
Smartphone-Integrated Paper-Based Electrochemical RT-LAMP Platform Targeting the N Gene for Rapid Point-of-Care Testing of SARS-CoV-2
 ; Sangmin Lee1
; Sangmin Lee1 ; Jihoon Kim1
; Jihoon Kim1 ; Wookjin Na1
; Wookjin Na1 ; Nomin Erdene Naranbaatar1 ; Youngung Seok1, +
; Nomin Erdene Naranbaatar1 ; Youngung Seok1, +
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
The COVID-19 pandemic has underscored the urgent need for rapid, accurate, and accessible diagnostic tools for use outside of centralized laboratories. While quantitative reverse transcription PCR (qRT-PCR) is considered the gold standard, it requires expensive instrumentation, trained personnel, and multi-hour protocols. To address these limitations, we developed a smartphone-integrated, paper-based electrochemical platform for the point-of-care detection of SARS-CoV-2. The system combines reverse transcription loop-mediated isothermal amplification (RT-LAMP) targeting the viral nucleocapsid (N) gene with differential pulse voltammetry (DPV) for electrochemical signal readout. Amplified products are deposited onto a glass microfiber filter (GMF) mounted over a commercial electrode (Metrohm 250AT), and signals are recorded using a portable potentiostat (PalmSens Sensit Smart) directly connected to a smartphone via the PStouch application. The platform successfully distinguished positive from negative samples, demonstrated concentration-dependent responses, and showed high specificity with no non-specific amplification. All assays were completed within one hour with minimal user intervention. In contrast to the centralized and instrument-intensive nature of qRT-PCR and antigen rapid tests, the proposed system offers a low-cost, portable, and quantitative solution. Its integration of amplification and detection in a compact format supports deployment in decentralized settings, enabling effective management of infectious diseases, response to outbreaks, and routine field diagnostics in both clinical and resource-limited environments.
Keywords:
Molecular diagnostics, Paper, Smartphone, Electrochemical detection, Point of care testing(POCT)1. INTRODUCTION
The global outbreak of coronavirus disease 2019 (COVID-19) has underscored the urgent need for rapid and sensitive molecular diagnostic technologies for early detection and response [1]. Although quantitative reverse transcription polymerase chain reaction (qRT-PCR) remains the gold standard due to its high analytical accuracy, its dependency on centralized laboratory infrastructure and thermal cycling instrumentation significantly limits its application in point-of-care testing (POCT) settings [2]. In response, integrated diagnostic platforms that combine paper-based electrochemical devices, smartphone-assisted readouts, and isothermal amplification techniques are being actively explored as viable alternatives [3,4].
Among various isothermal nucleic acid amplification methods, reverse transcription loop-mediated isothermal amplification (RT-LAMP) has emerged as a powerful tool for the rapid detection of viruses. RT-LAMP enables target amplification at a constant temperature of 65°C, eliminating the need for thermocyclers and making it highly compatible with resource-limited settings [5]. The method utilizes a set of primers to achieve high specificity and sensitivity, and the generation of pyrophosphate (PPi) and protons (H+) as amplification byproducts provides favorable conditions for downstream electrochemical signal transduction [6].
Differential pulse voltammetry (DPV) is a highly sensitive electrochemical detection technique that can measure subtle current changes. When integrated with paper-based electrode platforms, DPV allows for the quantitative detection of RT-LAMP amplification products in a label-free and rapid manner [7,8]. Paper substrates offer key advantages, including disposability, low cost, and scalability, while also enhancing device miniaturization and signal reproducibility [9].
In this study, we present a portable, smartphone-integrated diagnostic platform that combines RT-LAMP targeting the nucleocapsid (N) gene of SARS-CoV-2 with DPV-based electrochemical detection. This system is structurally distinct from previously reported paper-based recombinase polymerase amplification (RPA) assays [10] and dry-format LAMP devices [11], offering enhanced integration and analytical robustness. The proposed platform is designed for rapid, quantitative, and field-deployable molecular diagnostics, providing a promising solution not only for pandemic response but also for the broader detection of infectious diseases in decentralized environments.
2. EXPERIMENTAL
2.1. RT-LAMP Reaction Buffer and Conditions
In this study, the RT-LAMP reaction was performed using a customized buffer formulation rather than relying on commercially available kits. The composition and concentrations of reaction components were carefully optimized based on previous studies by Seok et al. [11] and Park et al. [12], which focused on paper-based and ambient-stable LAMP systems.
The final reaction volume was set at 20 μL, comprising 10 μL of 2× LAMP buffer. The 2× buffer solution included the following constituents: 2.8 mM of each dNTP, 16 mM MgSO4, 20 mM (NH4)2SO4, 20 mM KCl, 5 mM Tris-HCl (pH 8.8), 1.6 M betaine, and 0.2% Tween-20. These components were selected to ensure optimal enzyme activity, ionic balance, DNA strand separation stability, and the suppression of nonspecific interactions under isothermal amplification conditions [11,12].
Tris-HCl was used to maintain a stable reaction pH, while Mg2+ served as an essential cofactor for Bst polymerase activity. (NH4)2SO4 enhanced the specificity of primer-template binding, and betaine was included to destabilize secondary structures in GC-rich regions, thereby improving amplification efficiency. Tween-20 contributed to the homogeneity of the reaction mixture by reducing surface tension, while KCl improved enzyme stability and primer binding fidelity. The buffer composition was particularly optimized for liquid retention and reaction consistency within the paper-based format [11].
The enzymatic formulation consisted of Bst DNA polymerase (large fragment, with strand displacement activity) and M-MLV reverse transcriptase. Bst polymerase facilitated the isothermal DNA amplification, while M-MLV reverse transcriptase enabled cDNA synthesis from the RNA template. Both enzymes were configured to function concurrently under single reaction conditions, and amplification was conducted at 65°C for 60 minutes. Following the reaction, the amplified product was immediately used for electrochemical analysis via differential pulse voltammetry (DPV) without any additional purification steps, by being directly applied onto the electrode surface.
The buffer system was specifically optimized for compatibility with electrochemical detection, ensuring that factors such as conductivity, ionic strength, and pH stability did not interfere with the DPV signal resolution. In particular, the balance between Mg2+ and the resulting pyrophosphate concentrations was found to play a critical role in modulating electron transfer dynamics at the electrode–electrolyte interface, directly influencing signal quality and sensitivity.
The optimized formulation is consistent with those reported in earlier studies [11,12] on paper-based and portable LAMP systems, and it serves as a fundamental basis for the development of high-precision, field-deployable molecular diagnostic platforms.
2.2. Target Gene Selection and Primer Design
To develop a field-deployable electrochemical isothermal amplification platform for detecting SARS-CoV-2, the nucleocapsid (N) gene was selected as the target sequence. The N gene encodes a structural protein essential for viral replication and packaging, and is widely used in molecular diagnostics due to its high transcriptional abundance and strong sequence conservation across SARS-CoV-2 variants [14].
