
Wireless Bioelectronics for Bladder Function Monitoring
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Continuous bladder function monitoring is essential for the diagnosis and management of urological disorders; however, conventional methods such as catheter-based urodynamic studies are invasive, uncomfortable, and unsuitable for long-term use. Recent advances in flexible electronics, soft materials, and wireless communication have enabled the development of wearable and implantable bioelectronics that support real-time, continuous, patient-friendly bladder monitoring. This review summarizes progress across key modalities, namely, ultrasound- and bioimpedance-based wearable platforms, strain gauge-based implantable platforms, and multimodal implantable systems, which combine sensing with electrical stimulation. Their respective advantages, limitations, and potential clinical applications are discussed. In the future, the integration of multi-parameter systems, energy harvesting, and artificial intelligence-driven analytics could transform these devices into intelligent, self-regulating platforms that not only monitor but also actively manage bladder health. Such innovations are promising for advancements in personalized urological care from episodic clinical assessments toward continuous, adaptive interventions.
Keywords:
Bladder function monitoring, Wireless bioelectronics, Wearable device, Implantable device1. INTRODUCTION
Lower urinary-tract disorders, including an overactive bladder, urinary incontinence, a neurogenic bladder, and post-surgical recovery, are prevalent urological conditions that significantly impair the quality of life and impose a substantial economic burden on patients, caregivers, and health systems [1-5]. These disorders affect both men and women across different age groups and are closely associated with increased comorbidities and higher healthcare utilization. Neurogenic bladder dysfunction frequently arises in neurological conditions such as multiple sclerosis and spinal cord injury, necessitating structured, long-term monitoring to prevent urinary tract complications, preserve renal function, and optimize patient outcomes [6,7].
Given their high prevalence, associated comorbidities, and risk of long-term complications, accurate and continuous monitoring of bladder function is essential for the effective diagnosis and management of these disorders [8-11]. Conventional diagnostic approaches, such as catheter-based urodynamic studies, provide valuable physiological insights but are limited by their invasive nature, patient discomfort, infection risk, intermittent measurement, and high medical costs (Fig. 1 (a)) [12,13].
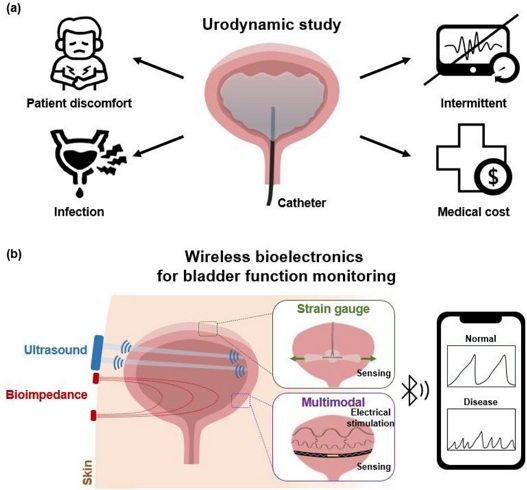
Overview of wireless bioelectronics for bladder function monitoring. (a) Limitations of catheter-based urodynamic studies, including patient discomfort, infection risk, intermittent measurement, and high medical costs. (b) Wireless bioelectronics for bladder function monitoring, comprising wearable platforms based on ultrasound and bioimpedance technologies and implantable platforms employing strain gauge-based technologies and multimodal systems that integrate sensing with electrical stimulation.
Early efforts to address these limitations focused on bladder-monitoring systems capable of recording data over extended periods without requiring the continuous presence of healthcare personnel [14-22]. Many of these prototypes relied on wired connections to external hardware for power and data acquisition [23,24]. Although this configuration enabled continuous measurements and introduced modalities such as ultrasound patches and bioelectrical impedance analysis, dependence on cables resulted in bulky form factors, restricted patient mobility, and increased susceptibility to motion artifacts.
