
Triboelectrification-Based Ultrasound Detection
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Ultrasound-to-electric signal conversion based on the triboelectric effect has emerged as a promising strategy for overcoming the inherent limitations of conventional piezoelectric ultrasound sensors, including structural complexity, high fabrication costs, and material constraints. In this review, we comprehensively examine the fundamental mechanisms and recent advances in ultrasound detection and energy harvesting, with a focus on triboelectric nanogenerators (TENGs). Particular focus is placed on ultrasound-driven TENGs (US-TENG), addressing key developments in device architecture, material engineering, and packaging techniques that facilitate effective transduction of ultrasonic excitation into mechanical deformation and subsequent electrical output via electrostatic induction and contact electrification. In addition, we discuss the potential of US-TENGs for integration into self-powered sensors and energy modules that are applicable to implantable medical devices and wearable electronics, highlighting their promise for overcoming the drawbacks of existing sensor platforms. This review offers a current and comprehensive overview of TENG-based ultrasound sensing technologies, highlighting their technological significance, current challenges, and prospects in the fields of sensing and energy conversion.
Keywords:
Ultrasound, Implantable medical device, Triboelectric effect, Energy conversion, Sustainable energy, Biomedical device1. INTRODUCTION
Since it was first reported in 2012, the triboelectric nanogenerator (TENG), which is based on the triboelectric effect, has attracted attention as an innovative energy conversion technology that can harvest mechanical energy from the surrounding environment and convert it into an electrical signal [1]. This device generates a charge by utilizing contact electrification and electrostatic induction, which occur when two different materials come into contact and separate. TENGs offer considerable freedom in terms of material selection and design flexibility, allowing for diverse material combinations and structural configurations. They also have significant advantages in terms of their low cost, large-area fabrication, and structural scalability. Owing to these features, TENGs have emerged as a key technology for implementing self-powered systems in various fields, including low-power sensors and implantable medical devices (IMDs) [2-6]. Notably, TENGs are well suited for sensor applications because they can convert mechanical vibrations across a wide range of frequencies into electrical signals. Recent studies have further demonstrated the ability of TENGs to detect high-frequency signals [7-8].
Leveraging this capability to transduce various mechanical vibrations in the environment into electrical signals, extensive research is underway to develop self-powered sensors that respond to everyday vibrations, such as wind, speech, and ambient noise [9-14]. For instance, a research group led by Professor Jun Chen demonstrated that TENGs can effectively convert even subtle environmental vibrations, thereby establishing their potential as sensors and energy-harvesting devices [15]. These studies confirmed that TENGs can detect low-frequency signals and convert energy across various frequency bands.
Recently, TENGs have even been reported to detect high-frequency vibrations such as ultrasound, which exceed the human audible range. This is a unique characteristic that distinguishes them from wearable devices, as ultrasound-driven TENGs (US-TENGs) exhibit substantially higher electrical signal conversion performance. This significantly enhances their potential for direct use as sensors or power sources in devices such as IMDs [16].
As opposed to conventional piezoelectric mechanisms, triboelectric-based ultrasonic sensors have unique advantages, including material selection flexibility, low cost, large-area fabrication, and the capability for non-contact energy conversion. Owing to these differences, the introduction of US-TENGs in Science in 2019 received significant attention, as their conversion characteristics were shown to vary according to the distance between the ultrasound source and device, as well as their interactions with biological tissue [17]. This research moves beyond ultrasound detection through TENGs to offer new insights into the influence of ultrasound energy transmission paths and structural characteristics on conversion behavior, thereby suggesting novel research directions. Although research on US-TENGs is still in its early stages, extensive efforts are being made to improve their conversion performance. These include the application of dual-electrode structures, development of theoretical frameworks, and creation of packaging technologies using flexible materials as alternatives to titanium (Ti) metal. In particular, research focusing on packaging materials that consider ultrasonic properties is crucial for enhancing the reliability and practical utility of sensors.
In this manner, US-TENGs harness the ability of ultrasound to propagate through biological tissues, converting externally delivered acoustic energy into mechanical vibrations, and subsequently, into electrical signals. These capabilities make US-TENGs promising for the real-time (or on-demand) charging and self-powered operation of IMDs and in vivo sensors. Moreover, related applications, including transient sensors and biodegradable electronics fabricated from resorbable materials, have recently attracted increasing interest [18-22].
In this review, we comprehensively examine the basic signal conversion principles and structures of US-TENGs, discuss recent research advances in this field, and explore their applications and prospects in IMDs.
2. TRIBOELECTRIFICATION SYSTEMS FOR ULTRASOUND DETECTION
2.1 Emergence of triboelectrification ultrasound detection systems
