Non-Invasive Glucose Measurement Using Electric Field and Temperature Sensor
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
A novel sensor system is presented for the non-invasive measurement of blood glucose concentration by applying an electric field to the skin. Unlike conventional invasive methods, the proposed sensor estimates glucose levels indirectly by analyzing temperature variations caused by dielectric loss, which arises from interactions between the electric field and glucose molecules. Experimental results across a glucose concentration range of 50–400 mg/dL demonstrated optimal performance, with the highest linearity and sensitivity achieved at 50 kHz and 24 V. Additionally, as the glucose concentration increased, the sensor output gradually decreased, exhibiting a strong linear relationship between the glucose level and the thermal response. In experiments simulating physiological conditions with the addition of 0.9% NaCl, the decreasing trend was more pronounced, confirming the sensor’s high responsiveness and reliability in electrolyte environments. The proposed sensor system has the potential to overcome the limitations of invasive techniques and improve the quality of life for patients with diabetes mellitus.
Keywords:
Dielectric loss, Electric field, Non-invasive glucose monitoring1. INTRODUCTION
Diabetes is a chronic metabolic disorder associated with significant complications affecting the cardiovascular system, kidneys, nerves, and vision. According to the World Health Organization (WHO), more than 95% of diabetes cases are classified as type 2 diabetes [1]. In 2024, an estimated 589 million adults—representing approximately 11% of the global population—were living with diabetes, and this figure is projected to increase to 853 million (12–13%) by 2050, as reported by the International Diabetes Federation (IDF) [2].
In Korea, the prevalence of diabetes among adults aged 30 years and older was 15.5% in 2022, with a markedly higher rate of 29.3% observed in those aged 65 years and older. An estimated 5.26 million individuals were living with diabetes; among them, 74.7% were aware of their diagnosis, and 70.9% were receiving treatment. However, only 32.4% of patients achieved the target blood glucose level of 120 mg/dL or lower [3]. The occurrence of diabetes in Korea has more than doubled since 1998, according to trend analyses from the Korea National Health and Nutrition Examination Survey (KNHANES). Furthermore, projections from the Korean Diabetes Association estimate that by 2030, nearly 18.9% of adults aged 20–79 years will be affected [4].
Conventional invasive blood glucose measurement methods, which have long been the standard for diabetes management, are associated with pain, risk of infection, psychological burden, and low compliance. Consequently, there is a growing demand for the development of non-invasive blood glucose monitoring technologies.
Over the past few decades, non-invasive blood glucose monitoring technologies have advanced considerably, driven by the development of diverse sensor modalities, including optical methods such as Near-Infrared (NIR) spectroscopy and Optical Coherence Tomography(OCT) [5,6], electrochemical methods [7], microwave sensing [8], and photoacoustic detection [9,14]. These emerging technologies demonstrate strong potential for commercialization, particularly through applications such as wearable devices, sweat-based biosensors, and smartphone-integrated systems [10,11].
However, these technologies present certain limitations. Recent reviews of non-invasive glucose monitoring methods have reported that optical approaches, such as NIR and OCT, typically yield mean absolute relative difference (MARD)—a standard metric of sensor accuracy representing the average percentage error between measured and reference values—in the range of 20–35% in clinical trials, often failing to meet ISO 15197 requirements. This international standard for self-monitoring blood glucose devices requires at least 95% of results to fall within ±15 mg/dL or ±15%. By contrast, electrochemical sensors based on enzyme reactions can achieve high sensitivity, with some systems demonstrating compliance with ISO 15197 and achieving Clarke Error Grid Zone A > 95%, where Zone A denotes clinically accurate results that would not lead to inappropriate treatment