Advances in flexible electronics, low-power circuitry, soft materials, and wireless communication protocols have driven the transition to fully wireless bioelectronic platforms [25-32]. By integrating sensing, power management, and data transmission into compact, skin-conformal, or implantable form factors, these wireless systems overcome the primary limitations of their wired predecessors, eliminating cable constraints, improving comfort, and enabling long-term, real-time continuous monitoring [25,27,33-37]. This wireless paradigm is also consistent with broader trends in digital healthcare, where remote patient monitoring, cloud-based data analytics, and personalized intervention strategies are becoming central to chronic disease management [35,38]. As research advances, wireless bladder function monitoring systems are evolving toward wearable platforms based on ultrasound- and bioimpedance-based technologies, as well as implantable platforms employing strain gauge-based technologies and multimodal systems that combine sensing with electrical stimulation, thereby setting the foundation for more proactive and individualized urological care (Fig. 1 (b)) [17,25-29].
In this review, we first examine the recent developments in wireless wearable bioelectronics for bladder function monitoring, focusing on key sensing technologies and representative designs. We then discuss wireless implantable bioelectronics, highlighting strain-gauge-based and multimodal systems that integrate sensing with electrical stimulation. Finally, we discuss practical considerations and remaining challenges, including long-term biocompatibility, signal reliability, power management, and artificial intelligence-driven analytics, and outline future research directions aimed at advancing these technologies toward reliable, patient-centered clinical deployment.
2. WEARABLE PLATFORMS
Wireless wearable bioelectronics for bladder function monitoring have translated broader technological advances into practical, patient-friendly tools [15,25]. These noninvasive systems overcome the limitations of intermittent, invasive catheter-based urodynamic studies. By integrating skin-interfaced platforms with miniaturized, low-power electronics, they enable continuous and quantitative monitoring of bladder function without compromising patient mobility or comfort.
Depending on the sensing technology, such as ultrasound-based wall distance measurement or bioimpedance analysis, wearable devices can capture volumetric trends, detect bladder filling patterns, and generate actionable data for clinical decision-making [38,39]. This capability enables real-time, continuous monitoring in both home and hospital settings, supports management strategies for bladder-related diseases, and creates opportunities for preventive care and early detection [3]. Ultimately, wearable bioelectronics for bladder function monitoring represent a pivotal step toward personalized digital healthcare systems in next-generation urological care.
2.1 Ultrasound-based technologies
Wearable ultrasound-based devices use acoustic time-of-flight measurements to continuously monitor bladder filling and emptying [25,26]. These devices estimate bladder volume noninvasively by transmitting and receiving ultrasound waves through the lower abdomen. When combined with flexible transducer arrays and soft, skin-conformal packaging, ultrasound technology offers a comfortable, gel-free option suitable for extended daily use.
Figs. 2 (a)–(c) illustrate a wireless ultrasound patch that integrates miniature piezoceramic transducers, control electronics, and Bluetooth low-energy (BLE) communication into a thin, soft platform [25]. The device adheres to the suprapubic area, emits ultrasonic pulses, and detects echoes from the bladder walls to calculate bladder volume. Its multi-layer construction consists of a bio-adhesive elastomer, a matching layer, and an encapsulated, flexible printed circuit board (PCB) (Fig. 2 (b)). The compact, lightweight form factor (Fig. 2 (c)) enables patients to move freely while continuously transmitting bladder volume data to a mobile device. An air-backed transducer design enhances sensitivity (≈4-fold higher SNR than a backed commercial transducer) and enables clear wall echoes at low drive voltages (~30 V).
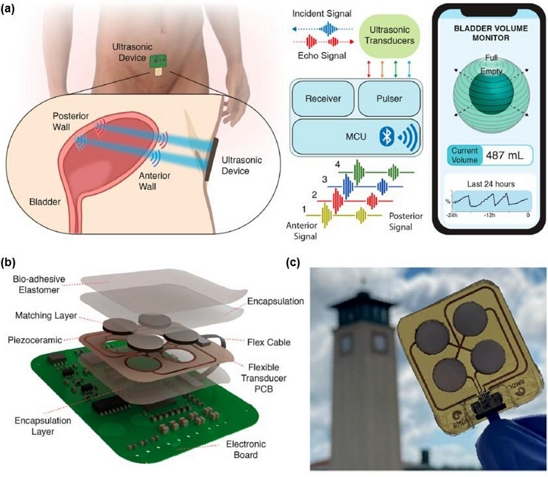
Wearable ultrasound-based device for bladder volume monitoring. (a) Schematic of the device positioned on the lower abdomen to emit and receive ultrasonic pulses. (b) Exploded view of the multilayer structure, comprising bio-adhesive elastomer, acoustic matching layer, piezoceramic transducers, flexible PCB, and encapsulation. (c) Photograph of the flexible ultrasonic transducer array. Adapted from Ref. [25].
