
Perspectives of Defects for Gas Detecting Performance of WSe2 Nano Flakes Selenized from Tungsten Metal Thin Film
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Nitrogen dioxide (NO2) is a toxic gas that poses serious health risks even at low concentrations, necessitating the development of highly sensitive gas detectors. In this study, WSe2 nano flakes were synthesized by selenizing tungsten thin films through a simple and economical annealing process. The synthesis conditions were optimized to produce abundant nano flakes with various structural defects, including tungsten metal residues, step terraces, and mixed 2H/1T phases. Gas detecting tests demonstrated that the WSe2 nano flakes exhibit p-type semiconductor behavior. The response was stable and reproducible under repetitive gas exposure cycles. These enhanced defect properties are attributed to the presence of structural defects, which provide additional active sites and improve charge transfer. These perspectives highlight the importance of defect engineering in transition metal dichalcogenides-based gas detectors and provide valuable insights into the development of various high-performance gas detectors using WSe2 or other transition metal dichalcogenides materials.
Keywords:
WSe2 nano flake, Gas detection, Defects, Selenization1. INTRODUCTION
With the continuous development of science, technology, and industry, air pollution has become an increasingly serious problem. The main cause of air pollution is the toxic gases generated by fossil fuel combustion. The most representative toxic gas is NO2, which can cause terrible effects (headache, eye, or respiratory irritation) in humans at only 1 ppm [1]. Therefore, various materials have been developed for toxic gas detection [2-6], with the most representative being metal oxides and transition metal dichalcogenides (TMDCs) [7].
Metal oxides have high operating temperatures, high sensitivity, and low selectivity. Most importantly, they are very unstable in humid environments. Graphite-based detectors do not detect low concentrations of toxic gases and have slow recovery times [8]. In contrast, TMDC-based detectors have various advantages that can be utilized in gas detectors [9]. First, they have a very high specific surface area because of the basal plane and clear active sites on the edge. Additional advantages are observed in manufacturing TMDC materials for 3D nanostructures [10]. For example, they are optimal for active-site exposure. In addition, the voids formed between the nanoparticles constituting the 3D nanostructure are favorable for the diffusion of the target gas, which is advantageous for detector performance [11,12]. Although many studies have been conducted on optimizing the structure or maximizing the specific area, very little interest has been displayed in the defects or phases that may occur in the material when manufacturing these TMDC nanostructures employing various processes for use as gas detectors. This is because analysis and control are difficult to implement. However, interest in these factors is growing, making it important to expand the industry of commercial gas detectors using TMDCs.