Fig. 1 illustrates the first report on the vibrating and implantable triboelectric generator (VI-TEG), a device that can detect ultrasonic energy and convert it into electrical signals [17]. Fig. 1 (a) shows a schematic of the VI-TEG operating environment and device architecture. The device, which is designed to function as a power source for IMDs, is typically implanted within 10 mm beneath the skin, where it senses ultrasound and charges components such as batteries, thereby serving as the primary power unit for implantable devices.
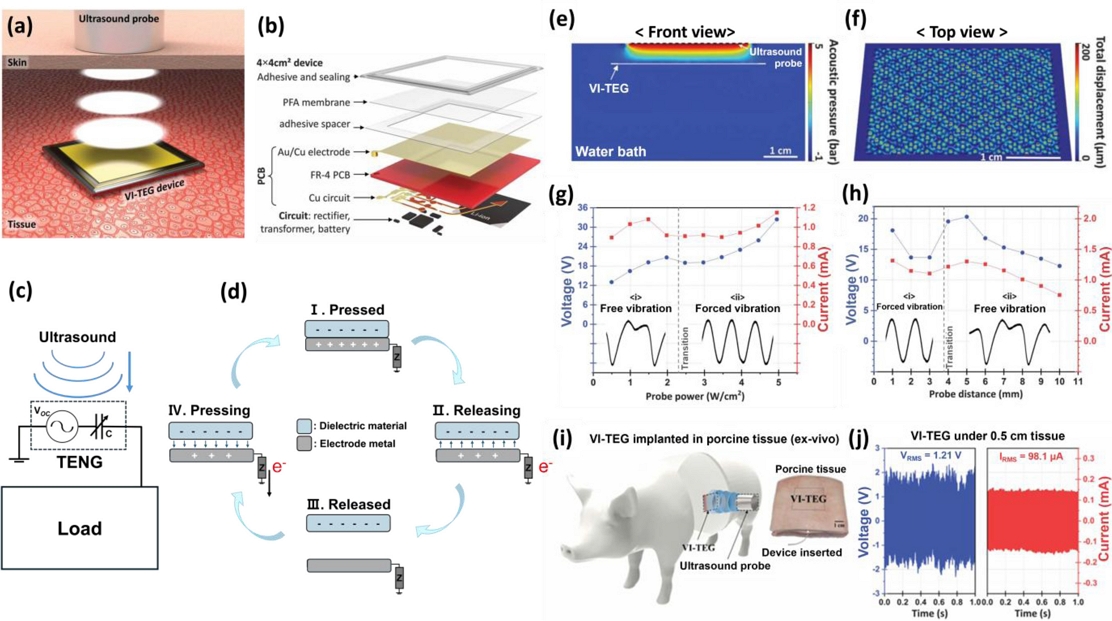
Emergence of the triboelectrification ultrasound detection system. (a) Schematic of the VI-TEG during ultrasound detection. Adapted from Ref. [12]. (b) Enlarged device structure of the VI-TEG. (c) Equivalent circuit of a single-electrode mode TENG. Adapted from Ref. [16]. (d) Energy conversion principle of the single-electrode mode TENG. (e) Simulation of ultrasound propagation in water and the VI-TEG using the FEM. (f) FEM simulation of the VI-TEG membrane vibration in water under 20 kHz ultrasound excitation. (g) Maximum voltage and current as a function of ultrasound output intensity, measured at a fixed distance of 5 mm. Insets i and ii show voltage waveforms measured under output intensities ranging from 0.5 to 2.0 W/cm² and from 2.5 to 5.0 W/cm², respectively. (h) Maximum voltage and current as a function of the distance from the ultrasound transducer, measured under a constant intensity of 3 W/cm². Insets i and ii show voltage waveforms measured at distances of 2 mm and 5 mm from the transducer, respectively. (i) Working illustration of the VI-TEG under an in vivo environment. (j) VI-TEG performance under 0.5 cm porcine tissue at 20 kHz and 1 W/cm².
As shown in Fig. 1 (b), the device incorporates a 50-μm-thick perfluoroalkoxy (PFA) membrane that vibrates under ultrasound and serves as both the encapsulation and triboelectric layer. An Au-coated copper electrode, designed as a 3.6 cm × 3.6 cm PCB, is positioned within the structure. A spacer maintains an 80 μm air gap between the immovable and vibrating layers, ensuring the physical separation required for triboelectric contact. The backside of the device integrates a rectifying circuit, maintaining a total device thickness of < 1 mm. Compared with conventional piezoelectric devices, the VI-TEG can be fabricated with a thinner form factor, yields a higher open-circuit voltage under comparable excitation, offers broader material choices, and operates without additional device-specific modifications because triboelectric transduction requires no poling or crystal-orientation control. These characteristics enable reliable operation in complex environments, including in vivo settings.
Figs. 1 (c) and (d) show an equivalent circuit diagram and the working principle of the VI-TEG [23]. The device operates in single-electrode contact (SEC) mode. When ultrasound induces vibration, the PFA membrane contacts the electrode, generating negative charges on the internal surface of the membrane through contact electrification effects. These negative charges produce electrostatic induction that reduces the main electrode potential relative to the reference electrode, attracting holes in the electrode and producing a current pulse in the circuit. When the membrane separates from the electrode, the potential increases, releasing holes and generating pulses of opposite polarities.
Acoustic and mechanical finite element method (FEM) simulations are illustrated in Fig. 1 (e), demonstrating that incident ultrasound waves are mostly reflected at the VI-TEG interface, thereby inhibiting their transmission through the device. When the VI-TEG is driven by a continuous 20 kHz ultrasound excitation, the membrane responds with complex multimodal vibration patterns, as shown in Fig. 1 (f). These oscillations give rise to multiple nodes and antinodes that are distributed across the membrane surface, moving in an alternating up–down pattern corresponding to the vibrational cycles.
The FEM simulation showed that when ultrasound energy penetrates through the encapsulation and reaches the metal electrode, part of the acoustic wave is reflected, inducing mechanical vibration primarily in the triboelectric membrane layer. The existence of micro-air gaps on the membrane surface enhances the localized sound pressure and facilitates distinct contact–separation cycles between layers, as confirmed by the sinusoidal electrical outputs. This ultrasound behavior enables efficient energy conversion, where the vibration amplitude and resulting signal strength are highly dependent on both the membrane structure and acoustic intensity. The coexistence of multiple vibration modes under these operating conditions shows the complex coupling between the acoustic excitation and membrane dynamics in the VI-TEG.
Ultrasound, as a wave-based energy source, exhibits operational characteristics that vary with both intensity and distance from the device. These variables are key determinants of efficiency and the process by which the device converts ultrasonic energy into electrical signals. The triboelectric signal varies with the acoustic radiation pressure, increasing with the ultrasound power (Fig. 1 (g)) and decreasing with an increasing distance from the ultrasound probe (Fig. 1 (h)). Two distinct electrical signal shapes are observed, corresponding to different types of PFA membrane vibrations. Radiation pressure forces the membrane against the electrode, altering the contact and vibration. At higher ultrasound power, strong radiation pressure drives membrane–electrode contact at the 20 kHz excitation frequency, producing a 20 kHz sinusoidal signal. At longer distances or lower ultrasound power, weaker pressure allows the membrane to rebound on the electrode, generating a 10 kHz signal with a 20 kHz harmonic. Because the triboelectric voltage is proportional to the membrane–electrode distance, the rebound every two ultrasound oscillations allows greater separation, maximizing the voltage.
As the acoustic radiation pressure decreases, the rebound amplitude also decreases. An optimal configuration exists in which, despite lower ultrasound power or a longer distance than that in the forced-vibration mode, the free-vibration mode can yield higher voltage and current peaks. This demonstrates that both the ultrasound intensity and device position are crucial factors for optimizing the power output.
