
Engineering Lipid Mesophases for Biosensors
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Self-assembled lipid mesophases are gaining attention as versatile platforms for biosensing. Their polymorphic forms—including lamellar, hexagonal, and bicontinuous cubic phases—offer nanoscale architectures with distinct transport, mechanical, and optical characteristics. These structures can preserve the activity of biomolecules, facilitate efficient charge and mass transfer, and provide membrane-like interfaces for the detection of biomolecules, including metabolites, nucleic acids, proteins, and viruses. This review summarizes the fundamental principles of lipid mesophase formation and examines how their properties can be tuned by altering composition, introducing polymers or nanoparticles, or applying external stimuli. We further highlight recent advances in their use for electrochemical, optical, and affinity-based biosensing. We also discuss current limitations related to reproducibility, stability, and device integration, and identify opportunities in rational design, hybrid systems, and portable or implantable platforms. Overall, engineered lipid mesophases hold significant potential as foundations for next-generation biosensors that unite sensitivity, selectivity, and practical usability.
Keywords:
Lipid mesophase, Self-assembly, Biosensors, Cubic phase, Nanostructured materials, Electrochemical and optical sensing1. INTRODUCTION
Biosensors are analytical devices that integrate a biological recognition element with a transducer to detect analytes of interest [1-6]. They play a vital role in medical diagnostics, environmental monitoring, and food safety by converting biochemical interactions into measurable signals [7]. Traditional biosensors often employ enzymes, antibodies, or nucleic acids as the biorecognition element, coupled with transducers like electrodes or optical systems [8]. In recent years, there is growing interest in using lipid-based mesophases as innovative matrices for biosensors [9]. These lipid mesophases offer a unique biomimetic environment akin to cell membranes, which can stabilize fragile biomolecules (such as membrane proteins or enzymes) and facilitate their function in sensor devices [10]. The high internal surface area and tunable nanostructure of certain lipid mesophases allow incorporation of both hydrophilic and hydrophobic compounds, making them attractive for immobilizing enzymes, receptors, or even entire cells in biosensing platforms [11].
Lipid mesophases bridge the gap between solid and liquid: they are liquid crystalline, exhibiting molecular order like a crystal yet fluidity like a liquid [12]. This combination yields materials that are optically transparent, semi-solid or gel-like, and biocompatible. These properties are highly advantageous for sensors. For example, optical transparency allows optical readouts, while biocompatibility and fluid channels enable biomolecules to remain active [13]. Fig. 1 conceptually illustrates how amphiphilic lipid molecules self-assemble into various mesophases, driven by the hydrophobic effect and molecular geometry (quantified by the critical packing parameter) [14]. By choosing appropriate lipids and conditions, one can obtain lamellar bilayers, hexagonal phases, or bicontinuous cubic structures, each with distinct nano-architectures [15].
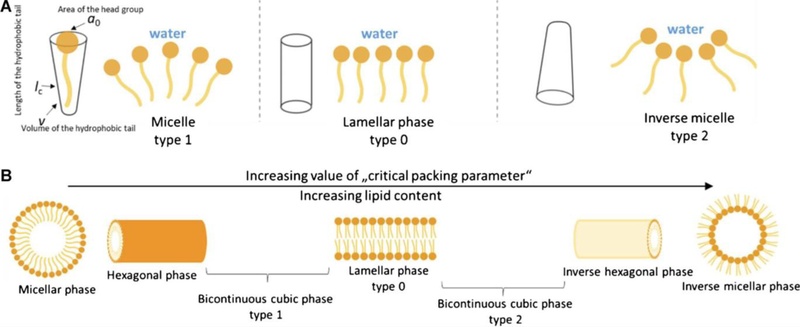
Self-assembly of amphiphilic lipids into different mesophases. (a) Lipid molecules can be approximated by shapes (cones, cylinders) based on the relative size of hydrophobic tails and hydrophilic headgroups. Upon contact with water, they spontaneously organize to minimize free energy. Cone-shaped amphiphiles (large head, small tail) tend to form type I (normal) structures like spherical or cylindrical micelles, whereas inverted-cone lipids (small head, bulky tail) favor type II (inverse) structures such as reverse micelles. (b) Schematic progression of self-assembled structures with increasing packing parameter: from spherical micelles (high positive curvature) to cylindrical micelles, lamellar bilayers (zero curvature), and further to inverse hexagonal and bicontinuous cubic phases (high negative curvature). These mesophases represent a continuum between liquid and solid states, combining fluidity with nanoscale order. Adapted from Ref. [9].