This study demonstrates how the monolithic integration of sensing, control, power, and wireless communication within a body-conformal form factor can enable accurate, continuous, and real-time bladder volume tracking. It provides a foundation for future wireless bladder monitoring systems designed for clinical translation and at-home management of urological disorders [10,25].
2.2 Bioimpedance-based technologies
Wearable bioimpedance-based devices estimate bladder volume by measuring the electrical resistance and reactance of tissues in the suprapubic region. Because urine exhibits higher conductivity than the surrounding tissues, bladder filling induces predictable changes in bioimpedance [26,39]. This approach supports simple, low-power hardware designs that operate with dry electrodes. Consequently, bioimpedance-based devices provide high wearability and are well-suited for the long-term monitoring of bladder function.
Fig. 3 shows a representative example of a bioimpedance-based device for bladder function monitoring [26]. Fig. 3 (a) illustrates the physiological mechanism of urination and the neural pathways involved in bladder control, highlighting bladder-related disorders that necessitate continuous monitoring. The device integrates flexible chitosan–glycerol composite electrodes (Fig. 3 (b)) with a compact electronic module containing a microcontroller, signal processing circuitry, a rechargeable battery, and BLE communication (Fig. 3 (c)–(d)). Designed as a patch that adheres to the suprapubic area (Fig. 3 (e)), it delivers a low-amplitude alternating current through the electrodes, continuously measuring impedance changes that correlate with bladder filling. Data are transmitted wirelessly to a mobile device for real-time visualization and storage, enabling remote monitoring without restricting mobility. In human testing, measurements from the integrated device demonstrated strong agreement with a commercial instrument (r ≈ 0.9699), supporting its feasibility for day-to-day, radiation-free assessment of bladder status.
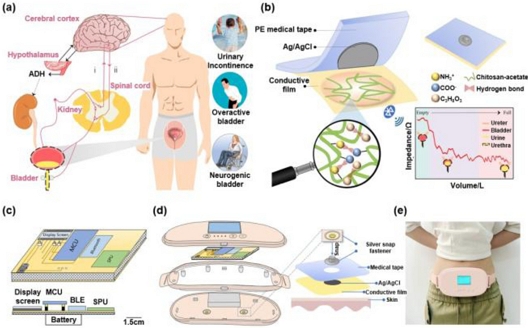
Wearable bioimpedance-based device for bladder volume monitoring. (a) Schematic illustration of the urination mechanism and neural pathways. (b) Flexible chitosan–acetic acid composite electrodes for impedance signal acquisition. (c) Cutaway view (top) and longitudinal section (bottom) of the system hardware circuit board. (d) Packaged electronics with connections to the patch electrodes. (e) Photograph of the device positioned on the suprapubic area. Adapted from Ref. [26].
This study demonstrates that integrating biocompatible electrodes, wireless communication, and low-power electronics into a skin-conformal form factor provides a practical, noninvasive solution for continuous bladder monitoring. It underscores the potential of bioimpedance-based devices as lightweight, patient-friendly, and cost-effective approaches to urological care [38,39].
3. IMPLANTABLE PLATFORMS
Wireless implantable bioelectronics for bladder function monitoring are designed to directly and accurately measure bladder activity and internal pressure within the body [17,27,28]. By establishing stable contact with the bladder wall, these devices enable precise detection of bladder wall deformation, tension changes, and related physiological signals with high temporal resolution. Unlike intermittent catheter-based urodynamic studies, implantable devices allow continuous monitoring over extended periods, ranging from hours to weeks. This capability is particularly valuable for tracking postsurgical recovery, monitoring patients with neurogenic bladders, and assessing underactive bladder cases in which gradual functional changes must be quantified over time.
Compared with wearable devices, implantable devices overcome key limitations such as signal attenuation, motion artifacts, and dependence on patient positioning, thereby providing more reliable and accurate long-term monitoring [27,28,40]. These platforms incorporate biocompatible encapsulation layers, stretchable sensing elements, and wireless telemetry modules, enabling operation without external leads. Power and data are typically managed through wireless transmission, eliminating the need for tethered connections and reducing associated risks [27,28,41]. Collectively, these features support real-time and long-term monitoring in home-based settings, aligning with the broader shift toward patient-centered, remote urological care.
