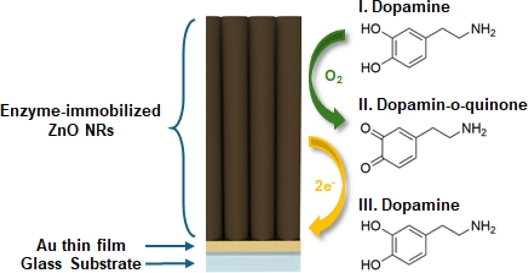
Nanostructured ZnO Interfaces for Highly Sensitive and Selective Dopamine Sensing
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
A tyrosinase-functionalized electrochemical biosensor based on vertically aligned ZnO nanorods (NRs) was developed for the selective and sensitive detection of dopamine. The sensor architecture features an Au working electrode modified with ZnO NRs, followed by sequential surface treatments using tetraethyl orthosilicate, 3-aminopropyl triethoxysilane, and glutaraldehyde to immobilize tyrosinase. Structural and chemical modifications were confirmed via scanning electron microscopy, energy-dispersive X-ray spectroscopy, and Fourier-transform infrared spectroscopy. Chronoamperometric measurements demonstrated a significant enhancement in sensing performance for the ZnO NRs-based sensor (3.6114 nA/μM) compared to a planar ZnO thin-film electrode (0.4318 nA/μM), with a nine-fold increase in sensitivity attributed to the larger enzyme loading area provided by the three-dimensional nanostructure. The sensor exhibited excellent linearity and a pronounced response at a low operating potential of -250 mV. Interference studies with ascorbic acid and norepinephrine confirmed high selectivity toward dopamine. These findings underscore the utility of nanostructured ZnO interfaces in advancing enzyme-based electrochemical biosensing platforms for neurotransmitter detection under physiologically relevant conditions.
Keywords:
Electrochemical biosensors, ZnO nanorods, Dopamine sensor, Enzyme immobilization1. INTRODUCTION
Dopamine is a critical neurotransmitter involved in numerous physiological processes like motor control, mood regulation, and cognitive function [1]. Abnormal dopamine levels are closely associated with neurological disorders, such as Parkinson’s disease, schizophrenia, and depression, necessitating the development of reliable detection platforms capable of operating under physiologically relevant conditions [2,3]. Among the available analytical techniques, electrochemical sensing has emerged as a powerful approach for the real-time, low-cost, and label-free detection of dopamine, particularly when integrated with enzyme-based selectivity [4].
Tyrosinase, an oxidase enzyme, facilitates the selective recognition of dopamine via enzymatic oxidation to o-dopaquinone, which can subsequently be electrochemically reduced to generate a measurable current (Fig. 1) [5]. However, the performance of tyrosinase-based sensors is primarily governed by the surface properties and morphology of the electrodes [6]. Conventional two-dimensional (2D) electrode surfaces often suffer from limited active surface areas and insufficient enzyme loading, restricting their sensitivity and dynamic range. These limitations are well recognized because the planar structure of 2D electrodes restricts the available surface for enzyme immobilization and constrains the mass transfer of analytes, ultimately leading to poor sensor performance [7].