Primer design was based on the reference genome of SARS-CoV-2 (GenBank accession number NC_045512.2). A set of six LAMP primers—including F3, B3, FIP (Forward Inner Primer), BIP (Backward Inner Primer), LF (Loop Forward), and LB (Loop Backward)—was designed using PrimerExplorer V5 (Eiken Chemical Co., Ltd., Japan). Primer design was performed using the built-in automatic judgment function, which applies the default parameter settings optimized for LAMP design. The target sequence was uploaded in FASTA format, and primer candidates were generated without manual parameter modification. These candidates were further evaluated based on their thermodynamic properties, including Gibbs free energy (ΔG), GC content, terminal stability, and potential secondary structure formation.
To ensure high specificity, all designed primer sequences were subsequently analyzed using the NCBI Primer-BLAST tool with default parameters, screening against the human genome and other representative respiratory viral genomes to exclude potential non-specific homology and off-target amplification [3,12].
The primers were designed to amplify a highly conserved region of approximately 200–250 bp within the N gene. Critical ΔG thresholds for the F1c and B1c regions were maintained below −4 kcal/mol to facilitate efficient strand invasion and primer annealing. Loop primers (LF and LB) were included to accelerate amplification kinetics. All primers were synthesized using high-performance liquid chromatography (HPLC) purification by Integrated DNA Technologies (IDT, USA) [12].
The amplification efficiency and analytical specificity of the designed primer set were experimentally validated using purified SARS-CoV-2 RNA and RNA extracted from viral transport medium (VTM)-based clinical specimens. Compared to commercially available reference primer sets, the custom-designed primers exhibited comparable amplification performance under identical reaction conditions.
Final primer concentrations were optimized according to previously reported LAMP protocols, with FIP and BIP at 1.6 μM, LF and LB at 0.8 μM, and F3 and B3 at 0.2 μM [14]. These concentrations were consistently applied across all subsequent RT-LAMP experiments. The complete primer sequences are summarized in Table 1.
All primers were synthesized using high-performance liquid chromatography (HPLC) purification, and their working concentrations were optimized according to amplification efficiency.
LAMP primers targeting the SARS-CoV-2 N gene were designed using PrimerExplorer V5. Primer specificity was subsequently verified by analyzing each primer sequence using the NCBI Primer-BLAST tool with default parameters against the human genome and other respiratory viral genomes to exclude potential off-target amplification.
2.3. Configuration of the RT-LAMP Reaction Interface and Sample Sealing Structure
To construct the electrochemical RT-LAMP diagnostic platform, a commercial integrated electrode system (Metrohm 250AT) was employed in this study. This electrode features a built-in configuration that combines the working, counter, and reference electrodes, ensuring compatibility with external potentiostat devices for downstream electrochemical analysis. For nucleic acid amplification, a glass microfiber filter (GMF; Whatman, USA) was mounted directly on the working electrode surface. The RT-LAMP reaction mixture (20 μL) was then dispensed onto the GMF to initiate the amplification process. The porous structure of the GMF23 plays a crucial role in uniformly absorbing and retaining the reaction mixture, thereby minimizing sample loss and ensuring consistent contact between the solution and the electrode surface. This setup effectively prevents local dehydration or migration of reaction components during amplification, thereby promoting both physical stability and reaction uniformity. After applying the sample to the GMF, it was stabilized at room temperature and subjected to amplification using a built-in isothermal heating module within a sealed reaction chamber.
This configuration enables both nucleic acid amplification and electrochemical analysis to occur sequentially on the electrode surface, eliminating the need for external reaction tubes or separate vessels. This approach provides a foundation for device miniaturization and system simplification. The use of a GMF for sample anchoring not only streamlines device design but also serves as a core structural element for the future development of all-in-one nucleic acid testing devices. Overall, this section focuses on optimizing the physical interface for sample immobilization and stabilization, while simultaneously providing a robust foundation for integrating electrochemical readout in the downstream analysis phase.
2.4. Electrochemical Signal Acquisition via Differential Pulse Voltammetry (DPV)
To quantitatively evaluate the amplified products generated from the RT-LAMP reaction, this study employed differential pulse voltammetry (DPV) as the electrochemical detection method. DPV is widely recognized for its high sensitivity and excellent signal-to-noise ratio, and is capable of detecting minute current variations in the nanoampere range [2,4,5]. These attributes make it particularly suitable for applications that require precise molecular quantification, such as nucleic acid diagnostics. Electrochemical analysis was performed using the portable Sensit Smart potentiostat (PalmSens, Netherlands). Prior to measurement, the RT-LAMP reaction mixture was directly immobilized onto the surface of a glass microfiber filter (GMF) attached to the working electrode. This setup enabled the monitoring of electron transfer dynamics at the electrode–electrolyte interface in response to the presence of amplification byproducts. The DPV measurement parameters were configured as follows: initial potential (E₀) at –1.0 V, terminal potential (E_end) at +1.0 V, step potential at 0.01 V, pulse amplitude at 0.2 V, pulse width at 0.02 s, and a scan rate fixed at 0.1 V/s (see Fig. 2).
The principle of DPV detection relies on electrochemical perturbations caused by biochemical species such as double-stranded DNA, pyrophosphate (PPi), and protons (H⁺) generated during the LAMP reaction. These molecules modulate the local charge distribution and electron transfer resistance at the electrode surface. Consequently, distinct peak currents appear in defined voltage ranges, depending on the presence and concentration of the target, enabling effective discrimination between positive and negative samples [4,6,9].
One notable advantage of this platform is its low background noise and minimal interference from nonspecific amplification or ionic contaminants. This performance is primarily attributed to the uniform liquid-holding structure of the GMF and the high reproducibility of the commercial screen-printed electrodes employed [3,7]. The resulting DPV signals were visualized in real time through the PStouch mobile application, providing immediate graphical feedback on a smartphone display.
Additionally, all data were stored in CSV format, facilitating subsequent numerical analysis and statistical validation. This integrated digital pipeline represents a significant advancement over traditional colorimetric LAMP systems by enabling objective, quantitative, and reproducible signal interpretation.
Overall, the implementation of DPV in this platform reinforces its suitability for point-of-care testing (POCT), offering a rapid, portable, and user-friendly diagnostic solution that maintains analytical precision in resource-limited or decentralized environments [1,9].
2.5. Smartphone-Integrated Data Acquisition System
To fulfill the essential requirements of point-of-care (POC) molecular diagnostics, namely real-time analysis and portability, this study implemented a smartphone-integrated electrochemical detection platform. Differential pulse voltammetry (DPV) measurements were conducted using the Sensit Smart portable potentiostat, developed by PalmSens. This compact device directly interfaces with an Android-based smartphone, specifically a Samsung Galaxy S9 (Android OS), enabling seamless control, real-time visualization, and data storage of electrochemical measurements through the proprietary PStouch application.
