Review of Ionic-Motion-based Chemimemristive Gas Sensors using Metal-Oxide Semiconductors
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Gas sensors are crucial for environmental monitoring, industrial safety, and healthcare, yet conventional metal-oxide chemiresistive devices require elevated temperatures (150–400°C) to activate surface oxygen redox reactions. Although approaches such as catalytic functionalization, UV illumination, and two-dimensional materials have been explored for room-temperature operation, they remain limited by incomplete recovery, poor selectivity, and instability. This review introduces ionic-motion-based chemiresistive sensors as a fundamentally different paradigm for room-temperature sensing. Three representative mechanisms are highlighted: proton conduction in hydrated layers, oxygen-vacancy migration in resistive-switching devices, and ion gating via electrolytes. These strategies not only enable efficient, low-power detection under ambient conditions but also open emerging opportunities, including memristive memory effects and neuromorphic olfactory functions. Ionic transport, therefore, provides a promising foundation for the development of next-generation, multifunctional gas sensors.
Keywords:
Gas sensors, Metal-oxide, Room temperature, Chemiresistive, Ionic motion1. INTRODUCTION
Gas sensors are indispensable for ensuring environmental safety, industrial process control, and human health. They provide real-time information on the presence of hazardous or trace gases, enabling applications such as air quality monitoring, explosion prevention, medical diagnostics, and the development of smart, interconnected devices within the Internet of Things (IoT) [1,2]. The increasing demand for portable, low-power, and highly selective gas sensors has driven decades of research into materials and mechanisms that can deliver reliable and reproducible performance [3,4].
Among the wide range of materials explored, metal oxide semiconductors have received the most attention owing to their low fabrication costs, simple processing, chemical stability, and compatibility with microelectronic platforms [5,6]. Conventional metal oxide-based chemiresistive sensors operate by modulating the carrier density through surface redox reactions with adsorbed gases [7]. These reactions are strongly activated at elevated temperatures, typically 150–400°C, which accelerates adsorption–desorption kinetics and ensures fast response, complete recovery, and stable performance over multiple cycles [8-10]. Despite these advantages, the requirement of high temperatures has significant drawbacks. Continuous heating leads to high power consumption, often exceeding hundreds of milliwatts, which limits portability [11]. Prolonged thermal stress can also cause grain growth or material degradation, thereby reducing device lifetime and reliability [12]. In addition, the integration with flexible or wearable electronics is hindered under such conditions. Therefore, the realization of room-temperature gas sensing remains a long-standing and critical goal in the field.
Various approaches, such as surface functionalization with catalytic metals [13], ultraviolet illumination [14,15], and the use of two-dimensional materials [16,17], have been actively explored for room-temperature sensing. Although these methods can lower activation barriers or enhance surface interactions, they rely on gas-induced surface reactions that modulate the electrical conductivity of the sensing layer. Consequently, their performances are limited by incomplete recovery, poor selectivity, and instability under practical operating conditions.
In this review, we summarize ionic-motion-based chemiresistive gas sensors that use metal oxide semiconductors as a fundamentally different paradigm for room-temperature operation. Instead of relying solely on thermally activated surface chemistry, these sensors exploit ionic processes such as proton conduction in adsorbed water networks, migration of oxygen vacancies within oxides, and ion gating through electrolyte dielectrics to modulate the sensor conductivity. By leveraging ionic transport, these devices not only enable efficient and low-power sensing at room temperature but also open new opportunities, including memristive memory behavior and neuromorphic functionalities for artificial olfactory systems. Therefore, ionic motion is a promising foundation for next-generation low-power multifunctional gas sensors.
2. MECHANISM OF CONVENTIONAL GAS SENSORS AND LIMITATIONS