In this study, to manufacture WSe2 nano flakes in a vertical 3D structure, the simplest and most economical process was used, which is to manufacture a tungsten (W) metal thin film by having it react with selenium vapor. Compared to other high-technology processes, this method is the most widely used in industry. Various defects of the WSe2 nano flakes were accurately manufactured using this method. The possible locations of the most important metal-phase residuals and their influence on gas detection performance are presented.
2. EXPERIMENTS
2.1 Fabrication of WSe2 nano-flake from W thin film
The substrate (c-plane Al2O3, Crystal Bank, SSP) was cleaned sequentially using acetone, isopropyl alcohol, and deionized water. The W thin film was deposited on the substrate through radio frequency (RF) magnetron sputter (base pressure was ~10-6 Torr), using the W target (ITASO, TW00LT0002, 2"Dia x 1/4"Th, 99.95% purity). Further heat treatment was performed in a tube furnace to convert the grown W thin films into WSe2 nano flakes. The base pressure was ~10-3 Torr, and heat treatment was performed with a flow of 100 sccm of He gas. The W thin film was placed at the center of the quartz tube. Selenium powder (Alfa Aesar, item No. 10 235 262) was loaded into an alumina boat and placed 18 cm away near the gas inlet side. After annealing, the chamber was allowed to cool to room temperature.
2.2 Analysis
The crystal structures of the obtained powders were examined by powder X-ray diffraction (XRD; ULTIMA III, Rigaku; scan rate = 2° min-1, Japan) using Cu-Kα radiation (λ = 0.154 nm). The morphologies of the particles were analyzed using field-emission scanning electron microscopy (FE-SEM; JEOL, JSM-7900F, accelerating voltage = 10 kV, Japan) and high-resolution transmission electron microscopy (HR-TEM; JEOL, JEM-2100, accelerating voltage = 200 kV, Japan).
Using silver paste, Cu wires were attached to both sides of the gas detector to fabricate the electrodes. The specimen was placed in a sealed chamber, controlling the flow of dry air and NO2 gas through mass flow controllers. The NO2 gas was diluted using dry air to achieve the desired concentration. Relative humidity (between 7% and 61%) was controlled by increasing the water temperature of the water bath system. Time-dependent responses of the specimens were measured using a source meter (Model 2400, Keithley, USA).
3. RESULTS AND DISCUSSIONS