3.1 Strain gauge-based technologies
Implantable strain gauge-based devices measure the mechanical deformation of the bladder wall during filling and voiding, providing direct and reliable data on bladder volume and internal pressure changes [27,40]. With rapid response, high sensitivity, and reliable operation over extended periods, this approach offers a robust foundation for continuous long-term monitoring and may facilitate earlier detection of functional impairments.
Fig. 4 (a) illustrates a fully implantable wireless platform that integrates a stretchable strain gauge attached to the bladder wall with a wireless data transmission system [27]. The strain gauge records resistance changes as the bladder expands and contracts, and it transmits data wirelessly to a mobile device to monitor recovery following partial cystectomy. Fig. 4 (b) and 4 (c) present the internal pressure profiles of a healthy bladder and a bladder after partial cystectomy, respectively, enabling real-time, continuous tracking of recovery and regeneration. The average difference and standard deviation of bladder pressure inferred from the strain gauge compared with direct measurement are 8.1 × 10−9 cmH2O and −1.2 cmH2O, respectively.
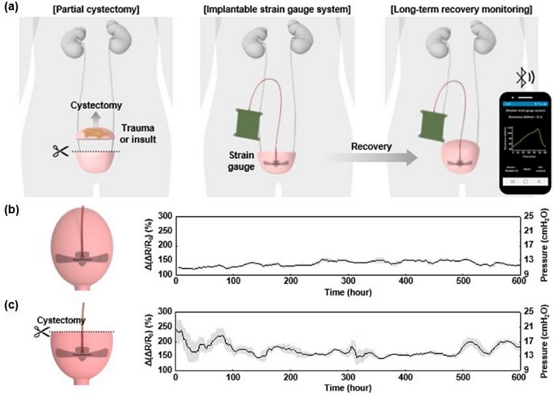
Implantable strain gauge-based device for bladder function monitoring. (a) Schematic of long-term monitoring to track recovery and regeneration after partial cystectomy. Internal pressure profiles in a non-human primate model (baboon) with (b) a healthy bladder and (c) a bladder after partial cystectomy. Adapted from Ref. [27].
By eliminating percutaneous leads and fully encapsulating all components with biocompatible materials, this technology reduces infection risk, enhances patient comfort, and supports long-term untethered monitoring [27,41]. These features underscore the potential of strain gauge-based devices for clinical postsurgical recovery assessment and chronic bladder function management, bridging the gap between preclinical research and future clinical translation.
3.2 Multimodal systems
Implantable multimodal systems integrate multiple sensing modalities and therapeutic stimulation within a single platform. By combining mechanical strain sensing, electromyography (EMG), and electrical stimulation, these systems can detect bladder filling, trigger voiding, and confirm successful emptying in one device [17,28].
Fig. 5 illustrates an implantable multimodal system for bladder sensing and electrical stimulation [28]. The soft, mesh-like expandable electronic web (Fig. 5 (a)) wraps around the bladder and incorporates strain gauges and EMG electrodes to monitor bladder wall deformation and detrusor muscle activity. Platinum-black-coated stimulation electrodes are integrated to deliver targeted electrical stimulation when voiding is required. The wireless module (Fig. 5 (b)) manages real-time sensing, BLE communication, and remote control of stimulation protocols, powered by radio-frequency coupling without the need for an onboard battery [28,41]. Fig. 5 (c) shows the device implanted in a freely moving rat, with a light-emitting diode (LED) indicator confirming active data acquisition from the bladder.
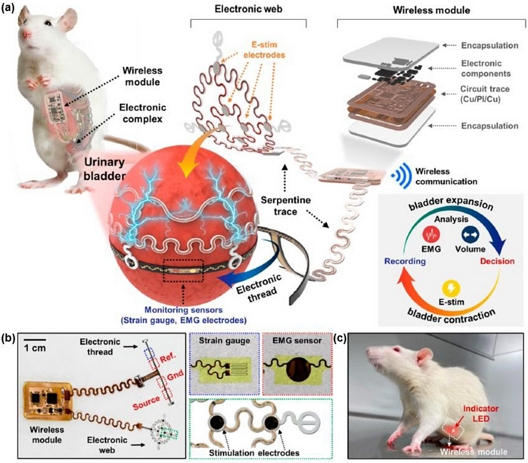
Implantable multimodal system for bladder function monitoring and electrical stimulation. (a) Schematic of the expandable electronic web wrapping around the bladder, incorporating strain gauges, EMG electrodes, and stimulation electrodes, connected to a wireless module. (b) Optical image of the complete system with inset views of individual sensing and stimulation components. (c) Photograph of a rat implanted with the system, showing an activated LED indicator during data acquisition. Adapted from Ref. [28].
By integrating sensing and electrical stimulation within a wireless, biocompatible form factor, this approach enables intelligent, self-regulating bladder management. Such bidirectional systems represent a potential pathway toward personalized closed-loop therapies for patients with detrusor underactivity or neurogenic bladders, reducing dependence on manual interventions and improving quality of life [17,28].