Fig. 1 illustrates the fundamental working principles of metal-oxide-based chemiresistive gas sensors, highlighting the distinct behaviors of n- and p-type semiconductors when exposed to reducing gases such as hydrogen (H2) [18]. The sensing mechanism is primarily governed by the adsorption and reaction of gaseous species on the oxide surface, which modulates the charge carrier density, and consequently, the electrical conductivity of the sensing layer. These surface reactions generally require elevated operating temperatures above 150°C to proceed efficiently. At the microscopic level, grain boundaries play an important role as they form potential barriers that are highly sensitive to surface chemistry.
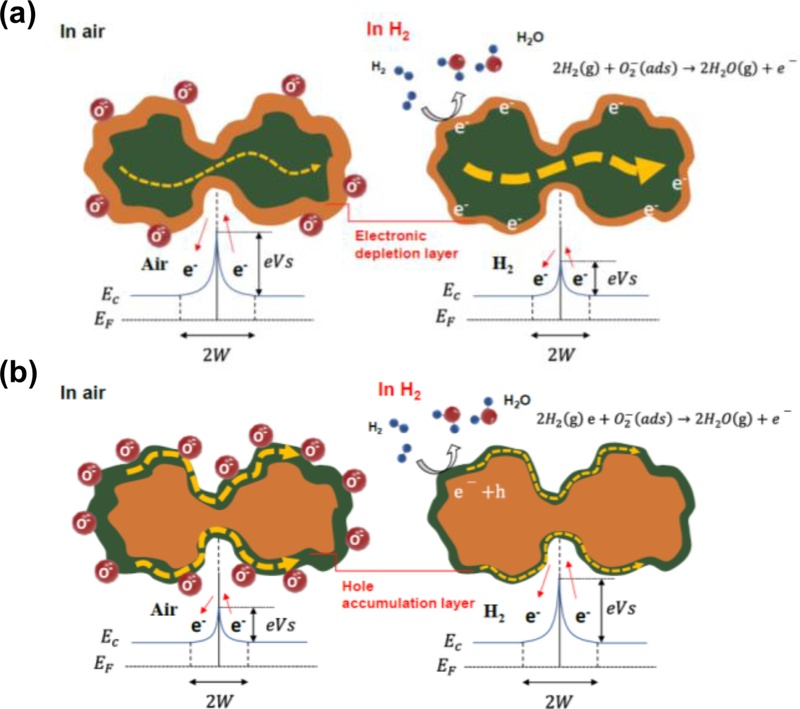
Conventional gas sensing mechanisms. Schematic illustration of (a) n-type and (b) p-type metal-oxide based gas sensing mechanisms. Adapted from Ref. [18]
In n-type semiconductors (Fig. 1(a)), such as SnO2, ZnO, and TiO2, oxygen molecules adsorbed on the surface under ambient air capture free electrons from the conduction band, forming ionized oxygen species. This process generated an electron-depletion region near the surface. At the grain boundaries, the overlapping depletion layers give rise to double Schottky barriers that dominate the overall sensor resistance. Upon exposure to hydrogen gas, the molecules react with the chemisorbed oxygen species, releasing the trapped electrons back into the conduction band. This reaction narrows the depletion region and lowers the barrier height at the grain boundaries, resulting in a significant decrease in resistance. The modulation of the depletion width (2 W) and surface potential (eVs), as illustrated in the band diagrams, is directly correlated with the observed change in conductance. This behavior also implies that the conduction pathway can shift from electron hopping or Poole–Frenkel conduction in the bulk to grain-boundary-limited conduction, emphasizing the critical role of intergranular junctions.
In contrast, p-type oxides (Fig. 1(b)) such as NiO and CuO exhibited opposite responses. In air, the adsorbed oxygen species extract electrons, leading to the formation of a hole accumulation layer (HAL) at the surface. This accumulation enhanced the majority carrier density, thereby lowering the resistance of the sensing material. Grain boundaries once again serve as active sites, where the modulation of potential barriers governs carrier transport. When exposed to hydrogen, the reducing gas reacts with the adsorbed oxygen species, decreasing their concentration on the surface. This process reduces the hole density near the surface and causes the accumulation layer to shrink. As the majority carrier concentration decreases, the resistance of the p-type oxides increases. In the band diagram, the modulation of 2 W and eVs shows a reverse tendency compared to the n-type case, resulting in an increase in the resistance rather than a decrease.
Overall, in both n- and p-type oxides, the key sensing principle lies in the dynamic modulation of the grain boundary potential barriers through surface redox reactions. The double Schottky barrier model provides an intuitive framework for explaining how carrier transport across grains is dictated by surface chemistry. However, the intrinsic requirements of high operating temperatures impose significant limitations, including high power consumption, reduced device stability, and poor compatibility with portable or wearable applications. Numerous attempts have been made to lower the operating temperature, for instance by incorporating catalytic noble metals, utilizing UV activation, or employing two-dimensional materials [13-17]. Nevertheless, these approaches rely on identical surface redox mechanisms, thereby inheriting their limitations. To overcome these constraints, recent studies have begun to explore gas-sensing mechanisms driven by ion motion, which represent a fundamentally different paradigm from conventional electron depletion or accumulation models.
3. IONIC-MOTION-BASED CHEMIRESISTIVE GAS SENSORS
3.1 Proton Conduction-based Gas Sensors
Humidity sensing was one of the earliest and most widely commercialized applications of metal-oxide-based chemiresistive devices, with electrical hygrometers already well established in consumer and industrial markets [19,20]. Unlike conventional metal oxide gas sensors, which depend on surface redox reactions with chemisorbed oxygen and therefore require elevated temperatures (typically >150°C), humidity sensors can operate effectively at room temperature. This remarkable difference arises from their fundamentally distinct mechanisms, which are not based on electron depletion or accumulation layers but rather on ionic motion within adsorbed water layers on the oxide surface.