Nanostructured materials, particularly vertically aligned zinc oxide nanorods (ZnO NRs), offer significant advantages owing to their high aspect ratios, large surface-to-volume ratios, and favorable electron-transport characteristics. Numerous studies have shown that three-dimensional (3D) nanostructured electrodes significantly enhance the effective surface area, thereby supporting greater enzyme immobilization and promoting efficient electron transfer between the biorecognition element and the electrode [8-10].
This study developed a 3D electrochemical biosensor platform by integrating tyrosinase into ZnO NRs arrays grown on gold (Au) electrodes. Surface functionalization was performed using an amine functionalization strategy involving tetraethyl orthosilicate (TEOS) and 3-aminopropyl triethoxysilane (APTES), followed by glutaraldehyde (GA) crosslinking for enzyme immobilization. A comparative analysis with a ZnO thin-film-based electrode was conducted to evaluate the impact of nanostructuring on the sensor performance in terms of sensitivity, selectivity, and signal stability.
This study highlights the role of 3D ZnO architectures in enhancing enzyme-based electrochemical sensing and offers a promising route for selective neurotransmitter detection.
2. EXPERIMENTAL
The Au electrodes were fabricated on glass substrates (5 × 50 mm2). A thin metal bilayer of Cr (7 nm) and Au (100 nm) was deposited via electron-beam evaporation, followed by photolithographic patterning and wet etching to define the working electrode (WE) geometry. A conformal ZnO seed layer (25 nm) was then deposited across the surface of the WE using RF sputtering in conjunction with a liftoff process to achieve a precise pattern definition. A polyimide (PI) encapsulation layer (Sigma-Aldrich, USA; ~2 μm thick) was applied by spin-casting and was subsequently cured in a vacuum oven (250°C, 2 h) to ensure thermal stability and mechanical integrity. Openings in the PI layer to expose the underlying WE were formed by reactive ion etching (RIE) under the following conditions: a chamber pressure of 100 mTorr, 20 sccm O2 flow rate, and 150 W RF power for 3–5 min.
Prior to the hydrothermal growth of the ZnO NRs, a photocurable epoxy was spin-cast onto the substrate to serve as a masking layer. Photolithographic patterning was used to define openings exclusively on the WE surface, thereby localizing the subsequent ZnO NR formation. Hydrothermal growth was performed in an aqueous nutrient solution composed of zinc nitrate hexahydrate and hexamethylenetetramine (HMTA) in a 1:1 molar ratio. Following growth, the epoxy mask was thoroughly removed using acetone, and the ZnO NRs were baked at 150°C for 15 min on a hotplate to enhance the adhesion. Residual debris was eliminated by brief ultrasonication in deionized (DI) water (1 min).
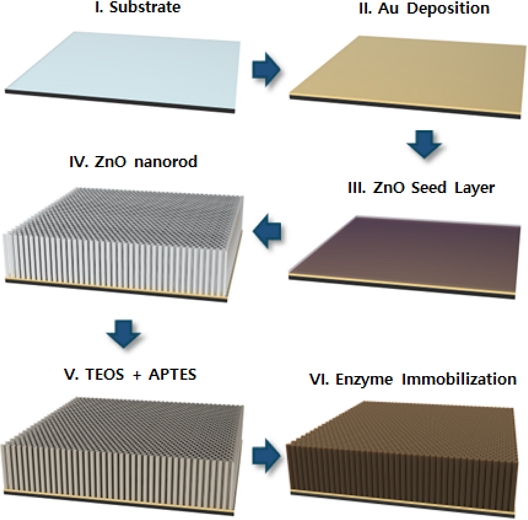
Schematic illustration of the fabrication process for a dopamine-sensitive electrochemical biosensor based on enzyme-functionalized ZnO NRs.
Before fabricating the ZnO NRs, spin-casting photocurable epoxy was used to form a masking layer and opened only on the WE surface by photolithography for a hydrothermal process for ZnO NRs growth with a reaction nutrient solution composed of a 1:1 ratio of zinc nitrate hexahydrate to HMTA. After growth, the masking layer was thoroughly removed with acetone, and the ZnO NRs were baked for 15 min at 150°C on a hotplate. Finally, ultrasonication (1 min) was performed to remove any residues.