decisions. However, these methods remain constrained in achieving fully non-invasive measurement owing to issues such as electrode stability and user discomfort [12]. Microwave- and RF-based sensors have demonstrated resonance frequency shifts on the order of tens of kilohertz per 100 mg/dL change in glucose concentration, offering advantages of simple circuitry and low-power operation; however, they remain highly sensitive to environmental variations such as temperature and humidity [8]. Photoacoustic approaches, while reagent-free and promising for deeper detection, are still at the proof-of-concept stage, with low signal-to-noise ratios and insufficient clinical validation [9,14]. Collectively, these limitations underscore the urgent need for novel, non-invasive blood glucose monitoring technologies. Therefore, the development of novel methods is critically required to overcome these challenges [12,13,23].
Meanwhile, dielectric loss- or permittivity-based approaches offer advantages such as reduced susceptibility to ambient light and the elimination of reagent requirements. In microwave-based sensors, this strategy also enables lower circuit complexity, and experimental studies have reported resonance frequency shifts corresponding to changes in glucose concentration [15,16].
In this study, a novel method utilizing an electric field and a temperature sensor is proposed.
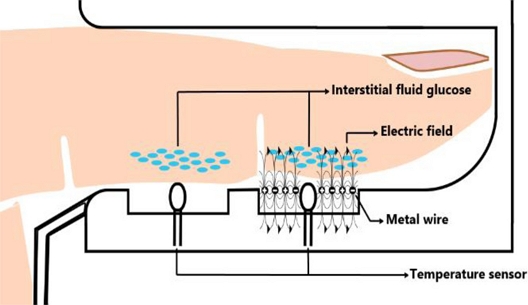
A schematic of the newly proposed non-invasive blood-glucose detector is shown in Fig. 1. In this structure, the thermistor output, which measures skin temperature without exposure to the electric field, serves as a reference. The output from the thermistor exposed to the electric field is then calibrated against this reference, enabling the measurement of glucose present in the interstitial fluid within the finger.
2. OPERATING PRINCIPLE AND CONFIGURATION
Glucose has a permanent dipole moment of 5.28 D, and when exposed to an oscillating electric field, it undergoes orientation polarization, which results in dielectric loss [17]. This dielectric loss arises from friction generated as the permanent dipoles of glucose rotate under the influence of the time-varying electric field.
The general expression for dielectric loss (P) can be written as
| (1) |
where V is the applied voltage,f is the frequency of the applied voltage,C0 is the capacitance of the electrodes, and represents the imaginary component of the complex relative dielectric constant under an AC electric field, commonly referred to as the dielectric loss factor [18].
Additionally, the relationship between the energy quantity and thermal variation of a general material can be expressed as
| (2) |
whereQ denotes the heat energy,ΔT is the change in temperature, andα is the specific heat capacity.
As dielectric loss is proportional to the generated heat change, monitoring temperature variation under constant voltage and frequency conditions enables the estimation of variations in the dielectric loss factor. As the dielectric loss factor of glucose is associated with glucose concentration, the glucose level can be determined by measuring the corresponding temperature increase [19].
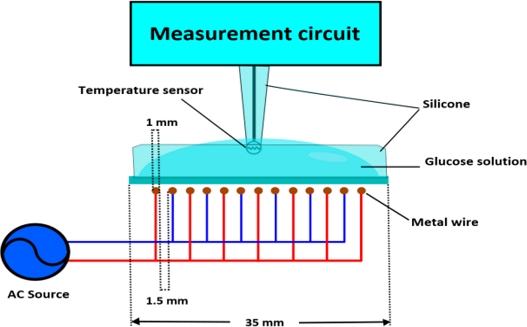
Based on this principle, a sensor system was designed, as shown in Fig. 2, to measure glucose concentration by applying an AC field and observing the resulting thermal response. The experiments were conducted using this structure.
To verify the operation of the proposed non-invasive glucose sensor, the sensor structure was configured as shown in Fig. 2. The region affected by the electric field consisted of a 35 mm-long polypropylene (PP) section into which the glucose solution was introduced, with silicone applied around it to ensure solution stability. Metal wires were arranged at 1.5 mm intervals at the bottom to apply an alternating voltage. The thermistor was positioned to avoid contact with the PP, and the wire section was sealed with silicone.
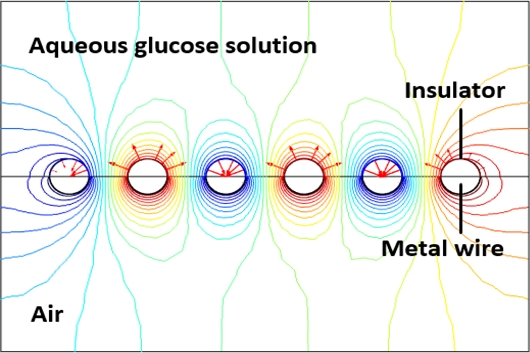