When water molecules interact with the surfaces of semiconducting oxides such as WO3, SnO2, and ZnO, a sequential adsorption process occurs [21]. The first monolayer of water is strongly chemisorbed at the oxygen vacancy sites in the form of hydroxyl groups. As the humidity increases, additional water molecules form physisorbed layers bound by hydrogen bonds. Charge transport within these hydrated layers proceeds predominantly via proton conduction, rather than electronic conduction. Two major mechanisms have been identified: Grotthuss and vehicle mechanisms [22-24]. In the Grotthuss mechanism, protons are transferred along hydrogen-bonded networks through successive proton hopping between neighboring water molecules or hydroxyl groups. In the vehicle mechanism, hydronium ions (H3O+) serve as mobile carriers, physically transporting charges across the adsorbed water layers. As illustrated in Fig. 2(a), these processes enable effective proton transfer across the oxide surface, which results in significant changes in the electrical resistance as a function of the relative humidity [25]. Fig. 2(b) shows the typical response of a SnO2 quantum dot (QD) humidity sensor, where the normalized resistance decreases systematically with increasing relative humidity, even at room temperature. Importantly, this proton-conduction-based mechanism not only enables room-temperature operation but also provides a fast response, high sensitivity, and stability, which explains why humidity sensors have become one of the most mature and commercially successful families of chemiresistive sensors.
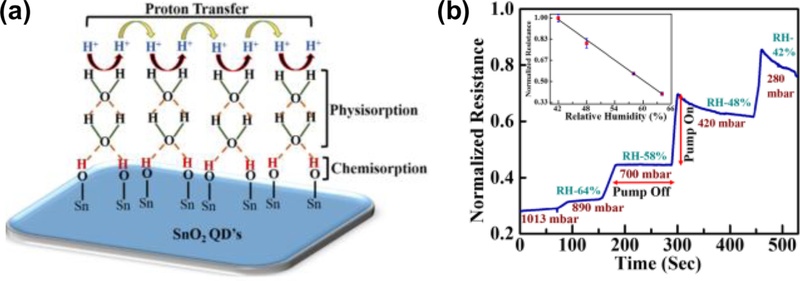
Relative humidity sensing using SnO2 quantum dots. (a) The mechanism of the relative humidity sensing in SnO2 QDs sensor. (b) The change of the electrical resistance with the variation of humidity at room temperature using as-prepared QDs. Adapted from Ref. [24]
The concept of utilizing ionic conduction for gas sensing was systematically demonstrated by Song et al., who reported an ionically activated chemiresistive mechanism using highly porous SnO2 nanorods [23]. As shown in Figs. 3(a) and 3(b), the sensors exhibited dramatic resistance changes to NO2 even at room temperature, with responses strongly enhanced under humid conditions compared to dry air. Importantly, the devices exhibited stable and linear sensing behaviors across different NO2 concentrations, proving that reliable and fast detection can be achieved without thermal activation. Figs. 3(c) and 3(d) schematically illustrate the underlying processes. Under dry conditions (Fig. 3(c)), NO2 adsorption was limited by the presence of pre-adsorbed oxygen species, which created a shielding effect that suppressed additional charge transfer. In contrast, under humid conditions (Fig. 3(d)), hydroxide groups formed on the SnO2 surface and established proton conduction pathways through hydrogen-bonded networks on the metal oxide surface. This ionic transport bypasses the restrictions of oxygen-mediated electron transfer, allowing for efficient interactions with NO2 molecules even at room temperature. As a result, the SnO2 nanorod-based sensors achieved ultrahigh sensitivity down to subparts-per-trillion levels with fast response and recovery, clearly demonstrating that ionic motion can replace high-temperature oxygen chemistry as the dominant sensing mechanism in metal oxide devices. However, the strong dependence on the ambient humidity level remains a limitation, highlighting the need to expand the operational window toward stable responses, regardless of the relative humidity.
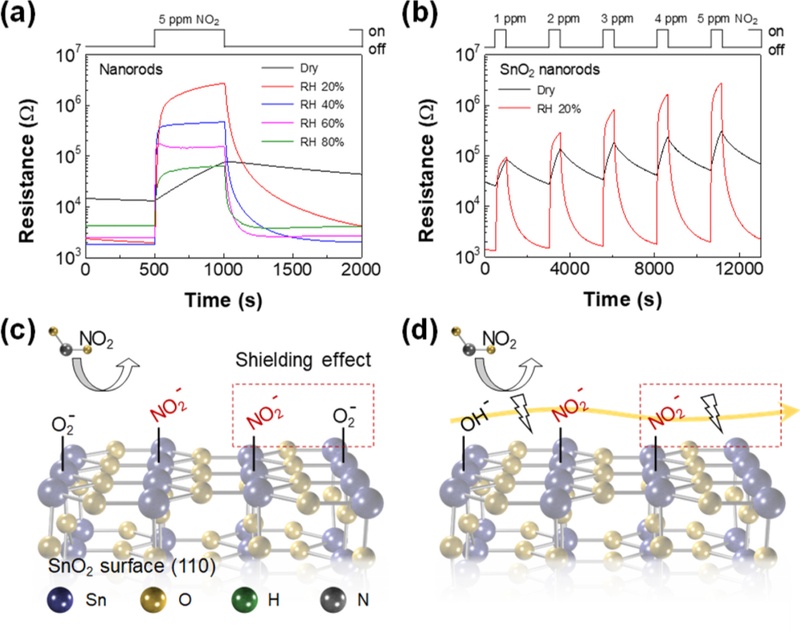
Humidity-mediated gas sensing characteristics of SnO2 nanorods. Response curves (a) the nanorods to 5 ppm NO2 as a function of RH at room temperature. (b) Response curves for 1–5 ppm NO2 in dry and RH 20%. Schematic illustrations of the sensor response to NO2 on the (c) oxygen adsorbed and (d) hydroxide functionalized SnO2 surface. Adapted from Ref. [23]
3.2 Oxygen Vacancy-related Gas Sensors
In the memory and neuromorphic fields, resistive switching (RS) devices are valued for their ability to reversibly switch between high- and low-resistance states through the formation and rupture of conductive filaments (CFs). These filaments, which are composed of oxygen vacancies, create localized conduction channels within the metal oxide layer. The application of an external voltage drives the migration of oxygen ions and vacancies, stabilizing or dissolving the CFs and thereby modulating the device resistance. This vacancy-mediated conduction supports both binary switching for nonvolatile memory and analog resistance tuning for artificial synapses [26,27].