The PStouch application provides real-time rendering of potential-current (I–V) curves on a user-friendly graphical user interface (GUI), enabling the immediate interpretation of results without the need for a laboratory-grade computer. This functionality allows users to intuitively monitor diagnostic outputs in situ, significantly reducing operational complexity in field settings [6,9,13]. All acquired electrochemical data are automatically saved in CSV format, which can be exported for quantitative post-analysis using standard tools such as Microsoft Excel or Python-based computational environments. The smartphone-based analysis framework not only enhances accessibility to raw data but also facilitates integration with cloud-based transmission systems for centralized monitoring and data sharing [5,8,13].
Moreover, maintaining a consistent and high-precision analytical environment is a critical requirement in electrochemical POCT platforms. The Sensit Smart device meets these demands by offering a broad range of potential control and high-sensitivity current detection capabilities. It is capable of reliably resolving electrical signals in the nanoampere (nA) range, which is particularly advantageous for detecting early-stage amplification products that produce minimal electrochemical perturbations. This contributes to the improved quantitative accuracy and reproducibility of the system, particularly under low template concentrations [4,7].
The incorporation of a smartphone as the primary readout and data management interface provides a compelling advancement in user-centered diagnostics. By simplifying traditionally complex laboratory workflows while maintaining analytical rigor, this approach lays a robust foundation for next-generation POCT systems. It enables end-users, including non-specialists, to conduct high-precision molecular diagnostics in decentralized settings, thus broadening the scope of infectious disease surveillance and early intervention strategies [1,6,13].
All electrochemical measurements and data visualization were performed using a Samsung Galaxy S9 smartphone (Android OS).
2.6. Schematic Design of the Portable Diagnostic Platform
The device proposed in this study is a compact, field-deployable electrochemical diagnostic platform optimized for the detection of SARS-CoV-2. It integrates isothermal amplification via RT-LAMP at a controlled temperature of 65°C and differential pulse voltammetry (DPV)-based electrochemical detection into a single portable structure. The complete system consists of a smartphone, the Sensit Smart potentiostat (PalmSens), a commercial three-electrode configuration (Metrohm 250AT), a Whatman glass microfiber filter (GMF), a sample injection port, a 12V positive temperature coefficient (PTC) heater, a power control module (comprising a 12V battery and switch), and an outer casing (Fig. 1(b)).
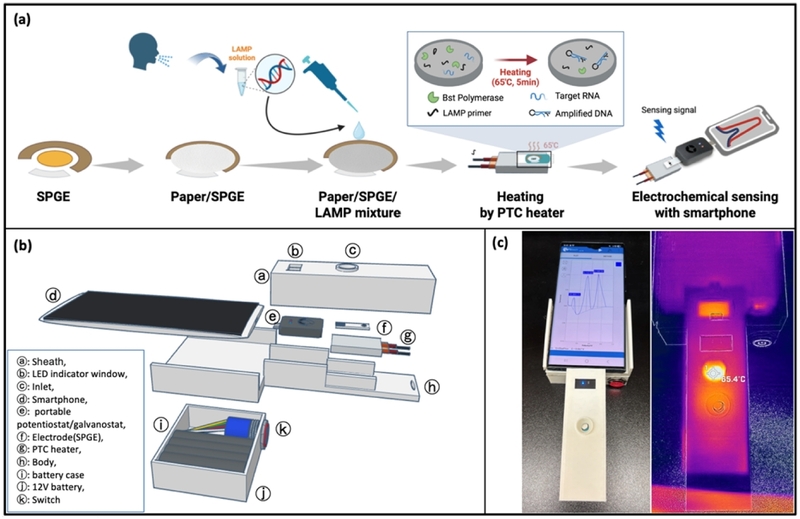
Conceptual overview and device configuration of the RT-LAMP/DPV platform. (a) Schematic illustration of the proposed workflow: SARS-CoV-2 N gene amplification via RT-LAMP, deposition of products on a GMF-assisted SPGE (screen-printed gold electrode), and electrochemical readout using DPV integrated with a smartphone. (b) Exploded view of the integrated device. The schematic shows the structural layout and integration of the amplification, electrochemical detection, heating, and power modules within the portable housing. Individual components are labeled directly in the figure. (c) Photograph and infrared thermal image of the assembled device during operation. The thermal profile confirms that the integrated PTC heater maintains a stable temperature of ~65°C, which is suitable for efficient RT-LAMP amplification.
The exterior housing of the device was designed using CAD software (Fusion 360, Autodesk) and fabricated using a fused filament fabrication (FFF)-based 3D printer. The final prototype measures 190 mm × 85 mm × 50 mm (L × W × H), with these dimensions chosen to ensure operational convenience and portability in field applications. The internal circuitry was designed to operate under low-power conditions, and the heater was optimized to maintain the target temperature for RT-LAMP reactions without requiring external thermal regulation units [6,9,11].
To validate the thermal control performance, the heating characteristics of the integrated 12V PTC heater were evaluated using a thermal imaging camera (FLIR ONE Pro). The temperature distribution was analyzed with a particular focus on the region where the GMF was positioned. As visualized in Fig. 1(c), the temperature at the electrode-contact interface was stably maintained within the optimal range for LAMP reactions (65°C ± 1°C). This result confirms that the self-regulating nature of the PTC heater and the thermally insulating design of the casing jointly contribute to stable thermal maintenance, eliminating the need for precision thermostatic control [7,12].
The system architecture presented here provides a foundational blueprint for the development of future all-in-one diagnostic platforms that integrate nucleic acid amplification and electrochemical detection into a single chip. Furthermore, the modular nature of the power system and the customizability afforded by 3D printing enable the expansion of this platform for detecting various pathogens by altering only specific structural or biochemical components [1,5,6]. Indeed, similar smartphone-integrated diagnostic devices have recently been deployed in various LAMP-based applications and are gaining attention as core technologies for next-generation point-of-care testing (POCT) systems due to their digital readout capabilities, low power requirements, and highly miniaturized form factors [6,8,13].
2.7 Clinical respiratory virus samples and nucleic acid extraction
Clinical respiratory specimens were obtained from Chonnam National University Hospital (CNUH) with approval from the Institutional Review Board (IRB No. CNUH-2025-402). For this study, positive samples of major respiratory pathogens were selectively collected, including SARS-CoV-2 (n = 13), influenza A (n = 6), adenovirus (n = 4), and respiratory syncytial virus (RSV A, n = 6; RSV B, n = 39). These pathogens were chosen because of their high prevalence in clinical respiratory infections and the frequent overlap in their symptoms.
All samples were confirmed positive by routine diagnostic assays at CNUH prior to use. Following collection, specimens were anonymized, stored at –80°C, and processed under standard biosafety protocols. Viral nucleic acids were isolated using a commercial extraction kit (QIAamp Viral RNA Mini Kit, Qiagen, Germany) according to the manufacturer’s protocol. The purified RNA was subsequently used as a template in RT-LAMP reactions to evaluate assay performance.
For the electrochemical specificity tests, a broader panel of clinically relevant viruses was also included to examine potential cross-reactivity. This panel consisted of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), Influenza A Virus (IAV), Respiratory Syncytial Virus A (RSV-A), Respiratory Syncytial Virus B (RSV-B), Human Immunodeficiency Virus (HIV), and Hepatitis B Virus (HBV). The inclusion of these viruses allowed direct assessment of primer specificity against both common respiratory pathogens and unrelated viral genomes, thereby reinforcing the robustness of the assay design.
3. RESULTS AND DISCUSSIONS
This study aimed to evaluate the feasibility of a field-deployable point-of-care testing (POCT) platform for the rapid detection of SARS-CoV-2 infection. The proposed system is based on reverse transcription loop-mediated isothermal amplification (RT-LAMP) and incorporates an electrochemical detection strategy that enables the quantification of nucleic acid amplification products in real time, eliminating the need for additional post-amplification handling steps or signal transduction delays. The complete platform consists of three sequential modules: (1) RNA-based RT-LAMP amplification, (2) electrochemical detection of the amplified product on a paper-based electrode interface, and (3) smartphone-integrated, real-time analysis and visualization [1,5,11].