An electrostatic modeling process using the COMSOL Multiphysics electric field simulator was conducted to analyze the electric field distribution of the experimental structure, as shown in Fig. 3 [20]. In the computational model, the odd-numbered electrodes were connected, whereas the even-numbered electrodes were linked to form a structure in which the oscillating potential was applied. All electrode surfaces were surrounded by insulators, with the upper region defined as the glucose sample and the lower region as air. The dielectric constant of the glucose sample was set to 77, corresponding to a glucose concentration of 100 mg/dL [19]. The simulation results indicated that a strong electric field was concentrated between adjacent electrodes. This field distribution represents the conditions under which the permanent dipoles of glucose in solution can rotate and produce dielectric loss. Based on Eqs. (1) and (2), the thermal response observed in the experiments can be quantitatively explained by the simulated electric field distribution, thereby establishing a direct correlation between the simulation and experimental findings.
Glucose samples were prepared by mixing purified water with glucose to obtain concentrations of 50, 100, 200, 300, and 400 mg/dL; glucose solutions containing 0.9% NaCl were also prepared to simulate the salt level of human plasma.
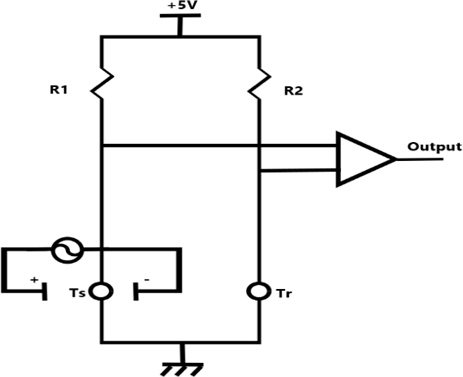
As shown in Fig. 4, the circuit used to apply an electric field to the proposed sensor and to measure temperature with thermistors was configured as a Wheatstone bridge. This circuit incorporated a thermistor (Tr) located in a region not influenced by the electric field and a thermistor (Ts) placed in a region exposed to the electric field. In other words,Tr measured the temperature of glucose, unaffected by the electric field, whereasTs measured the temperature of glucose under the influence of the field. These signals were processed through an op-amp circuit to cancel each other out, ensuring that the measured output was not affected by external temperature fluctuations. In this study, NTC (NTC-502F397F) thermistors and an OP177G operational amplifier were used.
The overall experimental setup for the proposed non-invasive glucose measurement system is illustrated in Fig. 5. The configuration comprised a function generator to produce alternating current (AC) signals, a direct current (DC) power supply to operate the measurement circuit, and two digital multimeters to monitor the thermistors. The AC source output was delivered through metal wires, whereas the measurement circuit was interfaced with the glucose sensor module. The sensor module contained a reference thermistor (Tr) and a sensing thermistor (Ts), with additional temperature sensors installed to monitor the ambient environment.
3. EXPERIMENTS AND RESULTS
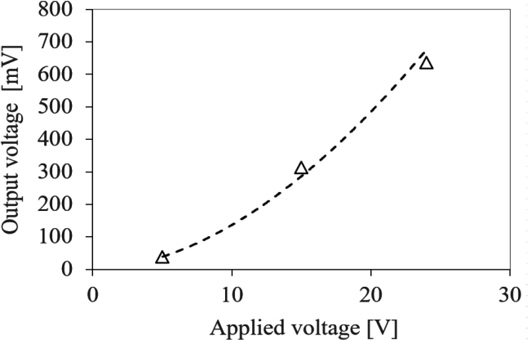
For a fixed glucose concentration of 100 mg/dL and a frequency of 10 kHz, the applied voltage was varied sequentially at 24, 15, and 5 V. Each experiment was repeated three times, and the average values are presented in Fig. 6.
This figure shows that dielectric loss approximately follows a relationship proportional to the square of the voltage, as described in Eq. (1). The maximum applied voltage was set to 24 V in accordance with the ILO regulations (AC 24 V or less), the Industrial Safety and Health Act (AC 30 V or less) [21,22], and the voltage tolerance of the Op-Amp. These results experimentally confirm that dielectric loss is proportional to the square of the applied voltage and demonstrate that stable measurements are achievable within the safe voltage range of 24 V or less.
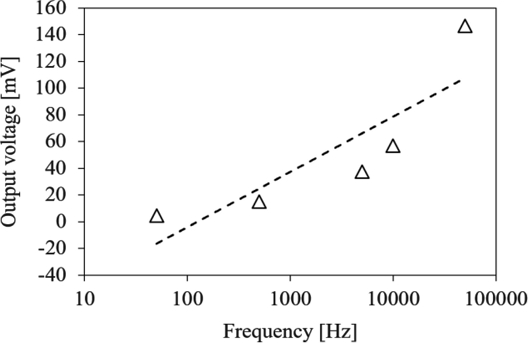
At a glucose solution concentration of 200 mg/dL, experiments were performed at frequencies of 0.05, 0.5, 5, 10, and 50 kHz to evaluate the effects of frequency. The results are shown in Fig. 7. As frequency increased, the rate of periodic changes in the electric field also increased, inducing polarization more frequently. This process generated greater dielectric loss, which in turn caused an increase in thermistor temperature. As described in Eq. (1), dielectric loss approximately correlates with frequency. The experimental findings confirmed that increasing frequency led to a higher rate of polarization events and greater dielectric loss, in good agreement with the theoretical equation.
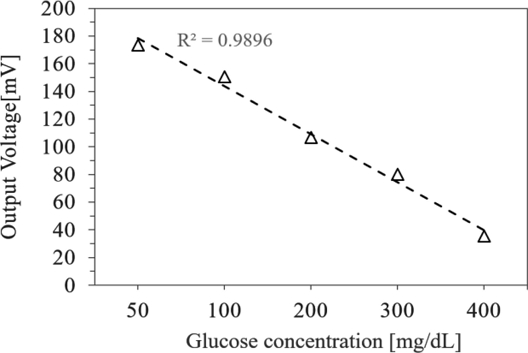