The motivation for using lipid mesophase systems in biosensors is multifold. First, they provide a biomembrane mimetic environment, crucial for maintaining the structure and activity of membrane-associated biomolecules such as G-protein coupled receptors or ion channels [16]. For instance, lipid membranes have been used to reconstitute ion channels in sensor devices, though traditional planar lipid membranes suffer from short lifetimes and instability [17]. Lipid mesophases (especially cubic phases) can overcome these limitations by forming stable 3D lipid matrices that are less prone to rupture [18]. Second, the high surface area and internal porosity of non-lamellar mesophases enable high loading of bioreceptors and efficient mass transport of analytes [19]. This can improve sensitivity and response time in sensors, as substrates and products can rapidly diffuse through the interconnected aqueous channels of a cubic phase or along the cylinder interiors of a hexagonal phase [20]. Third, lipid mesophases are inherently tunable – their nanostructure can be engineered by altering lipid composition, temperature, or additives [21]. This tunability allows tailoring of pore size, membrane curvature, and surface chemistry to optimize sensor performance for different analytes.
This review provides a comprehensive overview of the emerging field of lipid mesophases for biosensing applications. We first cover the fundamental self-assembly principles and characteristics of major lipid mesophases (lamellar, hexagonal, cubic), highlighting the material properties relevant to sensor design. Next, we discuss engineering strategies to modulate phase behavior and functionality, including lipid composition changes, polymer or nanoparticle incorporation, and stimuli-responsive control. We then survey a range of biosensing applications of lipid mesophases, categorized by transduction mode (electrochemical, optical) and sensing targets (small molecules, DNA/proteins, viruses), providing detailed examples from recent literature. Finally, we address the current challenges in this area – such as reproducibility, integration with devices, and stability – and outline future opportunities, including the potential of AI-guided design for novel lipid systems. Throughout the review, we integrate references to recent studies to illustrate the progress and prospects of lipid mesophase-engineered biosensors.
2. FUNDAMENTALS OF LIPID MESOPHASES
2.1 Lipid self-assembly and mesophase types
Lipids are amphiphilic molecules consisting of a hydrophilic “head” and one or more hydrophobic “tails.” In water, they spontaneously self-organize into supramolecular structures to shield their tails from water, a process driven by the hydrophobic effect. The specific structure formed depends on the lipid’s geometry and solution conditions. A useful predictor is the critical packing parameter (CPP), defined as v/(a0*lc) (where v is the tail volume, a0 the headgroup area, and lc the tail length) [22]. Lipids with small heads and bulky tails (CPP > 1) favor inverse (type II) phases with negatively curved lipid-water interfaces, while those with larger heads (CPP < 1) favor normal (type I) phases with positive curvature. When CPP ≈ 1, planar bilayer structures are favored (zero curvature). By adjusting parameters like lipid composition, hydration, temperature, and additives, one can access a rich polymorphism of lyotropic liquid-crystalline phases.
The principal lipid mesophases of interest in biosensing are: (1) Lamellar phase (Lα) – a stack of planar bilayers with no long-range curvature (like a stack of cell membranes); (2) Inverse hexagonal phase (HII) – a hexagonal array of lipid tubes (lipid monolayers wrapped around water cores), exhibiting high negative interfacial curvature; and (3) Bicontinuous cubic phases (QII) – three-dimensionally periodic lipid bilayer networks forming continuous tortuous aqueous channels. Table 1 compares these mesophase types in terms of structure, properties, and relevance to biosensors. Notably, the inverse bicontinuous cubic phase (often just called “cubic phase”) has drawn special attention for biosensors due to its unique combination of stability and internal nanoporosity [18]. In a cubic phase, a single lipid bilayer extends in 3D, separating two interpenetrating but non-intersecting water channel networks (typically with either gyroid (Ia3d), diamond (Pn3m), or primitive (Im3m) symmetry) [23]. This creates a robust, gel-like matrix with extensive internal surface area accessible to solvated analytes or biomolecules.