In this section, we present both quantitative and qualitative evaluations of the functional performance of each core component of the system. Specifically, the thermal stability of the RT-LAMP reaction, electrochemical sensitivity, signal reproducibility, and target specificity were experimentally validated. Furthermore, the platform was benchmarked against conventional detection systems, such as colorimetric and fluorescence-based methods, to highlight its advantages in terms of quantification accuracy, mobile readout capability, portability, and low power consumption [2,7,9].
This approach provides a novel POCT diagnostic strategy that addresses the limitations of traditional qRT-PCR systems, particularly their dependence on centralized laboratory infrastructure and extended turnaround times. Notably, the proposed platform integrates nucleic acid amplification and electrochemical signal transduction into a single, compact unit, without the need for microfluidic channels or optical detection modules. This design feature offers significant advantages for field-based applications, where simplicity, cost efficiency, and rapid diagnostics are paramount [5,13].
3.1. Thermal Stability Evaluation
The principle of loop-mediated isothermal amplification (LAMP) relies on efficient nucleic acid amplification under constant temperature conditions. Typically, LAMP reactions perform optimally at a temperature range of 60–65°C. In particular, the reverse transcription LAMP (RT-LAMP) system used in this study demonstrated its highest amplification efficiency at 65°C. Given this characteristic, precise and stable temperature control within the platform is considered a critical factor for successful amplification.
While laboratory-based thermocyclers and isothermal incubators enable precise temperature regulation, their size, cost, and energy requirements render them impractical for point-of-care testing (POCT) settings. To address this limitation, we implemented a compact and energy-efficient thermal control unit utilizing a 12V positive temperature coefficient (PTC) heater. This component inherently self-regulates its temperature: as it reaches the target threshold, its electrical resistance increases sharply, thereby limiting further heat generation without requiring complex external feedback circuitry.
To evaluate the heating performance and thermal precision of the integrated heater, we used an infrared thermal imaging camera (FLIR) to monitor the temperature distribution within the diagnostic platform. The thermal images captured during operation visualize both the spatial uniformity and temporal stability of heat delivery, particularly in the reaction zones located on the electrode surface and the attached glass microfiber filter (GMF). As shown in Fig. 1(c), the temperature around the heating region rapidly stabilized at approximately 65.1°C. Notably, the critical GMF zone where the RT-LAMP reaction occurs reached the target temperature within minutes of activation and maintained it continuously for over an hour.
These findings underscore the effectiveness of a resistance-based heating mechanism in achieving stable thermal control with minimal circuit complexity. The system satisfies the thermodynamic conditions required for successful isothermal amplification even under resource-limited field conditions. Furthermore, the low power consumption and 12V operating voltage enable standalone operation using portable battery modules, thus enhancing the platform’s viability for POCT applications.
In summary, the thermographic data presented in Fig. 1(c) provide visual evidence that the platform delivers optimal thermal conditions for RT-LAMP reactions. This confirms that precise and sustained temperature regulation can be achieved without the use of conventional temperature control instruments, supporting the realization of a fully portable molecular diagnostic system.
3.2. Electrochemical Detection Mechanism Based on Isothermal Amplification
Electrochemical nucleic acid sensing offers distinct advantages for point-of-care diagnostics by enabling the sensitive detection of amplification events through interface charge redistribution, pH variation, and ionic precipitation associated with the formation of double-stranded DNA (dsDNA) [4,10]. In the platform proposed in this study, reverse transcription loop-mediated isothermal amplification (RT-LAMP) is directly performed on the electrode surface to maximize the electrochemical response generated during the amplification process. This on-electrode reaction design eliminates the need for sample transfer between amplification and detection, thereby reducing reagent consumption, preventing cross-contamination, and enhancing reaction reproducibility.
As schematically illustrated in Fig. 1(a), the platform utilizes a set of LAMP primers targeting the nucleocapsid (N) gene of SARS-CoV-2, Bst polymerase, and the 5³ RNA template to drive isothermal amplification directly on the surface of the working electrode at 65°C. During amplification, dsDNA is produced alongside biochemical byproducts such as protons (H⁺) and inorganic pyrophosphate (PPi), which collectively alter the electrochemical environment at the electrode–electrolyte interface [10,14].
Specifically, the accumulation of H⁺ ions leads to localized acidification, which shifts the interfacial potential and results in modulations of the electrical double-layer capacitance and Faradaic current. These shifts manifest as detectable current peaks in differential pulse voltammetry (DPV) analysis [4,7]. In parallel, PPi reacts with divalent metal ions to form precipitates that either hinder or promote charge transfer at the electrode surface. This dual mechanism generates quantitative differences in the electrochemical signals between positive and negative samples, enabling not only binary target detection but also the semi-quantitative evaluation of amplification magnitude.
To support this on-site electrochemical readout architecture, the platform integrates the electrode, glass microfiber filter (GMF), and RT-LAMP reaction solution into a single-layered system. This unified configuration ensures consistent sample distribution and stable reaction conditions, eliminating the need for physical movement. The paper-based GMF substrate contributes to signal uniformity and sensitivity by leveraging its capillary-driven liquid retention and dispersion capabilities [14].
Collectively, this design overcomes the limitations of conventional LAMP-based diagnostic workflows, often reliant on multiple reaction chambers and auxiliary instruments, by enabling “One-Pot, One-Site” amplification and detection. The use of inexpensive metal electrodes and paper materials further improves the cost-effectiveness, portability, and field-deployability of the platform. As a result, this system offers a viable technological foundation for highly integrated POCT applications in both clinical and low-resource settings.
3.3. Differential Electrochemical Signal Analysis Between Positive and Negative Controls
A key objective of this study was to experimentally verify whether the proposed platform could reliably distinguish the presence or absence of SARS-CoV-2 RNA based on electrochemical signal variation. To this end, we compared the electrochemical profiles before and after the RT-LAMP amplification process. Two test conditions were established: a negative control (NC) using RNA-free buffer and a positive control (PC) containing a high concentration of SARS-CoV-2 RNA (10⁶ copies/μL). Both groups underwent identical RT-LAMP reaction conditions, after which the reaction mixtures were applied directly to the electrode surface and analyzed using differential pulse voltammetry (DPV).
The DPV results revealed that the NC exhibited no significant peak current throughout the entire potential scan range (–1.0 V to +1.0 V), suggesting the absence of nonspecific amplification or unintended electrochemical interference. In contrast, the PC produced a prominent oxidation peak within the potential window of +0.2 V to +0.4 V, accompanied by a marked increase in current in the tens of nanoamperes. This signal enhancement is attributed to the generation of double-stranded DNA (dsDNA), protons (H⁺), and inorganic pyrophosphate (PPi) during the RT-LAMP reaction, which collectively alter the electrochemical properties of the electrode–electrolyte interface [4,7,10].