Experiments were conducted at 50 kHz for glucose solution concentrations of 50, 100, 200, 300, and 400 mg/dL, and the results are presented in Fig. 8. The output voltage decreased as the glucose concentration increased, a trend consistent with previous studies on dielectric loss [19]. Additionally, the output exhibited a linear decrease with increasing glucose concentration. The sensitivity was –34.64 mV/mg/dL, and the linearity, represented by the coefficient of determination, was 0.9896. These results confirm that output voltage decreases linearly with increasing glucose concentration, and the high coefficient of determination demonstrates excellent linearity.
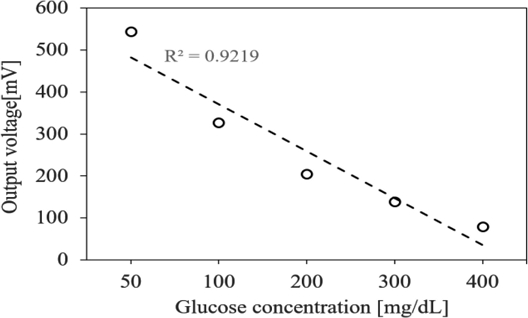
Glucose solutions containing 0.9% NaCl were used to evaluate the effects of glucose concentration under physiologically relevant conditions. Measurements were conducted under identical conditions for each concentration (50, 100, 200, 300, and 400 mg/dL), and the results are presented in Fig. 9.
Compared with Fig. 8, which shows results without NaCl, the output sensitivity of the glucose solution with NaCl increased to –111.78 mV/mg/dL, whereas the linearity decreased, with a coefficient of determination of 0.9219. The addition of NaCl enhanced electrical conductivity, steepened the output response slope relative to glucose concentration, and significantly enhanced sensitivity.
The observed phenomenon can be attributed to the increase in electrical conductivity (σ) resulting from the addition of NaCl. As electrical conductivity increases, the movement of ionic charges within the dielectric becomes more active, thereby increasing the dielectric loss rate in Eq. (1) and leading to greater overall dielectric loss.
These findings indicate that dielectric loss increases proportionally with electrical conductivity, and consequently, the sensitivity of the output response to changes in glucose concentration is enhanced.
Therefore, the results support the feasibility of employing the proposed system as a non-invasive blood glucose monitoring device.
4. CONCLUSIONS
This study experimentally verified the feasibility of a non-invasive blood glucose measurement technique based on the heat response induced by an electric field. The proposed sensor was designed to minimize the influence of external temperature by utilizing the difference between a reference thermistor and a measuring thermistor, thereby enabling the analysis of heat generation differences caused by dielectric loss with varying glucose concentrations.
The experimental results demonstrated that the output characteristics under different voltage and frequency conditions were consistent with the theoretical principles of dielectric loss. Furthermore, the decreasing trend in output with increasing glucose concentration showed a strong correlation, with a coefficient of determination of 0.9896. The highest linearity and sensitivity were achieved at 50 kHz and 24 V, respectively.
Additionally, in experiments using a glucose solution containing 0.9% NaCl, which approximates the NaCl concentration in blood, enhanced sensitivity was observed.
In conclusion, the proposed sensor system demonstrates the potential of electric field-based non-invasive blood glucose measurement technology. Future research directions include:
1. Conducting clinical trials and experiments with skin-mimicking tissue models to verify practical applicability.
2. Integrating multiple sensors to compensate for diverse environmental variables.
3. Performing further validation under real biological conditions, including noise analysis and comparison with existing methods for clinical application.
4. Simulating and optimizing electrode diameter and spacing to enable ultra-compact and portable sensor designs.
5. Developing methods to integrate electrodes and temperature sensors to further enhance the value and applicability of this research.
Such research may help overcome the limitations of conventional invasive blood glucose measurement methods and contribute to improving the quality of life of patients with DM.
Acknowledgments
This research received no external funding.
References
- World Health Organization, Fact sheets. https://www.who.int/news-room/fact-sheets, , 2025 (accessed 7 August 2025).
- International Diabetes Federation, Diabetes Facts & Figures. https://idf.org/about-diabetes/diabetes-facts-figures/, , 2025 (accessed 7 August 2025).
-
S.E. Park, S.-H. Ko, J.Y. Kim, K. Kim, J.H. Moon, N.H. Kim, et al., Diabetes Fact Sheets in Korea 2024, Diabetes Metab. J. 49 (2025) 24–33.
[https://doi.org/10.4093/dmj.2024.0818]