In a high-temperature heat-treatment atmosphere, the W thin films gradually agglomerate, even though the temperature is much lower than the melting point. When selenium vapor is attached to the agglomerated tungsten, WSe2 nucleates as tungsten is consumed, and it gradually grows along the basal plane into a nanoflake shape. The WSe2 nano flakes grow with a thermodynamically stable hexagonal facet. Even after the reaction, a small amount of the tungsten metal phase may remain, which is a residue that does not fully react because of the high vapor pressure of selenium. This growth process is illustrated in Fig. 1 (a). XRD images of a 100 nm thick W thin film are displayed as a function of selenization temperature in Fig. 1 (b). Selenization hardly occurred at 600℃, as no peak related to WSe2 are observable. At 700℃, WSe2 grows by selenization, and a tungsten metal phase residue remains. At 800℃, only tungsten metal remains, and the WSe2 phase does not appear. This is because selenium vapor is not adsorbed on the W thin film; thus, no reaction occurs. When heat-treated at 900℃, only the crystallinity of the tungsten metal increases. In the SEM image displayed as a function of selenization temperature in Fig. 1 (c), a slight sharp flake edge is observed when heat-treated at 600℃; however, the thermal energy is insufficient to achieve crystallinity. Another characteristic is that empty space exists between the flakes (pink dashed line circle), which is consumed as the tungsten thin film reacts with WSe2, resulting in the appearance of a void. At 700℃, the flake shapes become more abundant. At 800℃, only tungsten metal remains, and at 900℃, only agglomeration in the form of nano dots occur. The reaction at 700℃ was determined to be the most suitable.
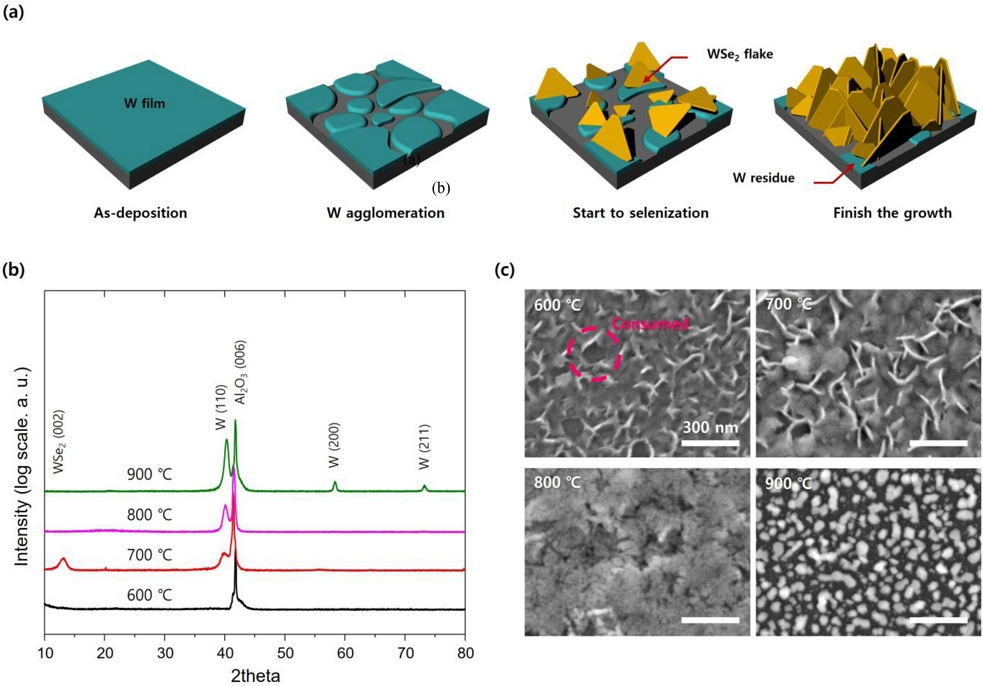
WSe2 nano-flake according to annealing temperature: (a) Schematic of WSe2 nano-flake growth from tungsten thin film. (b) XRD and (c) SEM images of WSe2 nano-flake as a function of selenization temperature. (W thin film thickness: 100 nm)
The amount of tungsten suitable for the growth of WSe2 nano flakes was determined by controlling the thickness of the thin W film. As shown in the SEM image in Fig. 2 (a), when the thickness is reduced, the density of the WSe2 nano flakes increases as the W thin film is sufficiently consumed. This means that as the amount of tungsten decreases, agglomeration and diffusion occur more easily due to thermal energy, and the selenization reaction becomes more favorable. At 50 nm, tungsten is sufficiently consumed, and the nano flakes are more abundant. This abundant structure formation is a positive factor for gas detection performance because it creates voids between the nanoparticles, which is advantageous for the diffusion of the gas to be detected. The XRD data in Fig. 2 (b) show a similar trend. At a thickness of 10 nm, the tungsten film is almost completely consumed and grows into WSe2 nano flakes. However, at a thickness of 50 nm, although WSe2 nano flakes are abundant, the tungsten metal phase remains. This tungsten residue plays a role in stabilizing the sensitivity and improving the performance of the detector with a nanostructured thin film. The detector performance of the WSe2 nano flakes as a function of tungsten thickness is