To guide modality selection and highlight practical trade-offs, Table 1 presents a side-by-side qualitative comparison of ultrasound, bioimpedance, and implantable strain-gauge approaches across four decision-critical axes: accuracy, invasiveness, power consumption, and long-term stability. Collectively, the comparisons emphasize the distinction between non-invasive wearables (ultrasound, bioimpedance) and higher-fidelity yet invasive implants (strain gauge), providing an at-a-glance reference to align technical capabilities with clinical context and engineering constraints.
4. CONCLUSIONS
The development of wearable and implantable bioelectronics for bladder function monitoring has advanced rapidly, driven by innovations in flexible electronics, soft materials, and wireless communication technologies. These systems enable high-performance, patient-friendly platforms capable of continuous, real-time data collection. Each technological approach offers distinct advantages. Wearable ultrasound-based devices provide direct anatomical measurements with high specificity, whereas wearable bioimpedance-based devices deliver low-cost, low-power alternatives well-suited for long-term home monitoring. Implantable strain gauge systems offer high-fidelity biomechanical data unaffected by external interference, while multimodal implants combine sensing with electrical stimulation to support closed-loop bladder management. The choice of modality should be tailored to the clinical context, balancing measurement accuracy, invasiveness, and patient comfort. These devices extend beyond diagnostic tools, representing a step toward personalized urological care that integrates continuous monitoring with adaptive therapeutic interventions.
Future research should prioritize multi-parameter platforms that integrate mechanical, electrical, and biochemical sensing to enable a more comprehensive assessment of bladder health. Closed-loop systems that combine sensing with automated neuromodulation or electrical stimulation hold considerable promise for the active management of neurogenic and underactive bladders. Realizing this potential will require explicit safety guardrails, human-in-the-loop overrides, verification of control latency, and transparent logging to support clinical auditability. Nevertheless, several practical challenges must be addressed before widespread clinical adoption can occur. Long-term biocompatibility and material stability are critical for extended use, particularly in implantable systems. Additionally, signal reliability must be ensured under diverse physiological conditions, including variations in posture, hydration, and abdominal pressure. Standardized stress-testing protocols should be established to quantify device performance under such perturbations and enable cross-study comparability. Power management remains a critical constraint, requiring a balance between device miniaturization and sufficient operating lifetime through battery capacity, wireless power transfer, or energy harvesting. Advances in energy harvesting from body motion, temperature gradients, and wireless power transfer may extend device lifetimes and eliminate the need for frequent recharging. Artificial intelligence-driven analytics hold the potential to transform continuous data streams into actionable clinical insights, supporting early detection and individualized treatment adjustments. Finally, scalable manufacturing, standardized testing, and multicenter clinical trials will be essential to translate these innovations from laboratory prototypes into widely adopted medical devices.
Acknowledgments
This research was supported by Global - Learning & Academic Research Institution for Master’s·PhD students, and Postdocs (G-LAMP) Program of the National Research Foundation of Korea (NRF) grant funded by the Ministry of Education (No. RS-2023-00285390). This research was supported by the Nano & Material Technology Development Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Science and ICT (RS-2025-25441823). This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education (RS-2025-25432139). This work was supported by the National Research Foundation of Korea (NRF), funded by the Korea government (MSIT) (RS-2025-24202976).
References
-
Y. Aoki, H.W. Broun, L. Brubaker, J.-N. Cornu, J.O. Daly, R. Cartwright, Urinary incontinence in women, Nat. Rev. Dis. Primers 3 (2017) 17042.
[https://doi.org/10.1038/nrdp.2017.42]