-
I. Baik, Projection of Diabetes Prevalence in Korean Adults for the Year 2030 Using Risk Factors Identified from National Data, Diabetes Metab. J. 43 (2019) 90–96.
[https://doi.org/10.4093/dmj.2018.0043]

-
A. Hina, W. Saadeh, A Noninvasive Glucose Monitoring SoC Based on Single Wavelength Photoplethysmography, IEEE Trans. Biomed. Circuits Syst. 14 (2020) 504–515.
[https://doi.org/10.1109/TBCAS.2020.2979514]

-
M. Shokrekhodaei, D.P. Cistola, R.C. Roberts, S. Quinones, Non-Invasive Glucose Monitoring Using Optical Sensor and Machine Learning Techniques for Diabetes Applications, IEEE Access 9 (2021) 73029–73045.
[https://doi.org/10.1109/ACCESS.2021.3079182]

-
Y.-T. Lee, S.-R. Lee, Development of the disposable glucose sensor using Cu/Ni/Au electrode, J. Sens. Sci. Technol. 15 (2006) 352–356.
[https://doi.org/10.5369/JSST.2006.15.5.352]

-
H. Choi, J. Naylon, S. Luzio, J. Beutler, J. Birchall, C. Martin, et al., Design and In Vitro Interference Test of Microwave Noninvasive Blood Glucose Monitoring Sensor, IEEE Trans. Microwave Theory Tech. 63 (2015) 3016–3025.
[https://doi.org/10.1109/TMTT.2015.2472019]