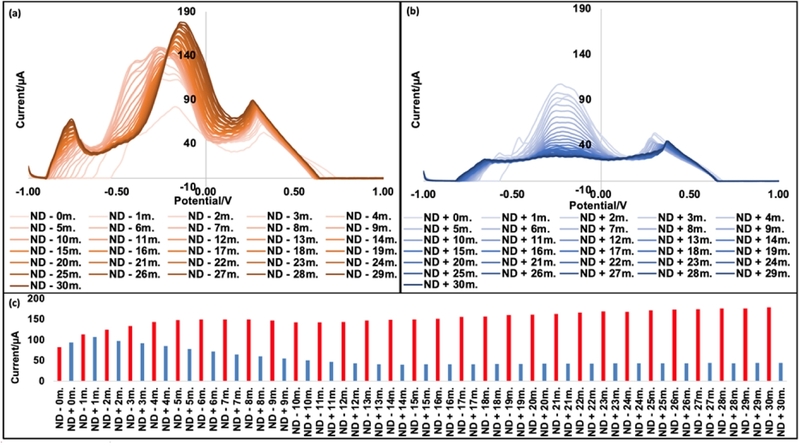
Real-time electrochemical discrimination between RT-LAMP negative and positive samples using DPV. (a) DPV profiles obtained from RT-LAMP negative control (NC) samples measured at 1 min intervals during the amplification process. The peak current remains within a narrow range over time. (b) DPV profiles obtained from RT-LAMP positive control (PC) samples measured at the same 1 min intervals under identical experimental conditions. Distinct peak currents are consistently observed within a comparable potential range. (c) Maximum DPV peak current values extracted from panels (a) and (b) as a function of reaction time (0–30 min).
In particular, the localized accumulation of H⁺ ions leads to a decrease in pH near the electrode surface, inducing a shift in the interfacial potential and enhancing Faradaic current through changes in charge distribution and potential gradients. Simultaneously, PPi interacts with metal ions or modulates interfacial reactions, either facilitating or impeding charge transfer depending on the presence of the target RNA, thereby contributing to the formation of differential current peaks [10].
These findings clearly demonstrate that the platform can distinguish positive and negative samples by quantifying the current variation induced by target amplification. In contrast to traditional colorimetric methods, which often lack sensitivity at low concentrations and suffer from subjective interpretation, this system provides numerical outputs through DPV, enabling objective and digitalized quantification. Such digital signal readout is advantageous for both clinical diagnosis and field-deployable applications, particularly in remote or resource-limited settings [4,9].
Furthermore, the reproducibility of the electrochemical signals was evaluated through repeated measurements (n ≥ 3), which consistently yielded peak currents within a narrow potential range (mean +0.32 V ± 0.03 V). The relative standard deviation (RSD) for peak current amplitude remained below 5% across all PC conditions. These results affirm the platform’s excellent signal stability and high-fidelity readout performance, underscoring its potential as a robust and reliable diagnostic system for molecular point-of-care applications [9].
Notably, a clear separation between positive and negative samples is already evident within the first 5 min of measurement, with the peak current of positive samples exceeding that of negative samples by approximately twofold. This result demonstrates that the proposed platform enables real-time electrochemical monitoring and early discrimination of target amplification.
3.4. Quantitative Validation via Concentration-Dependent Detection Experiments
This study further aimed to evaluate the relative quantification capability of the proposed electrochemical RT-LAMP platform, determining whether the system could not only distinguish between positive and negative results but also differentiate varying viral loads among infected individuals. To this end, RT-LAMP reactions were first performed under fixed conditions (65°C, 60 minutes) to generate a sufficient amount of amplified product from a positive SARS-CoV-2 RNA sample. The resulting amplicon solution was then serially diluted to obtain samples with varying concentrations, representing a gradient of target loads.
Each diluted sample was applied to the same electrode-filter-reaction configuration, followed by electrochemical analysis using differential pulse voltammetry (DPV). The DPV responses demonstrated a clear trend: as the dilution increased (i.e., the target concentration decreased), the peak current values consistently declined. Conversely, higher concentrations yielded significantly stronger peak currents, indicating a greater perturbation in electron transfer kinetics at the electrode–electrolyte interface. These changes are attributed to the accumulation of double-stranded DNA (dsDNA), hydrogen ions (H+), and inorganic pyrophosphate (PPi), which are generated as by-products of the LAMP reaction and directly influence the interfacial electrochemical behavior [4,7].
Repeated measurements (n = 3) at each concentration level showed that the relative standard deviation (RSD) of peak current values remained within 5%, confirming the platform's high signal reproducibility and robust quantitative reliability [9]. This quantitative approach addresses significant limitations of conventional colorimetric LAMP readouts, such as interpretational subjectivity and a lack of quantification, ultimately improving diagnostic precision.
In addition, the platform's integration with the smartphone-compatible PStouch system enabled real-time data acquisition, the automatic extraction of peak current values, result storage, and wireless transmission. Such functionality supports high-reliability quantitative reporting, even in remote or resource-limited diagnostic settings [10]. This feature enhances the system's clinical utility, making it suitable for tracking viral load dynamics during early-stage outbreaks or monitoring treatment efficacy in infected patients.
The results are visually summarized in Fig. 3, which illustrates the quantitative correlation between the dilution ratio of RT-LAMP amplicons and the corresponding DPV peak current intensity. Rather than relying on absolute quantification, this experiment employed a relative quantification strategy—an approach that aligns more closely with the demands of point-of-care (POC) diagnostics. The findings provide core validation for the platform's practical capability in delivering reliable, field-deployable quantitative diagnostics. As the RNA concentration increases, enhanced RT-LAMP amplification leads to a greater accumulation of double-stranded DNA and reaction by-products on the electrode surface, resulting in increased interfacial resistance and a corresponding decrease in the DPV peak current.
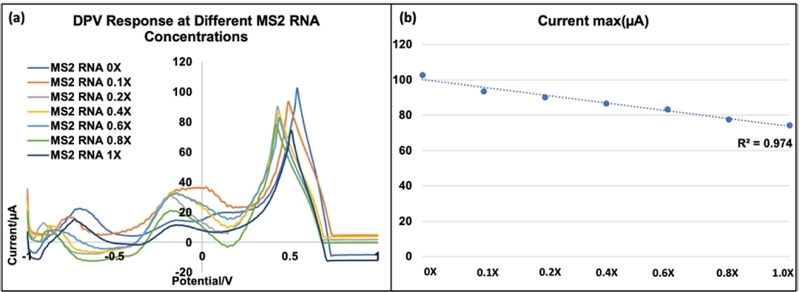
DPV analysis results following RT-LAMP amplification of SARS-CoV-2 RNA at various concentrations. The dilution series was prepared from a positive SARS-CoV-2 RNA sample, where 1× corresponds to approximately 1.5 × 10³ copies per reaction based on a 20 µL RT-LAMP reaction with a 1 µL RNA template input. Progressive dilution resulted in a proportional decrease in the DPV peak current intensity, thereby demonstrating the platform's relative quantitative capability. The lowest detectable concentration was approximately 150 copies per reaction.
Under the present experimental conditions, the lowest concentration that could be consistently distinguished from the negative control was approximately 150 copies per reaction, representing the estimated limit of detection (LOD) of the proposed electrochemical RT-LAMP platform.
3.5. Specificity Assessment: Evaluation of Non-Specific Amplification Suppression
One of the most critical performance indicators in nucleic acid-based diagnostics is specificity, which determines the diagnostic accuracy in distinguishing target-positive samples from those containing closely related non-target sequences. In this study, we evaluated whether the proposed electrochemical RT-LAMP platform exhibits high specificity toward SARS-CoV-2 RNA while effectively suppressing non-specific amplification from other respiratory viruses.
To assess this, total RNA from several representative respiratory pathogens—including influenza A, respiratory syncytial virus (RSV), and adenovirus—was subjected to RT-LAMP under identical reaction conditions (65°C for 60 minutes), followed by electrochemical analysis using differential pulse voltammetry (DPV) on the same electrode-filter assembly.
The results demonstrated that for all non-target RNA samples, no significant peak current was detected within the scan range of −0.2 to +0.6 V. The observed signals remained at baseline or background levels (typically below a few nanoamperes), confirming that the LAMP primer set effectively suppresses unintended amplification events due to its high sequence specificity [2,4,7].