-
K.S. Coyne, C.C. Sexton, D.E. Irwin, Z.S. Kopp, C.J. Kelleher, I. Milsom, The impact of overactive bladder, incontinence and other lower urinary tract symptoms on quality of life, work productivity, sexuality and emotional well-being in men and women: results from the EPIC study, BJU Int. 101 (2008) 1388–1395.
[https://doi.org/10.1111/j.1464-410X.2008.07601.x]

-
Y. Cheng, T. Chen, G. Zheng, Z. Song, G. Zhang, X. Rao, et al., Prevalence and trends in overactive bladder among men in the United States, 2005–2020, Sci. Rep. 14 (2024) 16284.
[https://doi.org/10.1038/s41598-024-66758-8]

-
D.E. Irwin, L. Mungapen, I. Milsom, Z. Kopp, P. Reeves, C. Kelleher, The economic impact of overactive bladder in six western countries, BJU Int. 103 (2009) 202–209.
[https://doi.org/10.1111/j.1464-410X.2008.08036.x]

-
L.C. Powell, S.M. Szabo, D. Walker, K. Gooch, The economic burden of overactive bladder in the United States: A systematic literature review, Neurourol. Urodyn. 37 (2018) 1241–1249.
[https://doi.org/10.1002/nau.23477]

-
V. Phé, E. Chartier-Kastler, J.N. Panicker, Management of neurogenic bladder in patients with multiple sclerosis, Nat. Rev. Urol. 13 (2016) 275–288.
[https://doi.org/10.1038/nrurol.2016.53]

-
J.-J. Wyndaele, The management of neurogenic lower urinary tract dysfunction after spinal cord injury, Nat. Rev. Urol. 13 (2016) 705–714.
[https://doi.org/10.1038/nrurol.2016.206]

-
A.P. Cameron, D.E. Chung, E.J. Dielubanza, E. Enemchukwu, D.A. Ginsberg, B.T. Helfand, et al., The AUA/SUFU guideline on the diagnosis and treatment of idiopathic overactive bladder, J. Urol. 212 (2024) 11–20.
[https://doi.org/10.1097/JU.0000000000003985]

-
P.F.W.M. Rosier, W. Schaefer, G. Lose, H.B. Goldman, M. Guralnick, S. Eustice, et al., International Continence Society Good Urodynamic Practices and Terms 2016: Urodynamics, uroflowmetry, cystometry, and pressure-flow study, Neurourol. Urodyn. 36 (2016) 1243–1260.
[https://doi.org/10.1002/nau.23124]

-
M.C. Masone, A wearable UBVM device to monitor bladder volume, Nat. Rev. Urol. 21 (2024) 577.
[https://doi.org/10.1038/s41585-024-00944-1]

-
N.R. Bhatt, A. Pavithran, C.P. Ilie, L. Smith, R. Doherty, Post-prostatectomy incontinence: a guideline of guidelines, BJU Int. 133 (2023) 513–523.
[https://doi.org/10.1111/bju.16233]

-
A.P. Cameron, L. Campeau, B.M. Brucker, J.Q. Clemens, G.T. Bales, M.E. Albo, et al., Best practice policy statement on urodynamic antibiotic prophylaxis in the non-index patient, Neurourol. Urodyn. 36 (2017) 915–926.
[https://doi.org/10.1002/nau.23253]

-
C.K. Pfeuti, M. Przydacz, B.J. Linder, Urodynamics for Female Stress Urinary Incontinence: When and Why, Neurourol. Urodyn. (2025).
[https://doi.org/10.1002/nau.70073]

-
H. Hu, H. Huang, M. Li, X. Gao, L. Yin, R. Qi, et al., A wearable cardiac ultrasound imager, Nature 613 (2023) 667–675.
[https://doi.org/10.1038/s41586-022-05498-z]

-
C. Wang, X. Chen, L. Wang, M. Makihata, H.-C. Liu, T. Zhou, et al., Bioadhesive ultrasound for long-term continuous imaging of diverse organs, Science 377 (2022) 517–523.
[https://doi.org/10.1126/science.abo2542]

-
A. Hafid, S. Difallah, C. Alves, S. Abdullah, M. Folke, M. Lindén, et al., State of the art of non-invasive technologies for bladder monitoring: a scoping review, Sensors 23 (2023) 2758.
[https://doi.org/10.3390/s23052758]