A silica solution was prepared by mixing ethanol (30 mL), aqueous ammonia (1.5 mL), and TEOS (0.1 mL). The WE tip was then immersed in the solution under constant stirring at room temperature for 2 h. Following silanization, the WE tip was thoroughly rinsed, sonicated with ethanol, and dried in an oven at 60°C to remove residual silane compounds. APTES (40 μL) was added to the previously prepared TEOS solution to yield an APTES-functionalization mixture for subsequent amine-functionalization. The silanized WE tip was immersed in this solution and stirred at room temperature for 4 h, resulting in the covalent attachment of amine groups (–NH2) to the ZnO NRs surfaces. Upon completion of the reaction, the probe was cleaned using sequential ultrasonic baths in DI water and ethanol, followed by drying at room temperature to remove the unbound reaction byproducts.
To enable tyrosinase (EC 1.14.18.1 2840 U mg-1, from mushroom) immobilization onto the amine-functionalized ZnO NRs, the tip of the WE was immersed in a GA solution (2.5% in 0.1 M phosphate buffer, pH 5.8) under gentle stirring at room temperature for 3 h to facilitate crosslinking. Following crosslinking, the WE was rinsed thoroughly with DI water, immediately immersed in a tyrosinase enzyme solution (5 mg mL-1 in 0.05 M phosphate-buffered saline, PBS), and incubated overnight at 4°C to allow for effective immobilization of the enzyme onto the functionalized ZnO NRs. The unbound enzyme was removed by extensive washing with DI water.
Electrochemical measurements, including cyclic voltammetry (CV) and chronoamperometry (CA), were performed using a potentiostat (Gamry Reference 1010E) in a standard three-electrode cell system. The as-prepared tyrosinase-modified electrode served as the WE. A saturated Ag/AgCl (1 M) electrode and a platinum wire were employed as the reference (RE) and counter electrodes (CE), respectively. All experiments were conducted in a 10 mL electrochemical cell containing 0.05 M PBS (pH 7.4) at room temperature. CV scans were performed at a scan rate of 150 mV s-1 unless otherwise noted.
A field-emission scanning electron microscope (FE-SEM, Hitachi S4200) equipped with an energy-dispersive X-ray spectrometer (EDS, Bruker) and a Fourier-transform infrared spectra (FTIR, Thermo Scientific) was used for surface analysis.
3. RESULTS AND DISCUSSIONS
Fig. 3 presents a series of SEM images illustrating the stepwise process of hydrothermally grown ZnO NRs on the WE, highlighting the critical role of sonication and the morphological transitions associated with each functionalization step. In the initial stage, the as-grown ZnO NRs surface exhibited scattered dark regions corresponding to residual byproducts generated during the hydrothermal synthesis, as shown in Fig. 3 (a). Following sonication, these residues are effectively removed, resulting in a cleaner and more uniform surface, as shown in Fig. 3 (b). This confirmed that sonication effectively removed nonspecific ZnO debris, thereby providing a cleaner interface for subsequent enzyme immobilization.