shown in Fig. S1. For the 50 nm thickness, the baseline of the current value is high, and the current noise is minimized during detection. This can be attributed to the effect of not only the increased density of the WSe2 nano flakes, but also the metal phase residue. Fig. 2 (c) shows a cross-sectional SEM image and EDS results of the nano flakes for a 50 nm W thin film. Point 1 is the part that directly contacts the selenium vapor during annealing; therefore, selenium is completely sufficient, whereas the ratio of selenium decreases sharply at point 2. A closer look at the cross-sectional SEM image reveals only a WSe2 flake-shaped particle at point 1, and point 2 reveals dot-shaped particles, not flakes. These dot particles are inferred to be metal residues. The location of these metal residues is also crucial. Since the gas-sensing region should be only provided by WSe2, the top surface (point 1) should be free of metal residues. Conversely, the path through which gas attaches to the sensing region transmits the electrical signal to the bottom interface (point 2). In addition, among the target gases, in certain cases, strong oxidizing properties are detected, and metal residues (especially tungsten) can be highly oxidized by the target gas; however, oxidation can degrade the effectiveness of the metal residue. This can be avoided by positioning the metal residue at Point 2. Thus, the metal-phase residue is distributed at the boundary with the substrate and is involved in the amplification and stability of the electrical signal rather than directly participating in NO2 gas detection.
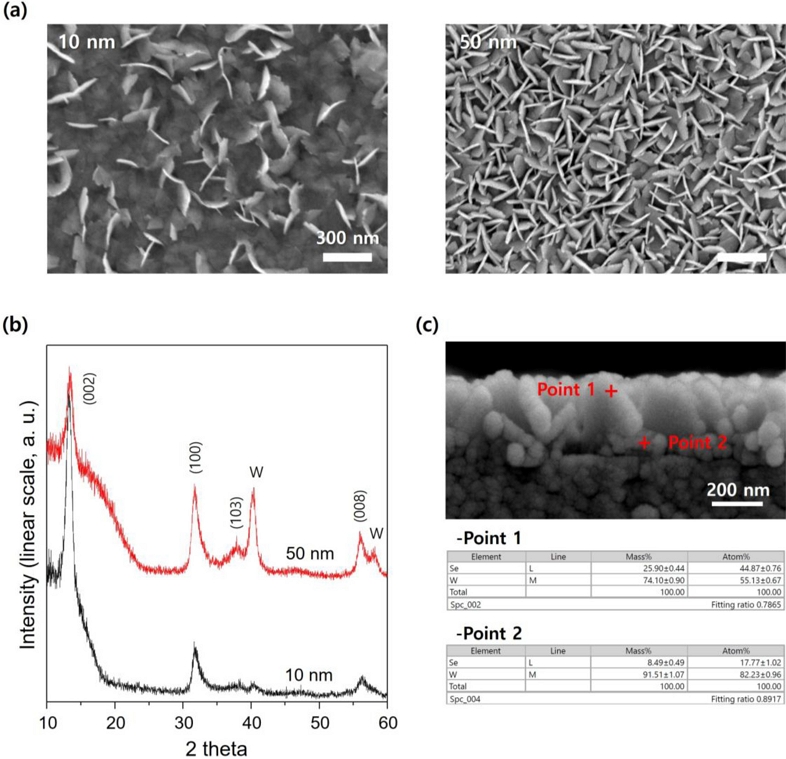
WSe2 nano-flake according to metal film thickness: (a) SEM and (b) XRD images of WSe2 nano flake as a function of W thin film thickness. (c) Cross-section image and EDS data of WSe2 nano flake of 50 nm thickness W thin film.
The TEM image in Fig. 3 (a) shows that the WSe2 nano flakes are uniformly thin and hexagonal, and the high-magnification image in Fig. 3 (b) shows that the crystal structure has hexagonal closed packing (HCP). Notably, the step terrace is on the basal plane of the flakes (Fig. 3 (c)). Because WSe2 flakes (Fig. 3 (c)) and TMDC are thermodynamically more unstable at the edges than at the basal plane, they are active sites. The drawback of the WSe2 nano flakes is that the density of these edges is very low compared to that of the basal plane. Therefore, the edge caused by the step terrace in the basal plane can amplify the sensor sensitivity of the WSe2 nano-flake structure. A factor that can further amplify the sensitivity of the sensor is the 1T phase, which has a relatively high free energy. As shown in Fig. 3 (d), the two phases coexist in one nano flake. This provides more active sites, which can improve gas detection performance. To demonstrate the gas detection performance of these defects, the response of the WSe2 nano flakes to NO2 gas was measured using a simple method.

