Engineering at Hanyang University (HYU). He received his B.S. degree in Materials Science and Engineering from HYU in 2010. He earned his M.S. degree in Electrical Engineering (2012) and Ph.D. degree in Materials Science and Engineering (2016), both from the Korea Advanced Institute of Science and Technology (KAIST). Following his doctoral studies, he conducted postdoctoral research in the Department of Chemistry at the Massachusetts Institute of Technology (MIT) from 2017 to 2019. His research interests include design, processing, and characterization of chemical sensors using multidimensional nanostructures for application in environmental and health monitoring.