SEM images of the surface modification of ZnO NRs on the WE for enzyme immobilization; (a) as-prepared ZnO NRs, (b) as-prepared ZnO NRs after sonication, (c) after TEOS silanization with unbound byproducts (red-dotted regions), (d) Sonicated sample after TEOS silanization, (e) after APTES functionalization, (f) after GA crosslinking, (g) after tyrosinase immobilization.
Subsequent silanization using TEOS led to a visible surface deposition, as shown in Fig. 3 (c). Without sonication, unbound silane byproducts persisted on the surface, particularly in the red-dotted regions. In contrast, the sonicated counterpart exhibited a markedly cleaner morphology, indicating that post-silanization sonication was effective in eliminating loosely attached silane residues, as shown in Fig. 3 (d). This clean interface is essential for enabling consistent and efficient coupling chemistry in the subsequent functionalization stages.
Amine functionalization via APTES resulted in a thin conformal coating on the ZnO NRs, as observed in Fig. 3 (e). This modification provided reactive amine groups for GA crosslinking, which introduced a roughened texture on the nanostructure surface, as shown in Fig. 3 (f), suggesting successful crosslinker attachment. After tyrosinase immobilization, the surface underwent an additional morphological transition characterized by a denser and more irregular topography, consistent with the formation of an enzyme layer on the functionalized ZnO NRs, as shown in Fig. 3 (g). These morphological transitions collectively support the sequential and controlled assembly of functional interfaces on the ZnO NRs.
Fig. 4 presents the EDS spectra that track the sequential surface modification of ZnO NRs for enzyme immobilization. The initial spectrum confirms the successful formation of ZnO NRs with strong signals from Zn and O, as shown in Fig. 4 (a). Following amine functionalization using TEOS (Fig. 4 (b)) and APTES, additional peaks corresponding to Si, N, and C emerged, consistent with silane attachment and amine group incorporation, as depicted in Fig. 4 (c). After tyrosinase immobilization, the intensities of the N, C, and O signals increased further, indicating successful enzyme binding to the functionalized ZnO NRs surface (Fig. 4 (d)).
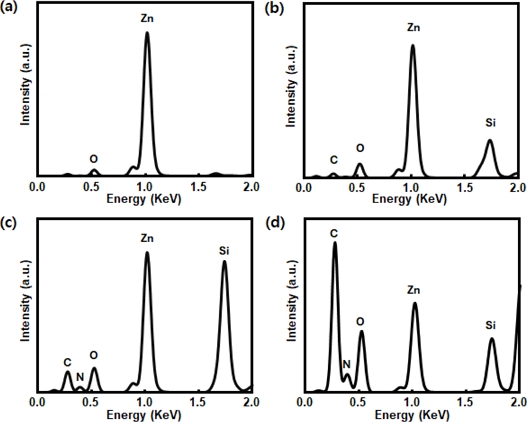
EDS spectra of ZnO NRs at various stages of surface modification; (a) as-prepared ZnO NRs, (b) after TEOS treatment, (c) after APTES functionalization, (d) after tyrosinase immobilization.
Fig. 5 presents FTIR spectra (400–4000 cm-1) of ZnO NRs at key stages of surface modification for enzyme immobilization. In Fig. 5 (a), the spectrum of as-prepared ZnO NRs displays a prominent absorption band around 430 cm-1, corresponding to the Zn-O stretching vibration [11]. After silanization and amine functionalization with TEOS and APTES, as shown in Fig. 5 (b), additional peaks appeared at 1020 cm-1, associated with Si-O-Si asymmetric stretching [12], while a broad band near 3300 cm-1 was attributed to N-H stretching vibrations from the primary amine