Recently, it was reported that this filamentary conduction concept could be extended to room-temperature gas sensing [28-30]. Unlike conventional chemiresistive sensors that rely on thermally activated gas adsorption, RS-based devices exploit the redistribution of oxygen vacancy filaments, wherein their conductance can be modulated by oxidizing gases, enabling resistance changes without heating. As illustrated in Fig. 4(a), the continuous vacancy filaments enable electron transport in air, thereby producing a conductive state [31]. When oxidizing gases are introduced, they interact with the vacancy sites along the filaments. At low concentrations, this partial occupation perturbs the conduction path and lowers the current, whereas at higher concentrations, stronger adsorption further disrupts filament continuity, leading to significant current suppression. These findings suggest that the modulation of conductive filaments by adsorbed gases provides a viable pathway for chemical detection under ambient conditions.
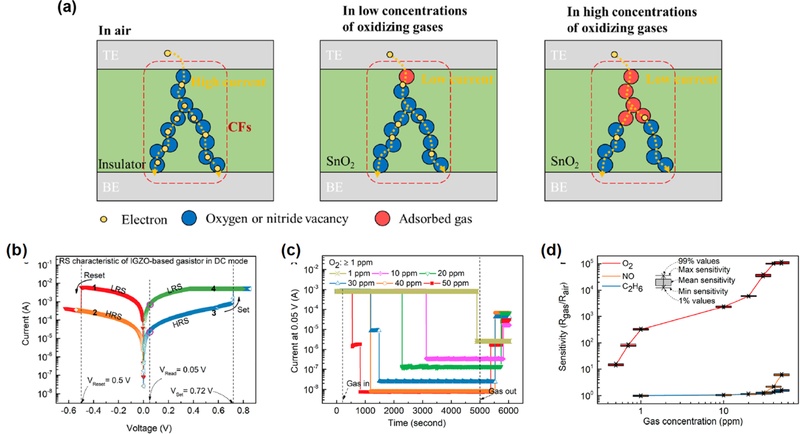
Oxygen vacancy-related gas sensors. (a) Schematic of gas sensing via oxygen-vacancy-based filaments in air and under low and high concentrations of oxidizing gases. Adapted from Ref. [31]. (b) Resistive switching (RS) characteristics of the IGZO-based gasistor in DC mode. (c) Transient response of the device to O2 above 1 ppm. (d) Sensitivity as a function of O2 concentration with error bars from five repeated tests. Adapted from Ref. [32]
As shown in Fig. 4(b), the resistive switching characteristics of IGZO-based devices exhibit clear set and reset transitions under DC bias [32]. When exposed to oxidizing gases at room temperature, the device showed an abrupt modulation of the current, as illustrated in Fig. 4(c). The transient response to increasing O2 concentrations from 1 to 50 ppm reveals a stepwise suppression of the current, indicating that filament conduction is progressively hindered by gas adsorption at the vacancy sites. Fig. 4(d) further highlights the gas selectivity. The response to O2 increased sharply with increasing concentrations, spanning several orders of magnitude, whereas negligible responses were observed for NO and C2H6 under identical conditions. This strong selectivity suggests that the oxidizing gases interact preferentially with vacancy-mediated filaments, whereas the reducing gases cannot effectively modulate the conduction pathways. Collectively, these results confirm that resistive switching devices can serve as dual-function platforms that operate as memory elements and highly selective room-temperature gas sensors through the interplay of oxygen vacancies, filamentary conduction, and gas adsorption.
The concept of employing RS devices as gas sensors was further advanced by Chun et al., who proposed an artificial olfactory system based on oxygen vacancy-mediated memristive dynamics [18]. Fig. 5(a) shows that the sensing response is prominent only in the high-resistance state (HRS), where residual cone-shaped filaments exist within the TiO2 nanorods. Upon exposure to H2, the surface reactions generate additional oxygen vacancies, which contribute to the growth and reconnection of the CFs, thereby reducing the resistance and markedly increasing the conductivity, as schematically illustrated in Fig. 5(b). Fig. 5(c) further demonstrates the transient response under repeated H pulses, where the current gradually increases with consecutive gas exposures and nearly recovers to its initial state upon removal. This gradual enhancement of the response was interpreted as an increase in the synaptic weight, representing the strengthening of the conductive pathways through ion-mediated modulation. In other words, repeated gas stimuli progressively modulate the charge-transport network, mimicking the learning and memory processes in biological synapses. This progressive potentiation implies a transition from short-term to long-term memory behavior, indicating that the device can retain a history of gas stimuli over extended periods [33]. Building on this functionality, an artificial olfactory synapse was successfully implemented in the device, demonstrating its potential to bridge gas sensing with neuromorphic functionality.
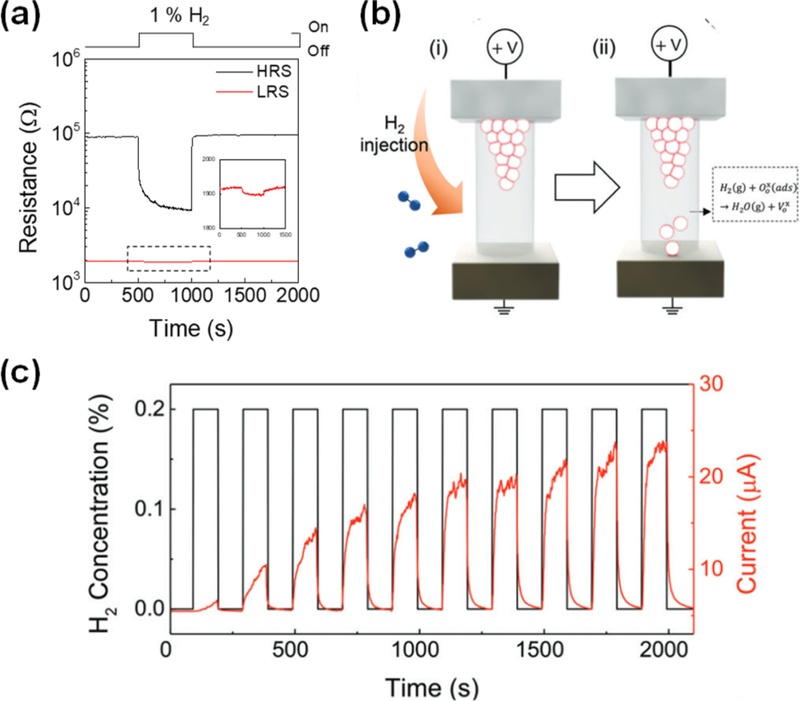
Chemi-memristive gas sensors enabling synaptic-like responses. (a) Sensing response of the chemi-memristive gas sensor at room temperature. (b) Schematic illustration of the H2 sensing mechanism. (c) Transient response under repeated H2 pulses, demonstrating gradual current increase analogous to synaptic weight potentiation. Adapted from Ref. [18]
These studies clearly demonstrate that oxygen vacancy filaments are not only central to resistive switching but can also be harnessed for selective and stable gas sensing at room temperature, opening the way toward multifunctional devices that unify memory, sensing, and neuromorphic operations.
3.3 Ion Gating-based Gas Sensors