In contrast, the positive control sample containing SARS-CoV-2 RNA consistently produced a distinct oxidation peak within the range of +0.3 to +0.4 V, validating the system’s capacity to sensitively and quantitatively detect changes in the interfacial electrochemical properties that occur specifically in the presence of the target nucleic acid.
This specificity is attributed to the intrinsic design of LAMP, which employs six primers that simultaneously bind to eight distinct regions of the target sequence. This multilayered binding mechanism inherently minimizes off-target hybridization, leading to a significantly reduced false-positive rate, even in the presence of closely related viral genomes [9].
Furthermore, the absence of detectable signal cross-reactivity in DPV measurements strongly suggests that the platform’s detection mechanism is robust against interference based on sequence similarity. This robustness enhances its reliability in complex clinical scenarios involving coinfections or seasonal outbreaks of multiple respiratory pathogens.
Fig. 4 visually summarizes these findings, illustrating that all non-specific RNA samples yielded current responses indistinguishable from the negative control (NC). This highlights the high specificity of the electrochemical readout and the diagnostic reliability of the integrated RT-LAMP-electrode platform, particularly in point-of-care testing (POCT) settings where rapid and accurate pathogen discrimination is crucial.
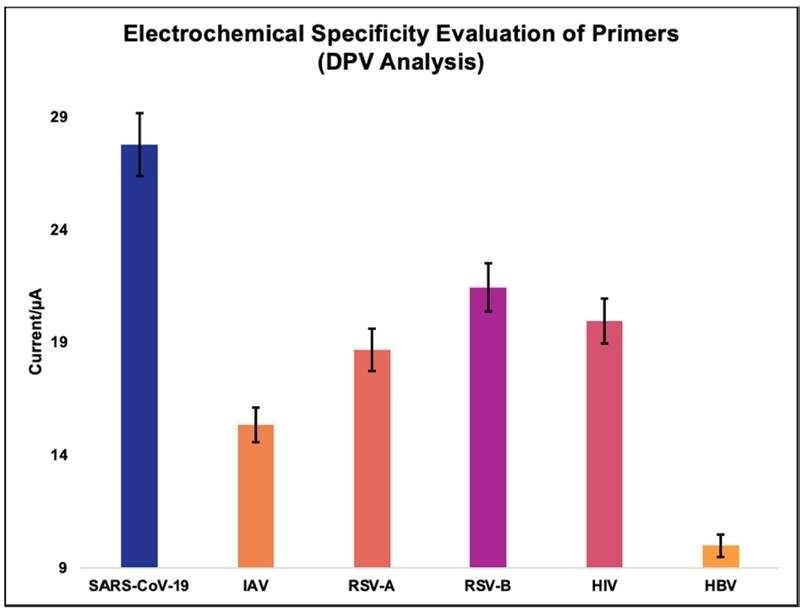
DPV analysis of RT-LAMP reactions targeting a variety of non-SARS-CoV-2 viral RNAs. No significant peak currents were observed under any non-specific conditions, confirming the high sequence specificity of the designed primer set.
Despite the promising performance demonstrated in this study, several limitations should be acknowledged. First, the clinical validation was conducted as a pilot study using a limited number of residual clinical specimens, which may not fully represent the heterogeneity of real-world clinical conditions. In addition, the current evaluation primarily focused on a restricted range of sample types, and further studies incorporating diverse specimen formats, such as saliva and nasopharyngeal swabs, are required to comprehensively assess clinical applicability. Moreover, the performance of the platform under low viral load conditions was not extensively evaluated in this study. Future work will therefore focus on large-scale clinical validation with expanded patient cohorts and varied sample types to further establish the robustness, sensitivity, and clinical utility of this point-of-care diagnostic platform.
3.6. Discussions
The results presented in this study demonstrate the feasibility of integrating RT-LAMP amplification with electrochemical signal transduction and smartphone-based readout into a compact and portable point-of-care testing (POCT) platform. Through a systematic evaluation of thermal stability, electrochemical signal generation, relative quantitative responsiveness, and target specificity, the proposed system was shown to reliably distinguish SARS–CoV–2–positive samples from negative and non-target controls while maintaining stable and reproducible signal outputs. These findings suggest that integrating isothermal amplification with electrochemical detection offers a practical analytical framework for decentralized molecular diagnostics.
Despite these promising results, several limitations of the present study should be acknowledged. First, the clinical validation was conducted as a pilot-scale investigation using a limited number of residual clinical specimens. As such, the current dataset may not fully capture the variability encountered in real-world clinical environments, including differences in patient populations, viral load distributions, and sample collection conditions. Second, the experimental evaluation primarily focused on a restricted range of specimen types, and the platform's performance has not yet been extensively assessed using alternative clinical samples, such as saliva or nasopharyngeal swabs. In addition, the analytical performance of the system under low viral load conditions was not systematically investigated in this study.
It is also important to note that the quantitative analysis presented here is based on relative electrochemical signal responses rather than absolute viral copy number determination. While this relative quantification strategy is well-aligned with the practical requirements of POCT applications—particularly for rapid screening and decentralized testing—it does not establish standardized clinical performance metrics, such as sensitivity, specificity, or dynamic range. Accordingly, the present study was designed as a proof-of-concept investigation, focusing on the feasibility and analytical signal responsiveness of the electrochemical RT-LAMP platform rather than on defining definitive clinical performance parameters through large-scale clinical validation studies.
Future work will therefore focus on expanding clinical validation with larger and more diverse patient cohorts, incorporating multiple specimen types, and evaluating system performance across a broader range of viral concentrations. In addition, direct comparative studies with established diagnostic modalities, including qRT-PCR and commercially available rapid antigen tests, will be necessary to define further the clinical utility, diagnostic accuracy, and translational potential of the proposed platform. Collectively, these efforts are expected to strengthen the positioning of the electrochemical RT-LAMP system as a robust, deployable molecular diagnostic tool for point-of-care applications in both clinical and resource-limited settings.
4. CONCLUSIONS
In this study, we developed and evaluated a portable, smartphone-integrated electrochemical point-of-care testing (POCT) platform for detecting SARS-CoV-2. The proposed system combines reverse transcription loop-mediated isothermal amplification (RT-LAMP) targeting the viral N gene with differential pulse voltammetry (DPV), enabling the direct electrochemical readout of nucleic acid amplification products without the need for optical components or complex instrumentation.
By integrating a paper-based electrode interface with isothermal amplification and smartphone-based signal acquisition, the platform demonstrated stable thermal control, reliable electrochemical signal generation, and reproducible analytical performance. The system was able to clearly discriminate SARS–CoV–2–positive samples from negative controls and non-target respiratory viruses, confirming its analytical specificity. In addition, concentration-dependent electrochemical signal responses were observed, supporting the feasibility of relative quantitative analysis within a compact and portable diagnostic format.
The incorporation of a battery-powered heating module, a portable potentiostat, and real-time smartphone visualization further highlights the practical advantages of the proposed platform for decentralized testing environments. These design features reduce dependence on centralized laboratory infrastructure, enabling on-site molecular diagnostics with minimal hardware and simplified operational workflows.
Overall, this work provides a proof-of-concept validation of an electrochemical RT-LAMP–based POCT platform that integrates amplification, detection, and digital readout into a single portable system. The results demonstrate that electrochemical signal transduction can serve as a viable alternative readout strategy for isothermal nucleic acid amplification, supporting the potential applicability of this approach in field-deployable molecular diagnostic settings.
Acknowledgments
This research received no external funding.
REFERENCES
-
H. Chen, K. Liu, Z. Li, P. Wang, Point-of-care testing for infectious diseases: Recent developments and trends, Clin. Chim. Acta 493 (2019) 138–147.
[https://doi.org/10.1016/j.cca.2019.03.008]