Finally, to assess the viability of the VI-TEG as an IMD power source, Fig. 1 (i) shows the device operating in a pig skin environment that closely mimics human tissue. As shown in Fig. 1 (j), the device delivers an output of over 2.4 V and 156 μA at a 5 mm depth, despite the attenuation of acoustic pressure by tissue. This performance meets the minimum power requirements (1–100 μW) for implantable devices such as pacemakers, confirming the VI-TEG as a promising early-stage technology for IMD power supply applications.
2.2 Advanced techniques for US-TENGs
The VI-TEG, as an initial implementation of the US-TENG, effectively converts ultrasonic energy into electrical signals for detection; however, achieving high conversion efficiency under limited ultrasound energy remains challenging [24]. Consequently, research is ongoing to enhance the conversion efficiency of TENG devices and improve their output performance. There are two primary approaches for increasing the conversion efficiency of TENGs: one involves modifying the device operating mode to achieve higher structural figures of merit (FOMs), thereby increasing the conversion rate; the other focuses on maximizing the density and pressure of the incident ultrasound on the device, thereby reducing energy loss.
Owing to the inherent properties of TENGs, which convert mechanical force into electrical signals through the triboelectrification and electrical induction effects, the conversion efficiency is highly influenced by the structural parameters of the device, such as the number and arrangement of electrodes. The comprehensive evaluation and quantitative expression of these influences are referred to as the FOM, which serves as a standardized metric for comparing device performances and can be calculated based on experimentally measured electrical outputs and structural design parameters.
Fig. 2 shows the FOM of the SEC mode TENG and compares it with that of the contact–separate (CS) mode TENG. In terms of the FOMs according to the TENG operating modes. Figs. 2 (a) and (b) illustrate the structures of the SEC mode and CS mode TENG devices [25,26]. In the SEC mode TENG, a pair of dielectric and metal electrodes face one another vertically, and the metal electrode operates by connecting to a reference electrode, playing the roles of the external circuit and ground. In the CS mode TENG, a dielectric is attached to one of the two vertically aligned metal electrodes, and an external circuit is connected between the electrodes to transmit electrical signals.
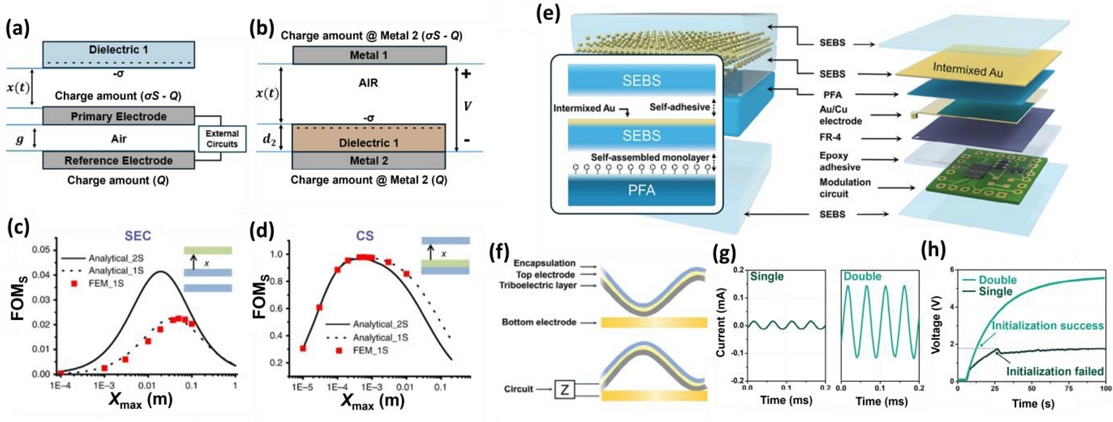
Structural FOMs relative to the TENG mode. Structure models of the (a) SEC and (b) CS modes. Adapted from Refs. [18] and [19]. FOMs for (c) SEC and (d) CS from analytical formulas and FEM simulation. Adapted from Ref. [20]. (e) Structural schematic and components of the DUS-TENG. Adapted from Ref. [21]. (f) Ultrasound detection mechanism of the DUS-TENG. Performance comparison of a single- vs. double-electrode US-TENG for (g) current and (h) capacitor charging.
Figs. 2 (c) and (d) show the calculation and analysis of the FOMs for the two TENG modes described above [27]. The analysis was conducted via FEM simulation, assuming the same charge density and identical area for the dielectrics. The FOMs used for the calculations were as follows:
where ε0 is the permittivity of vacuum, σ is the surface charge density, Em is the theoretically maximized output energy, A is the active area of the device, and xmax refers to the distance between the two layers. In addition, one-sided (1S) and two-sided (2S) effects due to the nonideal parallel-plate capacitance were considered, and it was confirmed that the calculations accounting for the 1S effect matched the simulation results. As shown in the graph, the FOM of the CS mode TENG was over 20 times higher than that of the SEC TENG, indicating that the conversion efficiency of the CS mode TENG was theoretically superior.
Based on this theory, Fig. 2 (e) describes the double-electrode US-TENG (DUS-TENG) for implementing an ultrasound-driven CS mode TENG [28]. The DUS-TENG enables efficient transmission of acoustic energy while realizing the CS mode. A close examination of the device structure reveals that a thin (≈ 25 μm) PFA layer is combined with a styrene-ethylene-butylene-styrene intermixed layer containing Au nanoparticles with a thickness under 45 nm, serving as both encapsulation and an electrode for the vibration layer. The immovable layer is fabricated in a manner identical to that of the VI-TEG. Through this structure, the DUS-TENG can achieve a CS mode TENG while minimizing changes in the PFA properties, thereby enhancing the efficiency of ultrasound signal conversion.
Fig. 2 (f) shows the operating principle of the DUS-TENG under ultrasonic irradiation. The DUS-TENG is constructed in a capacitor configuration, in which a dielectric layer is located between the Au nanofilm and Au/Cu electrodes. When the membrane vibrates upon exposure to ultrasound, contact electrification occurs between the PFA and bottom electrode, causing negative charges to move to the inner surface of the PFA. As the membrane separates, the potential of the bottom electrode increases relative to that of the top electrode, resulting in the release of holes and current flowing from the bottom to the top electrode. When the membrane returns to contact, the potential decreases, producing a current in the opposite direction. The repeated contact–separation cycles induced by the ultrasound produce a current waveform at a frequency of 20 kHz.
Through this mechanism, the DUS-TENG converts ultrasonic energy into electrical signals, and the output is compared with that of the single-electrode US-TENG in Fig. 2 (g). The DUS-TENG exhibits an output that is over 9.6 times greater than the single-electrode US-TENG, and, as illustrated in Fig. 2 (h), succeeds in charging a capacitor for a microcontroller, unlike the single-electrode US-TENG, which fails to accomplish this.