groups [13]. A band near 1570 cm-1 was also observed, corresponding to N-H bending [14], indicating successful introduction of amine functionalities on the ZnO surface. As shown in Fig. 5 (c), following GA crosslinking and tyrosinase immobilization, distinct amide bands emerged: amide I band around 1650 cm-1 (C=O stretching) [15] and amide II band near 1540 cm-1 (N-H bending and C-N stretching) [16]. These features provide strong evidence for the presence of proteins and confirm the covalent effect of tyrosinase on the functionalized ZnO NRs.
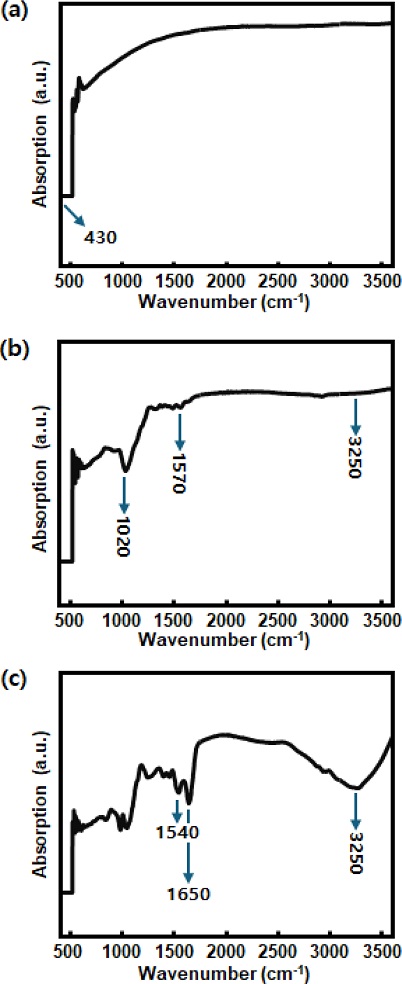
FTIR spectra of ZnO NRs at various stages of surface modification; (a) as-prepared ZnO NRs, (b), after TEOS/APTES functionalization, (c) after GA crosslinking and tyrosinase immobilization.
Fig. 6 shows the CV response of the tyrosinase-functionalized ZnO NRs electrode in 0.05 M PBS, with and without dopamine. In the absence of dopamine (grey dotted line), only the baseline capacitive current was observed. Upon dopamine addition (red solid line), a pronounced cathodic peak appeared near -250 mV, corresponding to the electrochemical reduction of enzymatically generated o-dopaquinone. This distinct response confirms the enzyme-mediated oxidation of dopamine and establishes an optimal potential window for subsequent chronoamperometric measurements.
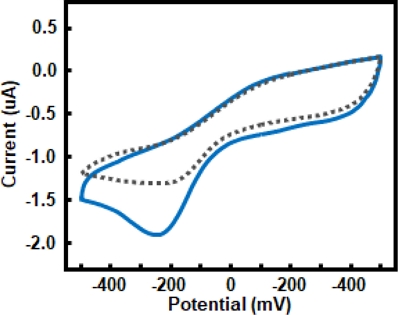
CV curves of the sensing electrode in 0.05 M PBS (pH 7.4) without (gray dotted line) and with 6 mM dopamine (blue solid line).
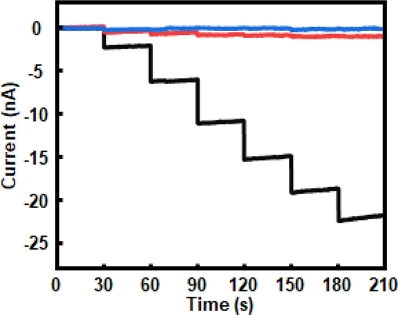
Fig. 7 illustrates the amperometric responses of enzyme-functionalized electrodes with varying surface morphologies upon successive additions of 1 μM dopamine every 50 s in 0.05 M PBS (pH 7.4) at -250 mV, as indicated by arrows. As shown in Fig. 7 (a), the sensing electrode composed of a planar ZnO thin film (25 nm) on Au, functionalized sequentially with TEOS, PATES, GA, and tyrosinase, exhibited a stepwise increase in current with dopamine addition. Fig. 7 (b) shows the response of an electrode featuring vertically aligned ZnO NRs on Au with the same surface chemistry. The NR-based sensor displayed a markedly higher current response, attributed to the increased surface area, enabling greater enzyme loading and enhanced sensitivity.
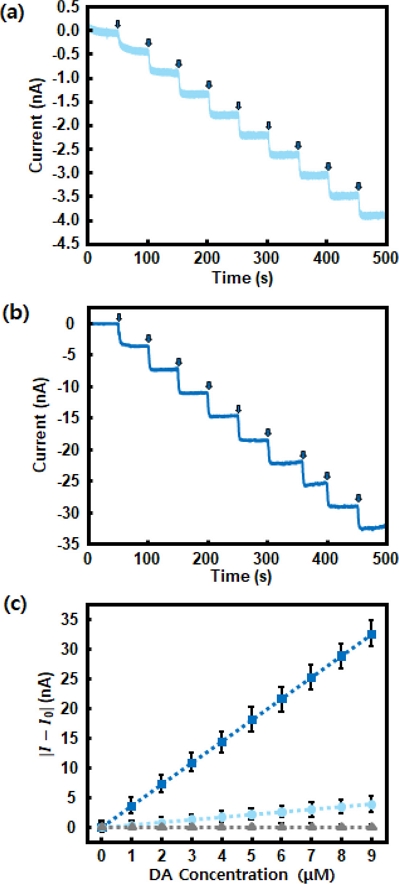
Chronoamperometric responses and corresponding calibration curves of tyrosinase-based dopamine sensors measured at -250 mV; (a) ZnO thin-film-based electrode functionalized with TEOS, APTES, GA, and tyrosinase, (b) ZnO NRs-based electrode with identical surface modification, (c) corresponding calibration curves for bare Au (gray triangle), ZnO thin-film-based electrode with identical surface modification (light blue circle), and ZnO NRs-based electrode with identical surface modification (blue square).