Another approach to utilize ion motion for gas sensing is ion gating, where a three-terminal transistor structure is employed with an electrolyte as the gate dielectric [34,35]. When a gate bias is applied, mobile ions accumulate at the electrolyte/metal oxide (MOx) interface to form an electric double layer (EDL), which strongly modulates the carrier density of the channel. This configuration enables low-voltage operation, amplification of the sensing signals, and room-temperature performance, offering clear advantages over conventional chemiresistive sensors.
Inaba et al. demonstrated this concept using an ionic-liquid-gated graphene field-effect transsistor (IL-GFET) for NH3 sensing [36]. The device employed a 1-ethyl-3-methylimidazolium tetrafluoroborate ([EMIM][BF4]) ionic liquid that covered the graphene channel, serving simultaneously as a gas-absorbing medium and an electrolyte gate. To illustrate this basic principle, Figs. 6(a) and 6(b) compare a conventional back-gate graphene FET (GFET) sensor with an IL-gated GFET. In the IL-gated configuration, a nanometer-thick EDL is formed at the channel/electrolyte interface, providing an extremely high gate capacitance and efficient carrier modulation at voltages below 1 V. As shown in Fig. 6(c), the transfer characteristics shift progressively toward negative voltages as the NH3 concentration increases from 18 to 2400 ppm, indicating n-type doping due to charge transfer from the NH3 molecules dissolved in the ionic liquid. The voltage shift slope (−0.057 V per decade of concentration) follows a logarithmic dependence on NH3 level, yielding a detection limit of 130 ppb. Although the graphene channel is fully covered by the IL layer, the sensor exhibits a sensitivity comparable to that of a back-gate GFET while operating below 1 V with ultra-low power consumption (<100 nW). The response time of ~30 s for a 23-µm-thick IL layer confirms that gas diffusion through the electrolyte governs the transient behavior.
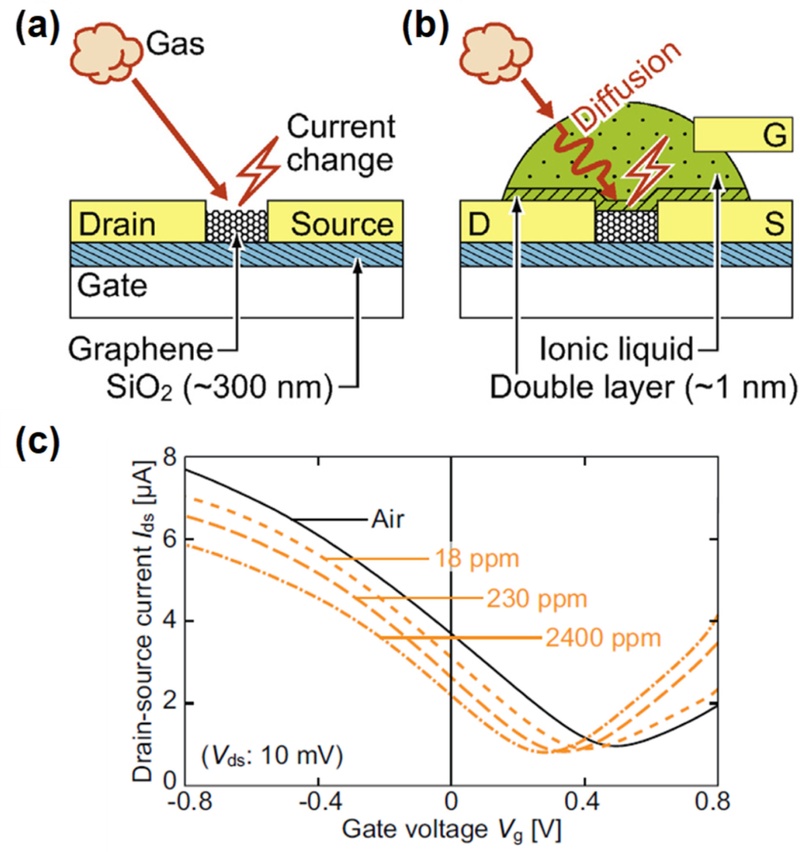
Ion gating-based graphene based gas sensors. Schematic illustration of (a) a conventional back-gate GFET gas sensor and (b) the proposed ionic-liquid-gated GFET sensor. (c) Transfer characteristics in air and under various NH3 concentrations (18, 230, 530, and 2400 ppm). Adapted from Ref [36].
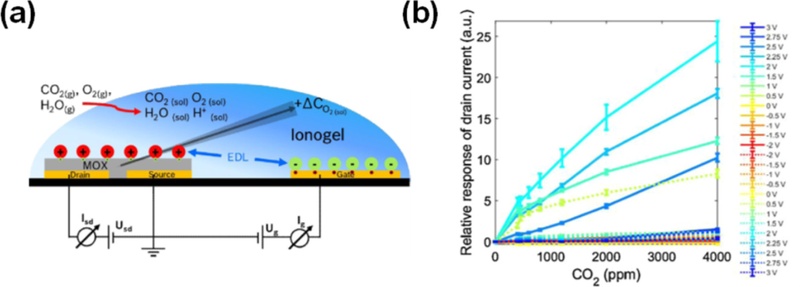
Although these results clearly demonstrate the advantages of ionic gating for low-voltage and high-gain gas sensing, the approach still faces several practical challenges. The electrolyte layer often suffers from limited stability, and its sensing response can be influenced by humidity and cross-sensitivity to other gases. To overcome these issues and extend the concept to metal-oxide semiconductors, Ersöz et al. developed an electrolyte-gated In2O3 transistor for CO2 sensing [37]. As illustrated in Fig. 7(a), the device employed a Nafion/[EMIM][BF4] ionogel that dissolved CO2 molecules and facilitated proton transport. Under an applied bias, an EDL is formed at the In2O3 channel interface, while the absorbed CO2 modifies the local oxygen chemistry, leading to modulation of the drain current. Fig. 7(b) demonstrates an almost linear increase in the relative drain current with CO2 concentration from 400 to 4000 ppm, with an optimal sensitivity of approximately 2 V. The sensor exhibited rapid response and recovery (~20 s), a detection limit below 200 ppm, and fully reversible operation under ambient conditions. These results highlight that the drain current is a reliable sensing metric, outperforming conventional FET parameters such as the threshold voltage.
4. CONCLUSIONS
Significant progress has been made in the development of room-temperature metal oxide gas sensors using nanostructured materials, two-dimensional systems, UV activation, and other approaches. However, these methods still rely on surface oxygen redox reactions and, therefore, face persistent challenges in terms of recovery, selectivity, and long-term stability. In this review, we highlight ionic-motion-based chemiresistive gas sensors as a new paradigm for room-temperature operation. Proton conduction, oxygen vacancy migration, and ion gating mechanisms provide alternative pathways to modulate the conductivity without thermal activation. These approaches not only enable efficient, low-power sensing but also introduce emerging opportunities such as memory effects and neuromorphic olfactory functions. Collectively, these advances have brought us closer to realizing ideal gas sensors for room-temperature applications.
Acknowledgments
This work was supported by the Academic Promotion System Tech University of Korea.
REFERENCES
-
H. Chai, Z. Zheng, K. Liu, J. Xu, K. Wu, Y. Luo, et al., Stability of metal oxide semiconductor gas sensors: A review, IEEE Sens. J. 22 (2022) 5470–5481.
[https://doi.org/10.1109/JSEN.2022.3148264]