Theoretical analysis and experimental results demonstrate that, for US-TENGs, the CS configuration yields higher energy conversion efficiency than the SEC configuration. These findings suggest that the device operating mode significantly influences the energy conversion efficiency and sensor performance.
Although modifications to the device structure can improve the efficiency of detecting and converting ultrasonic energy, the wave-based nature of ultrasound can also lead to energy losses depending on the medium encountered.
Fig. 3 shows strategies for improving the density and intensity of the ultrasound incident on the device by matching the acoustic impedance, thereby minimizing the loss of incident energy. Ti and its alloys have long been regarded as standard packaging materials for commercial IMDs [29]. Although Ti offers a significant advantage of high stability, it has notable limitations in terms of energy transfer. From the perspective of ultrasound propagation, greater differences in density (i.e., acoustic impedance) between materials result in increased reflection of ultrasound waves [30]. The acoustic impedance is defined as follows:
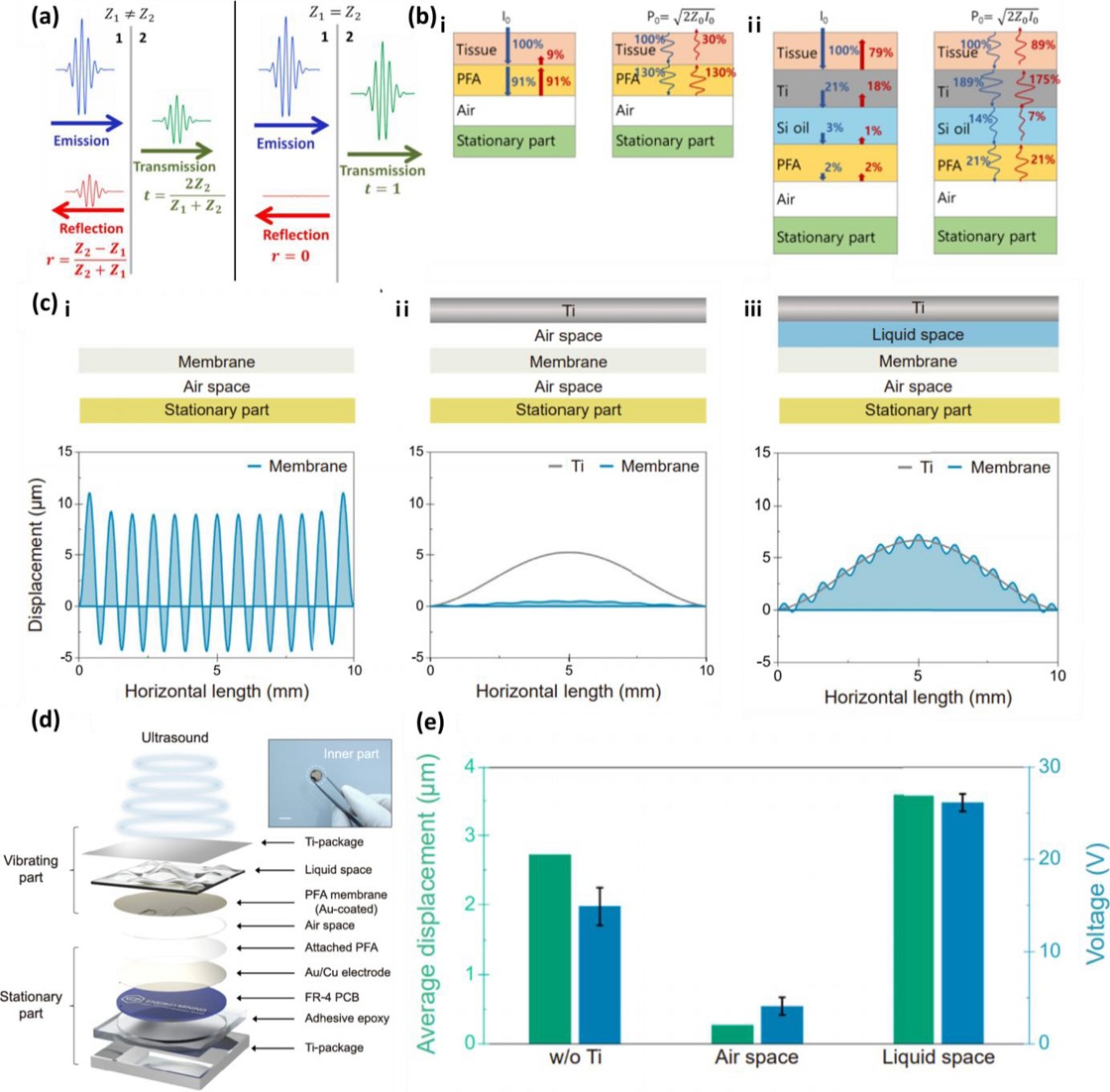
Mechanical design principles in ultrasound-driven energy delivery with Ti packaging. (a) Ultrasound interfacial propagation between media. Adapted from Ref. [25]. (b) Calculation of ultrasound intensity and pressure at each layer of the TENG. Insets i and ii present the intensity and pressure distributions for the TENG without Ti packaging and under ultrasound excitation, respectively. Adapted from Ref. [26]. (c) FEM analysis of vibration displacement across layered structures. Insets i–iii present the results for configurations without Ti, with Ti and an air gap, and with Ti and a liquid gap, respectively. (d) TI-TENG schematic/real device images. (e) Displacement–voltage correlations across structural designs.
where ρ denotes the density of the material, and κ, which is defined as κ = ω/υ + iα/2, represents the wavenumber. In this context, υ is the longitudinal phase velocity, ω is the angular frequency, and α is the attenuation coefficient. The acoustic impedance of Ti is approximately 20 times greater than that of skin tissue, leading to most of the ultrasonic energy being reflected [31].
Fig. 3 (a) provides a mathematical description of the ultrasound transmission and reflection at the interface between two media with differing densities [32]. The transmission coefficient (t) from medium 1 to medium 2 is expressed as
Similarly, the reflection coefficient (r) is determined as follows:
where Z1 and Z2 represent the acoustic impedances of medium 1 and medium 2, respectively. As Z1 and Z2 become more similar, the transmission coefficient increases, allowing for smoother propagation of the ultrasound.
Reflecting on these properties, Fig. 3 (b) illustrates a layer-by-layer structural design that maximizes acoustic impedance matching within the skin tissue for conventional US-TENGs and after Ti packaging [33]. The density and pressure of the incident ultrasound are also calculated using the relevant equations. In conventional US-TENGs, as shown in Fig. 3 (b)-i, the density difference between the PFA and tissue is minimal; however, the substantial disparity between the PFA and air gap results in significant ultrasound reflection. In addition, Ti packaging is essential for implantable devices to ensure biocompatibility and mechanical protection. As shown in Fig. 3 (b)–ii, when paired with an acoustic impedance-matching design, the addition of a liquid layer maximizes transmission, enabling the Ti-packaged US-TENG to perform similarly to or better than conventional configurations.
Based on the calculated results, Fig. 3 (c) presents the FEM simulations analyzing the vibration of the PFA membrane in both conventional US-TENG and Ti-packaged configurations. In the conventional US-TENGs shown in Fig. 3 (c)-i, the PFA membrane vibrated in multiple modes under ultrasound irradiation [17]. However, as shown in Fig. 3 (c)-ii, the US-TENG with Ti packaging exhibited single-mode vibration of the Ti plate because of its higher mass and stiffness relative to that of a polymeric membrane. The PFA membrane exhibited no vibration, as most of the ultrasonic energy was reflected at the Ti–air interface owing to the large difference in density between these layers. To address this issue, an intermediate medium that can buffer the density difference, specifically a liquid layer, can be inserted into the air gap, thereby enhancing the ultrasound transmission. Silicone oil, selected because of its high flexibility and excellent wettability (which minimizes the air gap), was used as the liquid layer. As shown in Fig. 3 (c)-iii, the PFA membrane vibrated following the single-mode resonance of Ti, and ultrasound energy was efficiently transmitted, resulting in a uniform contact area and vibration direction that enabled high conversion efficiency. These simulation results demonstrate that inserting an acoustic impedance-matching layer between media with large impedance mismatches can minimize the ultrasound reflection at the interface, thereby maximizing the density and pressure of the incident ultrasound.
Based on the preceding calculations and simulations, this concept was experimentally confirmed by fabricating a device. Fig. 3 (d) presents an enlarged schematic of the Ti-vibrating TENG (TI-TENG) design, which was developed based on the above calculations and simulations. The device is composed of a 0.5-mm-thick external Ti packaging, which is standard for commercial IMDs, with a 50-μm-thick silicone oil layer as the liquid medium, and an Au-coated PFA membrane serving as the electrode and vibrating part. The stationary part comprises the PFA membrane bonded to an Au/Cu PCB substrate, acting as the tribo-negative material, which is connected to the vibrating parts via a 5 μm air gap, thereby operating in the CS mode US-TENG structure. The entire assembly is encapsulated in adhesive epoxy, and the completed device is less than 10 mm in size. This configuration integrates the theoretically verified acoustic impedance-matching layer and operates in a CS mode TENG, forming a structural design that minimizes the loss of incident ultrasound density and pressure even after Ti packaging.