-
F.A. Hassani, H. Jin, T. Yokota, T. Someya, N.V. Thankor, Soft sensors for a sensing-actuation system with high bladder voiding efficiency, Sci. Adv. 6 (2020) eaba0412.
[https://doi.org/10.1126/sciadv.aba0412]

-
S.J.A. Majerus, B. Hanzlicek, Y. Hacohen, D. Carbal, D. Bourbeau, M.S. Damaser, Wireless and Catheter-Free Bladder Pressure and Volume Sensor, IEEE Sens. J. 24 (2024) 7308–7316.
[https://doi.org/10.1109/JSEN.2023.3267749]

-
A. Kim, C.R. Powell, B. Ziaie, An Implantable Pressure Sensing System With Electromechanical Interrogation Scheme, IEEE Trans. Biomed. Eng. 61 (2014) 2209–2217.
[https://doi.org/10.1109/TBME.2014.2318023]

-
D.J. Young, P. Cong, M.A. Suster, M. Damaser, Implantable wireless battery recharging system for bladder pressure chronic monitoring, Lab Chip 15 (2015) 4338–4347.
[https://doi.org/10.1039/C5LC00821B]

-
C.-C. Wang, C.-C. Huang, J.-S. Liou, Y.-J. Ciou, I.-Y. Huang, C.-P. Li, et al., A Mini-Invasive Long-Term Bladder Urine Pressure Measurement ASIC and System, IEEE Trans. Biomed. Circuits Syst. 2 (2008) 44–49.
[https://doi.org/10.1109/TBCAS.2008.921601]

-
S.J.A. Majerus, S.J. Offutt, T.S. Brink, V. Vasoli, I. Mcadams, M.S. Damaser, Feasibility of Real-Time Conditional Sacral Neuromodulation Using Wireless Bladder Pressure Sensor, IEEE Trans. Neural Syst. Rehabil. Eng. 29 (2021) 2067–2075.
[https://doi.org/10.1109/TNSRE.2021.3117518]

-
L. Zhang, C. Marcus, D. Lin, D. Mejorado, S.J. Schoen, T.T. Pierce, et al., A conformable phased-array ultrasound patch for bladder volume monitoring, Nat. Electron. 7 (2024) 77–90.
[https://doi.org/10.1038/s41928-023-01068-x]

-
S.J.A. Majerus, P.C. Fletter, E.K. Ferry, H. Zhu, K.J. Gustafson, M. S. Damaser, Suburothelial Bladder Contraction Detection with Implanted Pressure Sensor, PLoS One 12 (2017) e0168375.
[https://doi.org/10.1371/journal.pone.0168375]

-
A.T. Toymus, U.C. Yener, E. Bardakci, Ö.D. Temel, E. Koseoglu, D. Akcoren, et al., An integrated and flexible ultrasonic device for continuous bladder volume monitoring, Nat. Commun. 15 (2024) 7216.
[https://doi.org/10.1038/s41467-024-50397-8]

-
L. Duan, M.-L. Jin, A biocompatible integrated bladder electronics for wireless capacity monitoring assessment, Soft Sci. 5 (2025) 4.
[https://doi.org/10.20517/ss.2024.46]

-
J. Kim, M.I. Bury, K. Kwon, J.-Y. Yoo, N.V. Halstead, H.-S. Shin, et al., A wireless, implantable bioelectronic system for monitoring urinary bladder function following surgical recovery, Proc. Natl. Acad. Sci. U.S.A. 121 (2024) e2400868121.
[https://doi.org/10.1073/pnas.2400868121]

-
J.H. Lee, T.-M. Jang, J.-W. Shin, B.H. Lim, K. Rajaram, W.B. Han, et al., Wireless, Fully Implantable and Expandable Electronic System for Bidirectional Electrical Neuromodulation of the Urinary Bladder, ACS Nano 17 (2023) 8511–8520.
[https://doi.org/10.1021/acsnano.3c00755]

-
Q. Hu, Z. Wu, Y. Tian, J. Wang, Z. Pan, Y. Yu, et al., A magnetic soft robotic system for intelligent bladder volume control, npj Flex. Electron. 9 (2025) 33.
[https://doi.org/10.1038/s41528-025-00401-y]