-
R.A. Potyrailo, Multivariable sensors for ubiquitous monitoring of gases in the era of internet of things and industrial internet, Chem. Rev. 116 (2016) 11877–11923.
[https://doi.org/10.1021/acs.chemrev.6b00187]

-
T. Rault, A. Bouabdallah, Y. Challal, Energy efficiency in wireless sensor networks: A top-down survey, Comput. Netw. 67 (2014) 104–122.
[https://doi.org/10.1016/j.comnet.2014.03.027]

-
J. Yun, M. Cho, K. Lee, M. Kang, I. Park, A review of nanostructure-based gas sensors in a power consumption perspective, Sens. Actuators B Chem. 372 (2022) 132612.
[https://doi.org/10.1016/j.snb.2022.132612]

-
G.S. Kim, Y. Park, J. Shin, Y.G. Song, C.-Y. Kang, Metal Oxide Nanorods-Based Sensor Array for Selective Detection of Biomarker Gases, Sensors 21 (2021) 1922.
[https://doi.org/10.3390/s21051922]

-
M. Gardon, J.M. Guilemany, A review on fabrication, sensing mechanisms and performance of metal oxide gas sensors, J. Mater. Sci. Mater. Electron. 24 (2013) 1410–1421.
[https://doi.org/10.1007/s10854-012-0974-4]