To verify the operational performance experimentally, the conversion efficiency of each configuration, including the basic US-TENG before packaging, US-TENG with Ti packaging and an air gap, and TI-TENG, was assessed with a 20 kHz ultrasonic energy density of 430 mW/cm². Fig. 3 (e) shows the actual voltage signals and average displacement of the vibrating part under these conditions. The graph confirms that the physical displacement of the vibrating part was proportional to the amplitude of the signal pulses and that greater displacement led to higher conversion efficiency. This phenomenon was maximized in specific layered structures.
2.3 Ultrasound transmission and packaging beyond Ti
Two main strategies for improving the conversion efficiency of US-TENGs have been discussed. However, approaches for enhancing FOMs are limited by the specific TENG operation mode, and methods for increasing ultrasound transmittance within Ti packaging also have inherent limitations. Accordingly, active research on next-generation device packaging technologies that are capable of converting incident ultrasound energy without loss is ongoing. These developments are illustrated in Fig. 4.
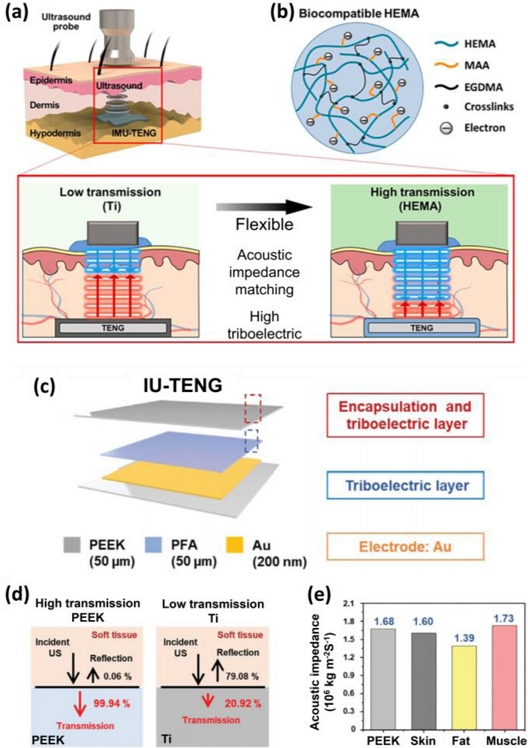
Ultrasound transmission and packaging beyond Ti. (a) Concept schematic of ultrasound-detecting IMU-TENG. Adapted from Ref. [27]. (b) Details of the layer used for both encapsulation and as a triboelectric material in the IMU-TENG. (c) Structure and function of each IU-TENG layer. Adapted from Ref. [28]. (d) Calculated ultrasound intensity transmission through PEEK versus Ti. (e) Acoustic impedance values of PEEK and soft tissue.
Fig. 4 (a) depicts the implantable, modulus-tunable, ultrasound-driven TENG (IMU-TENG) that incorporates poly(2-hydroxyethyl methacrylate) (HEMA), which enables the simultaneous encapsulation and formation of the triboelectric layer [34]. Owing to its high biocompatibility, as demonstrated by its use in contact lenses, HEMA offers exceptional flexibility and possesses an acoustic impedance similar to that of human skin tissue. These characteristics allow the efficient transmission of ultrasonic energy, block the penetration of moisture into the device, and, unlike Ti packaging, enable its application in flexible devices. As shown in Fig. 4 (b), the properties of HEMA can be further tuned by adding ionic methacrylic acid (MAA) to enhance the triboelectric performance, whereas ethylene glycol dimethacrylate (EGDMA) serves as a crosslinker to adjust the Young’s modulus of the material. This tunability of the physicochemical properties makes HEMA a highly promising packaging material for next-generation US-TENGs.
Fig. 4 (c) shifts the focus to acoustic impedance by introducing polyether ether ketone (PEEK) as both the packaging and triboelectric layers for implantable ultrasound-driven TENGs (IU-TENGs) [35]. PEEK is a biocompatible material containing multiple electron-donating groups and exhibits an acoustic impedance that closely matches that of skin tissue. As demonstrated in Figs. 4 (d) and (e), Ti exhibits a high acoustic impedance of 2.73 × 107 kg/m²s compared with 1.6 ×106 kg/m²s for skin tissue, resulting in an ultrasonic energy reflection rate exceeding 79.08%. In contrast, PEEK, with an acoustic impedance of 1.68 × 106 kg/m²s, demonstrates a reflection rate of less than 0.06%, ensuring that most ultrasonic energy is absorbed and converted, thereby minimizing energy loss.
Ongoing research seeks not only to overcome the obstacles posed by Ti packaging from the perspective of acoustic impedance and encapsulation, but also to explore diverse packaging materials that can function as triboelectric layers. These efforts continue to advance the field of high-efficiency implantable energy-harvesting devices.
3. CONCLUSIONS
Triboelectric-effect-based ultrasonic-to-electrical signal conversion has attracted increasing interest as a next-generation platform for ultrasound sensing and wireless power transfer. It detects ultrasound energy, which can be safely transmitted to the human body, and converts it into electrical signals, offering broad application potential. This technology, which is characterized by freedom of material selection, miniaturized device design, and high energy-conversion efficiency, can be applied across a wide range of fields, including power systems for low-power IMDs and neural stimulation sensors.
In this review, we comprehensively discussed the emergence and operating principles of US-TENGs, which are distinguished from conventional TENGs. We analyzed the conversion efficiencies according to the operating modes, explored the influence of acoustic impedance differences between ultrasound incident layers on the membrane behavior and output, and reviewed the latest trends in packaging materials. In addition, we examined the variations in the FOMs according to the operating modes and electrode structures, strategies to minimize ultrasonic energy loss through acoustic impedance optimization, and next-generation TENG packaging strategies to overcome the limitations of traditional Ti packaging.
However, ultrasound-driven electrostatic devices still face challenges. Key issues include output degradation owing to variations in the ultrasound incidence angles, energy loss during propagation within biological tissues, and device deterioration and output reduction caused by repetitive mechanical vibrations. In conclusion, although the current energy conversion efficiency remains somewhat limited, overcoming these technical constraints is anticipated to enhance the commercialization prospects and market competitiveness of US-TENG technologies significantly.
Acknowledgments
This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (No. RS-2025-00521476).
References
-
F.-R. Fan, Z.-Q. Tian, Z.L. Wang, Flexible triboelectric generator, Nano Energy 1 (2012) 328–334.
[https://doi.org/10.1016/j.nanoen.2012.01.004]