-
D. Das, C.W. Lin, H.S. Chuang, LAMP-based point-of-care biosensors for rapid pathogen detection, Biosensors 12 (2022) 1068.
[https://doi.org/10.3390/bios12121068]

-
G.C. Biswas, S. Choudhury, M.M. Rabbani, J. Das, A review on potential electrochemical point-of-care tests targeting pandemic infectious disease detection, Chemosensors 10 (2022) 269.
[https://doi.org/10.3390/chemosensors10070269]

-
C. Patiti, P.S. Sfragano, S. Laschi, S. Pillozzi, A. Boddi, O. Crociani, et al., Chip-based and wearable tools for isothermal amplification and electrochemical analysis of nucleic acids, Chemosensors 10 (2022) 278.
[https://doi.org/10.3390/chemosensors10070278]

-
W. Liu, F. Yue, L.P. Lee, Integrated point-of-care molecular diagnostic devices for infectious diseases, Acc. Chem. Res. 54 (2021) 4107–4119.
[https://doi.org/10.1021/acs.accounts.1c00385]

-
Q. Li, Y. Li, Q. Gao, C. Jiang, Q. Tian, C. Ma, et al., Real-time monitoring of isothermal nucleic acid amplification on a smartphone using a portable electrochemical device for home-testing of SARS-CoV-2, Anal. Chim. Acta 1229 (2022) 340343.
[https://doi.org/10.1016/j.aca.2022.340343]