-
J.Y. Sim, C.-G. Ahn, E.-J. Jeong, B.K. Kim, In vivo Microscopic Photoacoustic Spectroscopy for Non-Invasive Glucose Monitoring Invulnerable to Skin Secretion Products, Sci. Rep. 8 (2018) 1059.
[https://doi.org/10.1038/s41598-018-19340-y]

-
W. Gao, S. Emaminejad, H.Y.Y. Nyein, S. Challa, K. Chen, A. Peck, et al., Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis, Nature 529 (2016) 509–514.
[https://doi.org/10.1038/nature16521]

-
J. Lee, B. Ryu, E. Yeom, W.G. Koh, Skin-adhesive patch based on hydrogel-incorporated nanofiber for non-invasive colorimetric sweat sensing, Sens. Actuators B Chem. 443 (2025) 138211.
[https://doi.org/10.1016/j.snb.2025.138211]

-
J. Heikenfeld, A. Jajack, J. Rogers, P. Gutruf, L. Tian, T. Pan, et al., Wearable sensors: modalities, challenges, and prospects, Lab Chip 18 (2018) 217–248.
[https://doi.org/10.1039/C7LC00914C]

-
S. Imani, A.J. Bandodkar, A.V. Mohan, R. Kumar, S. Yu, J. Wang, et al., A wearable chemical-electrophysiological hybrid biosensing system for real-time health and fitness monitoring, Nat. Commun. 7 (2016) 11650.
[https://doi.org/10.1038/ncomms11650]

-
A. Aloraynan, S. Rassel, C. Xu, D. Ban, A Single Wavelength Mid-Infrared Photoacoustic Spectroscopy for Noninvasive Glucose Detection Using Machine Learning, Biosensors 12 (2022) 166.
[https://doi.org/10.3390/bios12030166]

-
A. Omer, G. Shaker, S. Safavi-Naeini, H. Kokabi, G. Alquié, F. Deshours, et al., Low-cost portable microwave sensor for non-invasive monitoring of blood glucose level: novel design utilizing a four-cell CSRR hexagonal configuration, Sci. Rep. 10 (2020) 15200.
[https://doi.org/10.1038/s41598-020-72114-3]

-
S. Dutt, R. Thakur, A. Kandwal, P. Kumar, R. Jasrotia, N. Lakshmaiya, et al., Emerging EM wave sensors for non-invasive glucose monitoring: Review, techniques and developments, Sens. Actuators Rep. 9 (2025) 100282.
[https://doi.org/10.1016/j.snr.2025.100282]

-
L.B.A. Oliveira, G. Colherinhas, TD-DFT and GIAO-NMR spectroscopy studies for maltose and (α- and β)-glucose in water solution using S-MC/QM polarization methodology, J. Mol. Liq. 237 (2017) 295–303.
[https://doi.org/10.1016/j.molliq.2017.04.108]

- H.B. Kang, Electrical and Electronic Materials, Cheongmungak, Seoul, 1986, pp. 316–359.
-
N. Zeng, J. Li, T. Igbe, Y. Liu, C. Yan, Z. Nie, Investigation on Dielectric Properties of Glucose Aqueous Solutions at 500 KHz–5 MHz for Noninvasive Blood Glucose Monitoring, Proceedings of IEEE International Conference on e-Health Networking, Applications and Services (Healthcom), Ostrava, Czech Republic, 2018, pp. 1–5.
[https://doi.org/10.1109/HealthCom.2018.8531182]

- Altsoft Incorporated, COMSOL Multiphysics Guidebook I: Functional Descriptions for Beginners, V.50, Altsoft, Seoul, 2025.
- International Labour Organization, Safety and health in the use of machinery. https://www.ilo.org, , 2025 (accessed 7 August 2025).
- Korea Occupational Safety and Health Agency, Industrial safety guidelines. https://oshri.kosha.or.kr/kosha/index.do, , 2025 (accessed 7 August 2025).
-
W.V. Gonzales, A.T. Mobashsher, A. Abbosh, The progress of glucose monitoring—A review of invasive to minimally and non-invasive techniques, devices and sensors, Sensors 19 (2019) 800.
[https://doi.org/10.3390/s19040800]