-
S.S. Kwak, H.-J. Yoon, S.-W. Kim, Textile-Based Triboelectric Nanogenerators for Self-Powered Wearable Electronics, Adv. Funct. Mater. 29 (2019) 1804533.
[https://doi.org/10.1002/adfm.201804533]

-
N. Rubab, S.-W. Kim, Triboelectric Nanogenerators for Self-powered Sensors, J. Sens. Sci. Technol. 31 (2022) 79–84.
[https://doi.org/10.46670/JSST.2022.31.2.79]

-
S. Biswas, Y.J. Lee, H.J. Choi, H. Kim, Current Development in Bio-implantable Sensors, J. Sens. Sci. Technol. 31 (2022) 403–410.
[https://doi.org/10.46670/JSST.2022.31.6.403]

-
J.H. Kim, H.J. Ryu, S.M. Kim, H.Y. Lee, A. Karami, D. Galayko, et al., Self-Boosting Energy Generation via Triboelectric Nanogenerator–Capacitor Coupling, Adv. Mater. Technol. 9 (2024) 2301309.
[https://doi.org/10.1002/admt.202301309]

-
J.Y. Kim, J.M. Yoo, H.T. Seo, R. Avila, G.Y. Chung, G.R. Shin, et al., Magnetically driven triboelectric nanogenerator for a wireless, versatile energy transfer system, Sci. Adv. 11 (2025) eadu5919.
[https://doi.org/10.1126/sciadv.adu5919]