TEM images of WSe2 nano-flake: (a) low magnification, (b) high magnification and Fourier-filtered image of WSe2 nano-flake (Inset: corresponding Fourier-transformed diffraction pattern). (c) Image of step-terrace and (d) 2H, 1T phase of WSe2 nano-flake.
The NO2 gas detection performance of WSe2 nano flakes grown from 50 nm thick W thin films was evaluated at an operating temperature of 80℃. An electrode was manufactured to evaluate the performance of the gas detector (Fig. 4 (a)). The relative response shown in Fig. 4 (b) was obtained from the following equation;
| (1) |
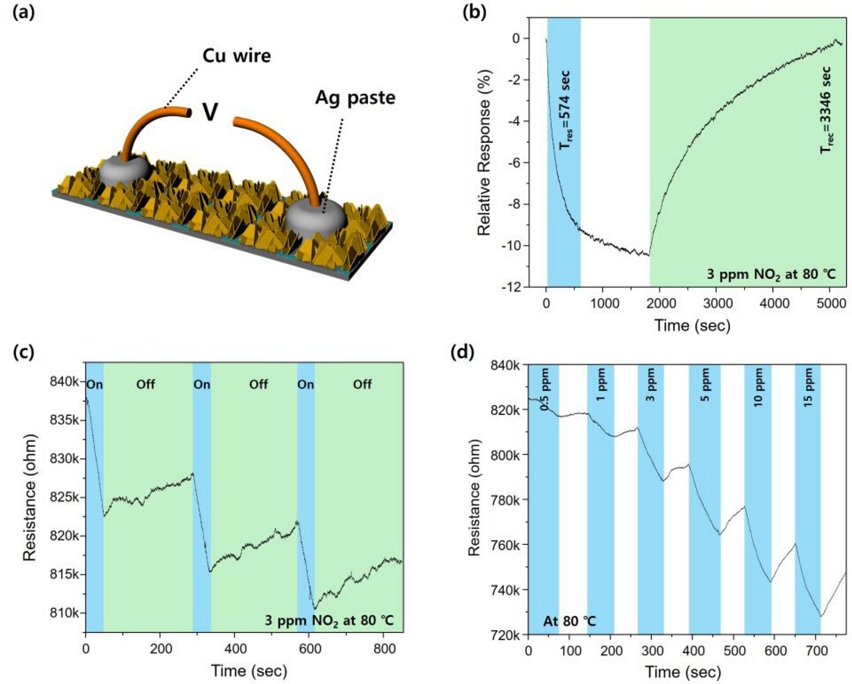
Responsivity of the fabricated gas detector: (a) Schematic of gas detector electrode. (b) Real-time relative response curve of WSe2 nano-flake at a concentration of 3 ppm NO2. (c) Real-time resistance changes of the detector in response to 3 ppm NO2 at 80℃ demonstrating repeatability. (d) Real-time response of WSe2 nano-flake at 80℃ and different NO2 concentrations. (W thin film thickness: 50 nm)
where Ra is the baseline resistance of the sensor in the background gas, and Rg is the resistance after exposure to the target gas. When NO2 gas was introduced, the resistance decreased because of the p-type semiconductor characteristics of WSe2. These p-type characteristics become more pronounced at the nanoscale, and very thin nano flakes can be synthesized using this method. The relative response increased to ~9% within 574 s and required 3346 s for recovery. This response was maintained under repeated NO2 gas on/off cycles (Fig. 4 (c)). Furthermore, the rate of resistance change varied with NO2 concentration (Fig. 4 (d)). This high performance is attributed to the various defects (metallic residues, step terraces, and thermodynamically unstable phases) that are easily generated by the simple selenization process of tungsten metal. The ability to generate such defects, along with the accurate identification and analysis methods presented in this study, is expected to serve as an important strategic asset for the development of NO2 gas sensors. Moreover, this approach can be extended to other TMDC materials beyond WSe2.
4. CONCLUSIONS
In this study, WSe2 nano flakes were synthesized by selenizing W thin films using a simple and economical method. The WSe2 nano flakes exhibited various structural defects, including tungsten metal residues, step terraces, and mixed phases, which played a crucial role in enhancing NO2 gas detection performance. The detector demonstrated a stable reproducible response to NO2 at an operating temperature of 80℃.
These results highlight the significance of defect engineering in TMDC-based gas sensors. By accurately identifying and controlling the defects generated during synthesis, electronic properties and active site availability can be tailored, thereby improving detector performance. The approach demonstrated herein can be extended to other TMDC materials, providing a valuable strategy for the development of highly sensitive and reliable gas detection devices for environmental monitoring applications.
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean Government (MSIT) (No. RS-2023-00208311).
References
-
Y. Duan, S. Feng, K. Zhang, J. Qiu, S. Zhang, Vertical Few-Layer WSe2 Nanosheets for NO2 Sensing, ACS Appl. Nano Mater. 4 (2021) 12043−12050.
[https://doi.org/10.1021/acsanm.1c02603]