-
S. Biswas, Y. Lee, H. Choi, H. Kim, Current Development in Bio-implantable Sensors, J. Sens. Sci. Technol. 31 (2022) 403–410.
[https://doi.org/10.46670/JSST.2022.31.6.403]

-
B.K. Jung, Y. Yang, S.J. Oh, Advances in Non-Interference Sensing for Wearable Sensors: Selectively Detecting Multi-Signals from Pressure, Strain, and Temperature, J. Sens. Sci. Technol. 32 (2023) 340–351.
[https://doi.org/10.46670/JSST.2023.32.6.340]

-
Y.S. lee, S. Jeon, D.W. Kim, C. Pang, Biodegradable Adhesive Interfaces for Bioelectronic and Biomedical Applications, J. Sens. Sci. Technol. 34 (2025) 224–230.
[https://doi.org/10.46670/JSST.2025.34.3.224]

-
H. Guo, W. Bai, W. Ouyang, Y. Liu, C. Wu, Y. Xu, et al., Wireless implantable optical probe for continuous monitoring of oxygen saturation in flaps and organ grafts, Nat. Commun. 13 (2022) 3009.
[https://doi.org/10.1038/s41467-022-30594-z]

-
E.S. Sani, C. Xu, C. Wang, Y. Song, J. Min, J. Tu, et al., A stretchable wireless wearable bioelectronic system for multiplexed monitoring and combination treatment of infected chronic wounds, Sci. Adv. 9 (2023) eadf7338.
[https://doi.org/10.1126/sciadv.adf7388]

-
H. Kim, B. Rigo, G. Wong, Y.J. Lee, W.-H. Yeo, Implantable Electronics for Real-Time, Continuous Physiological Monitoring, Nano-Micro Lett. 16 (2024) 52.
[https://doi.org/10.1007/s40820-023-01272-6]

-
T. Kim, I. Park, Skin-interfaced Wearable Biosensors: A Mini-Review, J. Sens. Sci. Technol. 31 (2022) 71–78.
[https://doi.org/10.46670/JSST.2022.31.2.71]

-
N. Rubab, S.-W. Kim, Self-powered Sensors based on Piezoelectric Nanogenerators, J. Sens. Sci. Technol. 31 (2022) 293–300.
[https://doi.org/10.46670/JSST.2022.31.5.293]

-
H.T. Crane, J.A. Berkebile, S. Mabrouk, N. Riccardelli, O.T. Inan, Digital twin driven electrode optimization for wearable bladder monitoring via bioimpedance, npj Digit. Med. 8 (2025) 73.
[https://doi.org/10.1038/s41746-025-01441-4]

-
S. Abasi, J.R. Aggas, G.G. Garayar-Leyva, B.K. Walther, A. Guiseppi-Elie, Bioelectrical Impedance Spectroscopy for Monitoring Mammalian Cells and Tissues under Different Frequency Domains: A Review, ACS Meas. Sci. Au 2 (2022) 495–516.
[https://doi.org/10.1021/acsmeasuresciau.2c00033]

-
T.-M. Jang, J.H. Lee, H. Zhou, J. Joo, B.H. Lim, H. Cheng, et al., Expandable and implantable bioelectronic complex for analyzing and regulating real-time activity of the urinary bladder, Sci. Adv. 6 (2020) eabc9675.
[https://doi.org/10.1126/sciadv.abc9675]

-
X. Tian, Q. Zeng, S.A. Kurt, R.R. Li, D.T. Nguyen, Z. Xiong, et al., Implant-to-implant wireless networking with metamaterial textiles, Nat. Commun. 14 (2023) 4335.
[https://doi.org/10.1038/s41467-023-39850-2]


Seongjin Lim is an undergraduate student in the Department of Applied Chemistry and Biological Engineering at Ajou University. His research focuses on implantable bioelectronics for human healthcare monitoring.

Jihye Kim is an assistant professor in the Department of Molecular Science and Technology and the Advanced College of Bio-convergence Engineering at Ajou University. She received her B.S. and PhD degrees from the School of Advanced Materials Science and Engineering at Sungkyunkwan University, Korea, in 2015 and 2020, respectively. She then completed postdoctoral research at Northwestern University’s Center for Bio-Integrated Electronics (CBIE). Her research focuses on wearable and implantable bioelectronics for monitoring physiological fluids. For more information, please refer to http://jkim.ajou.ac.kr.