-
N. Barsan, D. Koziej, U. Weimar, Metal oxide-based gas sensor research: How to?, Sens. Actuators B Chem. 121 (2007) 18–35.
[https://doi.org/10.1016/j.snb.2006.09.047]

-
Y.G. Song, Y.-S. Shim, S. Kim, S.D. Han, H.G. Moon, M.S. Noh, et al., Downsizing gas sensors based on semiconducting metal oxide: Effects of electrodes on gas sensing properties, Sens. Actuators B Chem. 248 (2017) 949–956.
[https://doi.org/10.1016/j.snb.2017.02.035]

-
Y.G. Song, J.Y. Park, J.M. Suh, Y.-S. Shim, S.Y. Yi, H.W. Jang, et al., Heterojunction Based on Rh-Decorated WO3 Nanorods for Morphological Change and Gas Sensor Application Using the Transition Effect, Chem. Mater. 31 (2019) 207–215.
[https://doi.org/10.1021/acs.chemmater.8b04181]

-
J.M. Suh, W. Sohn, Y.-S. Shim, J.-S. Choi, Y.G. Song, T.L. Kim, et al., p–p Heterojunction of Nickel Oxide-Decorated Cobalt Oxide Nanorods for Enhanced Sensitivity and Selectivity toward Volatile Organic Compounds, ACS Appl. Mater. Interfaces 10 (2018) 1050–1058.
[https://doi.org/10.1021/acsami.7b14545]

-
A. Baranov, D. Spirjakin, S. Akbari, A. Somov, Optimization of power consumption for gas sensor nodes: A survey, Sens. Actuators A Phys. 233 (2015) 279–289.
[https://doi.org/10.1016/j.sna.2015.07.016]

-
Z. Yuan, R. Li, F. Meng, J. Zhang, K. Zuo, E. Han, Approaches to enhancing gas sensing properties: A review, Sensors 19 (2019) 1495.
[https://doi.org/10.3390/s19071495]

-
Y. Tang, Y. Zhao, H. Liu, Room-temperature semiconductor gas sensors: challenges and opportunities, ACS Sens. 7 (2022) 3582–3597.
[https://doi.org/10.1021/acssensors.2c01142]

-
F. Meng, H. Zheng, Y. Sun, M. Li, J. Liu, UV‐activated room temperature single‐sheet ZnO gas sensor, Micro Nano Lett. 12 (2017) 813–817.
[https://doi.org/10.1049/mnl.2017.0148]

-
D. Cho, J.M. Suh, S.-H. Nam, S.Y. Park, M. Park, T.H. Lee, et al., Optically activated 3D thin‐shell TiO2 for super‐sensitive chemoresistive responses: toward visible light activation, Adv. Sci. 8 (2021) 2001883.
[https://doi.org/10.1002/advs.202001883]

-
I.-H. Baek, J.J. Pyeon, Y.G. Song, T.-M. Chung, H.-R. Kim, S.-H. Baek, et al., Synthesis of SnS Thin Films by Atomic Layer Deposition at Low Temperatures, Chem. Mater. 29 (2017) 8100–8110.
[https://doi.org/10.1021/acs.chemmater.7b01856]

-
J.J. Pyeon, I.-H. Baek, Y.G. Song, G.S. Kim, A.-J. Cho, G.-Y. Lee, et al., Highly sensitive flexible NO2 sensor composed of vertically aligned 2D SnS2 operating at room temperature, J. Mater. Chem. C 8 (2020) 11874–11881.
[https://doi.org/10.1039/D0TC02242J]

-
S.Y. Chun, Y.G. Song, J.E. Kim, J.U. Kwon, K. Soh, J.Y. Kwon, et al., An Artificial Olfactory System Based on a Chemi‐Memristive Device, Adv. Mater. 35 (2023) 2302219.
[https://doi.org/10.1002/adma.202302219]

-
D. Zhang, H. Chang, P. Li, R. Liu, Q. Xue, Fabrication and characterization of an ultrasensitive humidity sensor based on metal oxide/graphene hybrid nanocomposite, Sens. Actuators B Chem. 225 (2016) 233–240.
[https://doi.org/10.1016/j.snb.2015.11.024]

-
J.J. Steele, M.T. Taschuk, M.J. Brett, Nanostructured metal ox ide thin films for humidity sensors, IEEE Sens. J. 8 (2008) 1422–1429.
[https://doi.org/10.1109/JSEN.2008.920715]

-
T. Xie, A.F. Abdul Rahman, A. Abu Bakar, A. Arsad, Design and Optimization of Metal Oxide-Based Humidity Sensors: A Review on Mechanisms and Material Engineering, J. Clust. Sci. 36 (2025) 148.
[https://doi.org/10.1007/s10876-025-02869-0]