To quantitatively compare the sensor performance, Fig. 7 (c) presents the calibration curves derived from the three electrode configurations: bare Au, enzyme-functionalized ZnO thin film, and enzyme-functionalized ZnO NRs. The bare Au electrode shows negligible sensitivity (slope: 0.0025 nA/μM, R2 = 0.7989), indicating minimal electrochemical response to dopamine. The thin-film-based electrode demonstrates significant improvement, with a sensitivity of 0.4318 nA/μM and excellent linearity (R2 = 0.9998). Notably, the NRs-based electrode exhibits a nine-fold enhancement in sensitivity (3.6114 nA/μM, R2 = 0.9999), confirming the superior electrochemical performance of the 3D structure of dopamine detection.
These results highlight the critical role of electrode surface morphology in determining enzyme-based electrochemical biosensor performance. Although both the 2D (ZnO thin film) and 3D (ZnO NRs) electrodes were identically functionalized with tyrosinase, the 3D geometry enabled a much higher density of immobilized enzymes and improved analyte accessibility, leading to enhanced electrochemical signal transduction. The strong linear correlation and high sensitivity observed for the ZnO NR-based sensors underscore their potential for sensitive and reliable dopamine detection in physiological environments.
Fig. 8 presents the selectivity profile of the tyrosinase-functionalized ZnO NRs-based dopamine sensor in the presence of typical interfering species. Upon successive additions of 5 μM ascorbic acid at 30 s intervals, the sensor exhibited negligible current changes, indicating minimal cross-reactivity. Similarly, the addition of 1 μM norepinephrine at the same interval resulted in no significant current response. In contrast, the addition of 1 μM dopamine produced clear, stepwise increases in current with excellent linearity. These results demonstrate the high selectivity of the sensor toward dopamine, enabled by the enzyme-specific catalytic activity of tyrosinase and the engineered interface.
4. CONCLUSIONS
The integration of vertically aligned ZnO NRs with enzyme immobilization chemistry enabled the development of high-performance electrochemical platforms for dopamine sensing. The electrode was constructed by sequentially layering a glass substrate, an Au WE, ZnO NRs, and tyrosinase immobilized through silanization and crosslinking. This architecture provides a stable and high-surface-area interface that supports efficient bioelectrocatalytic activity.
Compared to a planar ZnO thin-film electrode, the NR-based sensor exhibited markedly improved sensitivity, which was attributed to greater enzyme loading and enhanced analyte interaction. Electrochemical measurements confirmed strong linearity, reliable response at low potential, and high selectivity for dopamine over standard interferents like ascorbic acid and norepinephrine. These results demonstrate the utility of 3D nanostructured interfaces in improving the performance of enzyme-based electrochemical biosensors for neurotransmitter detection.
Acknowledgments
This study was supported by the 2024 Hannam University Research Fund.
References
-
A. Nieoullon, Dopamine and the regulation of cognition and attention, Prog. Neurobiol. 67 (2002) 53–83.
[https://doi.org/10.1016/S0301-0082(02)00011-4]