-
J. Chen, Z.L. Wang, Reviving Vibration Energy Harvesting and Self-Powered Sensing by a Triboelectric Nanogenerator, Joule 1 (2017) 480–521.
[https://doi.org/10.1016/j.joule.2017.09.004]

-
D.M. Tiruneh, G.R. Jang, K.H. Kwon, H.J. Ryu, Highly compact inertia-driven triboelectric nanogenerator for self-powered wireless CO2 monitoring via fine-vibration harvesting, Nano Energy 138 (2025) 110872.
[https://doi.org/10.1016/j.nanoen.2025.110872]

-
J.G. Jeong, E.H. Jo, J.-A. Choi, Y.S. Kang, S.J. Pyo, Structural design strategies of triboelectric nanogenerators for omnidirectional wind energy harvesting, Micro Nano Syst. Lett. 13 (2025) 4.
[https://doi.org/10.1186/s40486-025-00224-6]

-
S.J. Lee, H.-J. Ko, J.B. Kim, Self-powered gas sensor based on triboelectric nanogenerators (TENG): a comparative review of sensing mechanisms, Micro Nano Syst. Lett. 13 (2025) 2.
[https://doi.org/10.1186/s40486-025-00222-8]

-
H.J. Ryoo, C.W. Lee, J.W. Han, W. Kim, D.H. Choi, A Triboelectric Nanogenerator Design for the Utilization of Multi-Axial Mechanical Energies in Human Motions, J. Sens. Sci. Technol. 29 (2020) 312–322.
[https://doi.org/10.46670/JSST.2020.29.5.312]

-
S.H. Ji, Y.C. Chang, J.H. Park, Module-type Triboelectric Nanogenerator for Collecting Various Kinetic Energies, J. Sens. Sci. Technol. 31 (2022) 376–382.
[https://doi.org/10.46670/JSST.2022.31.6.376]

-
H.S. Park, G.S. Gbadam, S. Niu, H.J. Ryu, J.-H. Lee, Manufacturing strategies for highly sensitive and self-powered piezoelectric and triboelectric tactile sensors, Int. J. Extrem. Manuf. 7 (2025) 012006.
[https://doi.org/10.1088/2631-7990/ad88be]

-
C.J. Lee, J.Y. Yang, S.-K. Kim, Y.G. Jin, H.S. Park, S.J. Gwak, et al., Self-powered directional dust removal via wind-driven phase-controlled TENG for solar panel maintenance, Nano Energy 142 (2025) 111262.
[https://doi.org/10.1016/j.nanoen.2025.111262]

-
X. Fan, J. Chen, J. Yang, P. Bai, Z. Li, Z.L. Wang, Ultrathin, Rollable, Paper-Based Triboelectric Nanogenerator for Acoustic Energy Harvesting and Self-Powered Sound Recording, ACS Nano 9 (2015) 4236–4243.
[https://doi.org/10.1021/acsnano.5b00618]

-
F.-C. Kao, S.-F. Hung, C.-C. Yang, P. Parashar, C.-J. Huang, M.-K Hsieh, et al., Ultrasound-driven triboelectric and piezoelectric nanogenerators in biomedical application, J. Phys. Energy 6 (2024) 022002.
[https://doi.org/10.1088/2515-7655/ad307c]

-
R. Hinchet, H.-J. Yoon, H.J. Ryu, M.-K. Kim, E.-K. Choi, et al., Transcutaneous ultrasound energy harvesting using capacitive triboelectric technology, Science 365 (2019) 491–494.
[https://doi.org/10.1126/science.aan3997]

-
D.-M. Lee, N. Rubab, I.A. Hyun, W.S. Kang, Y.-J. Kim, M.K. Kang, et al., Ultrasound-mediated triboelectric nanogenerator for powering on-demand transient electronics, Sci. Adv. 8 (2022) eabl8423.
[https://doi.org/10.1126/sciadv.abl8423]

-
I.M. Imani, B.S. Kim, X. Xiao, N. Rubab, B.-J. Park, Y.-J. Kim, et al., Ultrasound-Driven On-Demand Transient Triboelectric Nanogenerator for Subcutaneous Antibacterial Activity, Adv. Sci. 10 (2023) 2204801.
[https://doi.org/10.1002/advs.202204801]