-
Q. Li, Q. Yin, Y.-S. Zheng, Z.-J. Sui, X.-G. Zhou, D. Chen, et al., Insights into hydrogen transport behavior on perovskite surfaces: transition from the grotthuss mechanism to the vehicle mechanism, Langmuir 35 (2019) 9962–9969.
[https://doi.org/10.1021/acs.langmuir.8b04138]

-
Y.G. Song, Y. Shim, J.M. Suh, M. Noh, G.S. Kim, K.S. Choi, et al., Ionic‐Activated Chemiresistive Gas Sensors for Room‐Temperature Operation, Small 15 (2019) 1902065.
[https://doi.org/10.1002/smll.201902065]

-
Y.G. Song, I. Jung, J. Shin, Y.-S. Shim, G.S. Kim, B.-K. Ju, et al., Ionic-activated semiconducting gas sensors operated by piezoelectric generators at room temperature, Sens. Actuators B Chem. 332 (2021) 129481.
[https://doi.org/10.1016/j.snb.2021.129481]

-
S. Pan, G. Gayathri, T.S. Reshma, G. Mangamma, A.K. Prasad, A. Das, A sensitive humidity sensor at low pressure with SnO2 QDs, Sens. Actuators Phys. 346 (2022) 113835.
[https://doi.org/10.1016/j.sna.2022.113835]

-
A. Chen, A review of emerging non-volatile memory (NVM) technologies and applications, Solid-State Electron. 125 (2016) 25–38.
[https://doi.org/10.1016/j.sse.2016.07.006]

-
E.W. Lim, R. Ismail, Conduction mechanism of valence change resistive switching memory: A survey, Electronics 4 (2015) 586–613.
[https://doi.org/10.3390/electronics4030586]

-
M. Vidiš, T. Plecenik, M. Moško, S. Tomašec, T. Roch, L. Satrapinskyy, et al., Gasistor: A memristor based gas-triggered switch and gas sensor with memory, Appl. Phys. Lett. 115 (2019) 093504.
[https://doi.org/10.1063/1.5099685]

-
P. Qiu, Y. Qin, Q. Xia, Ultrasensitive memristor-based gas sensor (gasistor) with gas-triggered switch and memory function for dilute NH3 detection, Sens. Actuators B Chem. 373 (2022) 132730.
[https://doi.org/10.1016/j.snb.2022.132730]

-
M. Ali, I. Ahmad, I. Geun, S.A. Hamza, U. Ijaz, Y. Jang, et al., A Comprehensive Review of Advanced Sensor Technologies for Fire Detection with a Focus on Gasistor-Based Sensors, Chemosensors 13 (2025) 230.
[https://doi.org/10.3390/chemosensors13070230]

-
M. Ali, D. Lee, I. Ahmad, H.-D. Kim, Recent progress in memristor-based gas sensors (Gasistor; gas sensor+ memristor): Device modeling, mechanisms, performance, and prospects, Sens. Actuators Rep. 9 (2025) 100269.
[https://doi.org/10.1016/j.snr.2024.100269]

-
D. Lee, J. Jung, K.H. Kim, D. Bae, M. Chae, S. Kim, et al., Highly sensitive oxygen sensing characteristics observed in IGZO based gasistor in a mixed gas ambient at room temperature, ACS Sens. 7 (2022) 2567–2576.
[https://doi.org/10.1021/acssensors.2c00484]

-
T. Chang, S.-H. Jo, W. Lu, Short-term memory to long-term memory transition in a nanoscale memristor, ACS Nano 5 (2011) 7669–7676.
[https://doi.org/10.1021/nn202983n]

-
S. Thiemann, S. Sachnov, S. Porscha, P. Wasserscheid, J. Zaumseil, Ionic liquids for electrolyte-gating of ZnO field-effect transistors, J. Phys. Chem. C 116 (2012) 13536–13544.
[https://doi.org/10.1021/jp3024233]

-
T. Wasilewski, J. Gębicki, W. Kamysz, Prospects of ionic liquids application in electronic and bioelectronic nose instruments, TrAC Trends Anal. Chem. 93 (2017) 23–36.
[https://doi.org/10.1016/j.trac.2017.05.010]

-
A. Inaba, K. Yoo, Y. Takei, K. Matsumoto, I. Shimoyama, Ammonia gas sensing using a graphene field–effect transistor gated by ionic liquid, Sens. Actuators B Chem. 195 (2014) 15–21.
[https://doi.org/10.1016/j.snb.2013.12.118]

-
B. Ersoez, K. Schmitt, J. Wöllenstein, Electrolyte-gated transistor for CO2 gas detection at room temperature, Sens. Actuators B Chem. 317 (2020) 128201.
[https://doi.org/10.1016/j.snb.2020.128201]


Ilho Myeong is an Assistant Professor in the Department of Electronics Engineering at Myongji University, South Korea. He received his Ph.D. in Electrical Engineering from Seoul National University in 2021. Until 2025, he worked at Samsung Electronics, where he was engaged in advanced semiconductor research and development. His research interests include Logic and Memory devices, nanostructures, and chemical sensors.

Young Geun Song is currently an Assistant Professor in the Department of Electrical Engineering at the Tech University of Korea, South Korea. He earned his Ph.D. in Electrical Engineering from Korea University in 2020. Prior to joining academia, he worked at Samsung Electronics until 2025, focusing on advanced semiconductor research and development. His research interests include semiconducting nanostructures, chemical sensors, and memristors.