-
E. Reinertsen, G.D. Clifford, A review of physiological and behavioral monitoring with digital sensors for neuropsychiatric illnesses, Physiol. Meas. 39 (2018) 05TR01.
[https://doi.org/10.1088/1361-6579/aabf64]

-
D. Weintarub, D. Aarsland, K.R. Chaudhuri, R.D. Dobkin, A.F. Leentjens, M. Rodriguez-Violante, A. Schrag, The neuropsychiatry of Parkinson’s disease: advances and challenges, Lancet Neurol. 21 (2022) 89–102.
[https://doi.org/10.1016/S1474-4422(21)00330-6]

-
Z. Fredj, M. Sawan, Advanced nanomaterials-based electrochemical biosensors for catecholamines detection: challenges and trends, Biosensors 13 (2023) 211.
[https://doi.org/10.3390/bios13020211]

-
F. Monica, M. David, Tyrosinase-Based Biosensors for Selective Dopamine Detection, Sensors 17 (2017) 1314.
[https://doi.org/10.3390/s17061314]

-
Z. Lu, Y. Zhang, Y. Shen, M. Li, Tyrosinase modified poly (thionine) electrodeposited glassy carbon electrode for amperometric determination of catechol, Electrochemistry 85 (2017) 17–22.
[https://doi.org/10.5796/electrochemistry.85.17]

-
J. Wang, V. Malgras, Y. Sugahar, Y. Ymauchi, Electrochemical energy storage performance of 2D nanoarchitectured hybrid materials, Nat. Commun. 12 (2021) 3563.
[https://doi.org/10.1038/s41467-021-23819-0]

-
H. Wu, X. Zhang, C. Wei, C. Wang, M. Jiang, X. Hong, et al., Modular assembly of enzyme loaded nanoparticles in 3D hollow fiber electrode for electrochemical sensing, Chem. Eng. J. 421 (2021) 129721.
[https://doi.org/10.1016/j.cej.2021.129721]

-
Z. Pu, J. Tu, R. Han, X. Zhang, J. Wu, C. Fang, et al., A flexible enzyme-electrode sensor with cylindrical working electrode modified with a 3D nanostructure for implantable continuous glucose monitoring, Lab Chip 18 (2018) 3570–3577.
[https://doi.org/10.1039/C8LC00908B]

-
C. Zhang, Q. Lai, W. Chen, Y. Zhang, L. Mo, Z. Liu, Three-Dimensional Electrochemical Sensors for Food Safety Applications, Biosensors 13 (2023) 529.
[https://doi.org/10.3390/bios13050529]

-
G. Nagaraju, S.A. Prashanth, M. Shastri, K.V. Yathish, C. Anupama, D. Rangappa, Electrochemical heavy metal detection, photocatalytic, photoluminescence, biodiesel production and antibacterial activities of Ag-ZnO nanomaterial, Mater. Res. 94 (2017) 54–63.
[https://doi.org/10.1016/j.materresbull.2017.05.043]

-
Q. Hu, H. Suzuki, H. Gao, H. Araki, W. Yang, T. Noda, High-frequency FTIR absorption of SiO2/Si nanowires, Chem. Phys. Lett. 378 (2003) 299–304.
[https://doi.org/10.1016/j.cplett.2003.07.015]

-
T. Threlfall, The infrared spectra of amides. Part 1. The stretching vibrations of primary carboxamides, Vib. Spectrosc. 121 (2022) 103386.
[https://doi.org/10.1016/j.vibspec.2022.103386]

-
B.C. Smith, Infrared Spectroscopy of Polymers XIII: Polyurethanes, Spectroscopy 38 (2023) 14–16.
[https://doi.org/10.56530/spectroscopy.fn3378a3]

-
D. Usoltsev, V. Sitnikova, A. Kajava, M. Uspenskaya, Systematic FTIR spectroscopy study of the secondary structure changes in human serum albumin under various denaturation conditions, Biomolecules 9 (2019) 359.
[https://doi.org/10.3390/biom9080359]

-
L.M. Miller, M.W. Bourass, R.J. Smith, FTIR spectroscopic imaging of protein aggregation in living cells, Biochim. Biophys. Acta Biomembr. 1828 (2013) 2339–2346.
[https://doi.org/10.1016/j.bbamem.2013.01.014]