-
Y. Zhang, F. Zheng, Z. Zhou, Y. Zhang, F. Wang, H. Tao, A Transient Triboelectric Nanogenerator with Optical Feedback, Proceedings of the IEEE International Conference on Micro Electro Mechanical Systems (MEMS), Northern Ireland, UK, 2018, pp. 632–634.
[https://doi.org/10.1109/MEMSYS.2018.8346633]

-
D.Y. Park, H.-M. Lee, S.-H. Kim, Y.M. Sim, Y.J. Kang, G.-R. Jang, et al., Recent Advances in Bioresorbable Biomedical Applications: From Materials to Devices, Adv. Electron. Mater. 11 (2025) 2400997.
[https://doi.org/10.1002/aelm.202400997]

-
H.-M. Lee, H.J. Ryu, Bioresorbable Materials for Wound Management, Biomimetics 10 (2025) 108.
[https://doi.org/10.3390/biomimetics10020108]

-
L. Zhang, Y. Liu, X. Sun, Z. Wen, Advances in triboelectric nanogenerators in acoustics: Energy harvesting and Sound sensing, Nano Trends 8 (2024) 100064.
[https://doi.org/10.1016/j.nwnano.2024.100064]

-
H.-J. Yoon, S.-W. Kim, Nanogenerators to Power Implantable Medical Systems, Joule 4 (2020) 1398–1407.
[https://doi.org/10.1016/j.joule.2020.05.003]

-
S. Niu, Y. Lui, S. Wang, L. Lin, Y. S. Zhou, Y. Hu, et al., Theoretical Investigation and Structural Optimi- zation of Single-Electrode Triboelectric Nanogenerators, Adv. Funct. Mater. 24 (2014) 3332–3340.
[https://doi.org/10.1002/adfm.201303799]

-
S. Nui, S. Wang, L. Lin, Y. Liu, Y. S. Zhou, Y. Hu, et al., Theoretical study of contact-mode triboelectric nanogenerators as an effective power source, Energy Environ. Sci. 6 (2013) 3576–3583.
[https://doi.org/10.1039/c3ee42571a]

-
Y. Zi, S. Niu, J. Wang, Z. Wen, W. Tang, Z.L. Wang, Standards and figure-of-merits for quantifying the performance of triboelectric nanogenerators, Nat. Commun. 6 (2015) 8376.
[https://doi.org/10.1038/ncomms9376]

-
Y.W. Chung, H. Yuan, Z. Wang, J.-M. Jeong, B.-J. Park, J.-H. Hwang, et al., Acoustic Tunable Battery-Free Implants Based on Sustainable Triboelectric Nanogenerators With Metal-Polymer Intermixing Layers, Adv. Energy Mater. 15 (2025) 2403712.
[https://doi.org/10.1002/aenm.202403712]

-
Y.-H. Joung, Development of Implantable Medical Devices: From an Engineering Perspective, Int. Neurourol. J. 17 (2013) 98–106.
[https://doi.org/10.5213/inj.2013.17.3.98]

-
S.Y. Jung, J.S. Park, M.-S. Kim, H.W. Jang, B.C. Lee, S.-H. Baek, Piezoelectric Ultrasound MEMS Transducers for Fingerprint Recognition, J. Sens. Sci. Technol. 31 (2022) 286–292.
[https://doi.org/10.46670/JSST.2022.31.5.286]

-
M.O. Culjat, M.J. Choi, R.S. Singh, W.S. Grundfest, E.R. Brown, S.N. White, Ultrasound detection of submerged dental implants through soft tissue in a porcine model, J. Prosthet. Dent. 99 (2008) 218–224.
[https://doi.org/10.1016/S0022-3913(08)60046-3]

-
R.-M. Guillermic, M. Lanoy, A. Strybulevych, J.H. Page, A PDMS-based broadband acoustic impedance matched material for underwater applications, Ultrasonics 94 (2019) 152–157.
[https://doi.org/10.1016/j.ultras.2018.10.002]

-
Y.W. Chung, J.-M. Jeong, J.-H. Hwang, Y.-J. Kim, B.-J. Park, D.S. Cho, et al., Gigantic triboelectric power generation overcoming acoustic energy barrier using metal-liquid coupling, Joule 8 (2024) 2681–2695.
[https://doi.org/10.1016/j.joule.2024.06.016]

-
B.S. Kim, H.-J. Yoon, Y.-J. Kim, B.-J. Park, J.-H. Jung, S.-W. Kim, Ultrasound-Driven Triboelectric Nanogenerator with Biocompatible 2-Hydroxyethyl Methacrylate, ACS Energy Lett. 8 (2023) 3412−3419.
[https://doi.org/10.1021/acsenergylett.3c00953]

-
S. Jeon, X. Meng, N. Rubab, D.B. Kim, H. Mo, X. Xiao, et al., Ultrasound-Driven Highly Stable Implantable Triboelectric Nanogenerator with Human-Tissue Acoustic Impedance-Matched Polyether Ether Ketone, Adv. Mater. Technol. 9 (2024) 2400317.
[https://doi.org/10.1002/admt.202400317]


Ki-Yoon Park is a BS/MS Crouse researcher at Gachon University. His research interests include piezoelectric, triboelectric, and ferroelectric materials, mechanical-electrical energy harvesting device designs, and implantable bioresorbable medical devices and sensors. For more information, please visit the website https://sites.google.com/view/yoonresearchlab/home.

Prof. Hong-Joon Yoon is an Assistant Professor in the Department of Electronic Engineering at Gachon University, Korea. He received his Ph.D. from the School of Advanced Materials Science and Engineering, Sungkyunkwan University in 2018. Prof. Yoon’s current research interests include the design and development of piezoelectric and triboelectric nanogenerators for wearable/implantable applications, and bioresorbable materials-based 3D sensors/devices. Further details can be found at https://sites.google.com/view/yoonresearchlab/home.