-
M. Trotter, A. Schreiber, D. Kleinknecht, Z. Bagherian, F. von Stetten, N. Borst, Pathogen-specific electrochemical real-time LAMP detection using universal solid-phase probes on carbon electrodes, ACS Sens. 10 (2024) 1788–1796.
[https://doi.org/10.1021/acssensors.4c02492]

-
D. Das, M. Masetty, A. Priye, Paper-based loop-mediated isothermal amplification (LAMP) platforms: Integrating the versatility of paper microfluidics with accuracy of nucleic acid amplification tests, Chemosensors 11 (2023) 163.
[https://doi.org/10.3390/chemosensors11030163]

-
A. Rivas-Macho, U. Eletxigerra, R. Diez-Ahedo, Á. Barros, S. Merino, F. Goñi-de-Cerio, et al., Development of an electrochemical sensor for SARS-CoV-2 detection based on loop-mediated isothermal amplification, Biosensors 13 (2023) 924.
[https://doi.org/10.3390/bios13100924]

-
H. Ahn, B.S. Batule, Y. Seok, M.-G. Kim, Single-step recombinase polymerase amplification assay based on a paper chip for simultaneous detection of multiple pathogens, Anal. Chem. 90 (2018) 10211–10216.
[https://doi.org/10.1021/acs.analchem.8b01309]

-
Y. Seok, H.-A. Joung, J.-Y. Byun, H.-S. Jeon, S.J. Shin, S. Kim, et al., A paper-based device for performing loop‑mediated isothermal amplification with real-time simultaneous detection of multiple DNA targets, Theranostics 7 (2017) 2220–2230.
[https://doi.org/10.7150/thno.18675]

-
K. Nagamine, K. Watanabe, K. Ohtsuka, T. Hase, T. Notomi, Loop-mediated isothermal amplification reaction using a non-denatured template, Clin. Chem. 47 (2001) 1742–1743.
[https://doi.org/10.1093/clinchem/47.9.1742]

-
Y.H. Baek, J. Um, K.J.C. Antigua, J.H. Park, Y. Kim, S. Oh, et al., Development of a reverse transcription–loop‑mediated isothermal amplification assay for rapid detection of SARS-CoV-2, Emerg. Microbes Infect. 9 (2020) 998–1007.
[https://doi.org/10.1080/22221751.2020.1756698]

-
A. Ainla, M.P.S. Mousavi, M.-N. Tsaloglou, J. Redston, J.G. Bell, M.T. Fernández-Abedul, et al., Open-source potentiostat for wireless electrochemical detection with smartphones, Anal. Chem. 90 (2018) 6240–6246.
[https://doi.org/10.1021/acs.analchem.8b00850]

-
A. Priye, S.W. Bird, Y.K. Light, C.S. Ball, O.A. Negrete, R.J. Meagher, A smartphone-based diagnostic platform for rapid detection of Zika, chikungunya, and dengue viruses, Sci. Rep. 7 (2017) 44778.
[https://doi.org/10.1038/srep44778]

-
X. Zhang, S.B. Lowe, J.J. Gooding, Brief review of monitoring methods for loop-mediated isothermal amplification (LAMP), Biosens. Bioelectron. 61 (2014) 491–499.
[https://doi.org/10.1016/j.bios.2014.05.039]