-
H.-D. Lee, S. Lee, MoS2-based Ultra-Low-Power NH3 Gas Sensor with Room-Temperature Operation, J. Sens. Sci. Technol. 34 (2025) 159−162.
[https://doi.org/10.46670/JSST.2025.34.3.159]

-
D.H. Jeong, D.G. Jung, D. Jung, Fabrication and Evaluation of Single Layer Graphene/SnO2 Based Gas Sensor for NO2 Detection, J. Sens. Sci. Technol. 33 (2024) 493−498.
[https://doi.org/10.46670/JSST.2024.33.6.493]

-
I. Kim, E.D. Park, H.-S. Kim, S.-D. Han, Fabrication of Au-In2O3 Thin/Thick-Film Gas Sensors and Their Sensing Characteristics for Toxic Gases, J. Sens. Sci. Technol. 33 (2024) 516−524.
[https://doi.org/10.46670/JSST.2024.33.6.516]

-
T.W. Ha, D.-Y. Lim, C.-H. Lee, High-Temperature Stable NOx Sensor for Exhaust Gas Monitoring in Automobiles, J. Sens. Sci. Technol. 33 (2024) 439−447.
[https://doi.org/10.46670/JSST.2024.33.6.439]

-
S.L. Jeon, J. Lee, S.E. Kim, J. Park, Optimization of Growth Environments Based on Meteorological and Environmental Sensor Data, J. Sens. Sci. Technol. 33 (2024) 230−236
[https://doi.org/10.46670/JSST.2024.33.4.230]

-
E. Llober, Transition metal dichalcogenide based toxic gas sensing, Curr. Opin. Environ. Sci. Health 37 (2024) 100533.
[https://doi.org/10.1016/j.coesh.2024.100533]

-
A. Sharma, U. Varshney, A. Yadav, P. Vashishtha, P. Singh, G. Gupta, Fabrication of ultra-sensitive NO sensor based on vacuum selenized WSe2 nanorods, Mater. Chem. Phys. 296 (2023) 127241.
[https://doi.org/10.1016/j.matchemphys.2022.127241]

-
L.-H. Cheng, U. Kumar, Z.-Y. Deng, C.-H. Wu, Improved Charge Transfer for NO2 Gas Sensors by Using 0D SnS Quantum Dot/2D WSe2 Heterostructures, ACS Appl. Nano Mater. 6 (2023) 9506−9514.
[https://doi.org/10.1021/acsanm.3c01181]

-
J.H. Kim, S.E. Lee, J.H. Park, H.J. Jin, J.S. Choe, J. Ko, et al., Next-Generation Sensors with Three-Dimensional Micro-/Nano-Structures, J. Sens. Sci. Technol. 33 (2024) 419−428.
[https://doi.org/10.46670/JSST.2024.33.6.419]

-
A. Alagh, F.E. Annanouch, A. Sierra-Castillo, E. Haye, J.-F. Colomer, E. Llobet, Three-Dimensional Assemblies of Edge-Enriched WSe2 Nanoflowers for Selectively Detecting Ammonia or Nitrogen Dioxide, ACS Appl. Mater. Interfaces 12 (2022) 54946−54960.
[https://doi.org/10.1021/acsami.2c16299]

-
Y. Duan, S. Zhang, Y. Yu, J. Qiu, S. Feng, Facile microwave plasma driven 3D-WSe2 2H-1T phase modulation for improving NO2 gas sensing performance, Sens. Actuators B Chem. 387 (2023) 133822.
[https://doi.org/10.1016/j.snb.2023.133822]