Other lyotropic phases exist (e.g., micellar cubic, sponge phase, cholesteric, etc.), but the lamellar, hexagonal, and bicontinuous cubic are most encountered with lipid amphiphiles like glycerol monooleate or phytantriol under relevant conditions [24]. For example, monoolein (glycerol mono-oleate) in excess water at room temperature forms a Pn3m cubic phase, whereas certain additives or higher temperatures can induce a transition to the HII phase [25]. Understanding these phase behaviors is crucial: the phase diagram of a lipid/water system tells us which mesophase will form at a given composition and temperature. In biosensing contexts, one must often ensure that the desired mesophase is stable under operating conditions (aqueous environment, ambient temperature, presence of salts or buffers). Significant research has been devoted to mapping lipid phase diagrams and identifying conditions to reliably obtain target mesophases [26].
2.2 Mesophase properties relevant to sensing
Each mesophase’s structure imparts distinct physical properties that can be harnessed in biosensors. Lamellar bilayers, for instance, provide a planar fluid membrane that is ideal for reconstituting membrane proteins and creating biomimetic interfaces (such as in field-effect transistor sensors or supported bilayer arrays) [27,28]. However, unsupported bilayers are fragile, and even supported ones can suffer from limited stability and protein diffusion into the solid support [29]. The cubic phase, by contrast, is a viscous, transparent gel that is stable and self-supporting – it does not require a solid substrate to maintain its structure [18]. It offers a bicontinuous aqueous network that facilitates 3D diffusion of ions and small molecules, which is beneficial for electrochemical sensor response and maintaining enzyme activity throughout the matrix [30]. The cubic phase is also isotropic (non-birefringent), which means it does not scatter light strongly; this optical clarity is useful for optical or spectroscopic biosensors [31]. The hexagonal HII phase is intermediate – it has high curvature like cubic, but only one-dimensional long-range order. It features straight cylindrical water cores, which could act as nanochannels [32]. HII phases tend to be less viscous than cubic phases, and while they are less studied for biosensors, they might be useful for creating aligned nanopore arrays or for hosting functional membrane proteins such as ion channels in a more ordered environment [33].
2.3 Phase behavior and transitions
It is important to note that lipid mesophases can transform from one type to another with changes in conditions. Many lipid systems exhibit temperature-dependent transitions (e.g., a lamellar-to-hexagonal transition at elevated temperature for certain phospholipids) [34]. Hydration level and pressure also influence mesophase stability. For biosensor operation, maintaining a stable phase is often critical. Unintended phase transitions (e.g., cubic-to-hexagonal) could alter the sensor’s performance by changing diffusion paths or expelling incorporated biomolecules. Therefore, researchers typically formulate the lipid composition to ensure the desired mesophase is the equilibrium state under operating conditions. For example, adding helper lipids like phosphatidylcholine can broaden the cubic phase stability range or prevent an unwanted transition by adjusting the lipid packing parameter [35]. Conversely, phase transitions can sometimes be exploited deliberately (as a sensing mechanism or for controlled release of a reporter molecule) if they can be triggered by an external stimulus [26]. In summary, a solid grasp of mesophase fundamentals and phase behavior is the foundation for engineering lipid mesophases to function reliably in biosensors.
3. ENGINEERING STRATEGIES FOR LIPID MESOPHASE SYSTEMS
3.1 Lipid composition tuning
Designing effective lipid mesophase-based biosensors requires the ability to modulate the phase structure and functionality to suit a given sensing application. Several engineering strategies have been developed to tune lipid mesophases. The simplest lever is to change the lipid molecules or mixtures thereof. Different lipids have different spontaneous curvature tendencies (e.g., monoglycerides like monoolein favor cubic phases, phosphocholine favors lamellar) [36]. By blending lipids, one can obtain intermediate structures or expand the stability of a desired phase. For example, incorporating a small fraction of a charged lipid (such as DOTAP, a cationic lipid) into a monoolein cubic phase can induce electrostatic swelling of the water channels, increasing their size [37]. Such swelling has been used to tune cubic pore size and even create “swollen” cubic phases that enhance diffusion rates. Charged or bulky headgroup lipids increase the hydration repulsion, often favoring the cubic over the hexagonal phase or converting a gel to a more fluid cubic structure. In a recent example, adding 4–5% DOTAP (a cationic amphiphile) to monoolein enabled the incorporation of large nucleic acids (mRNA strands ~4 kb) into cubosomes by electrostatic complexation [38]. The cationic charges helped load the negatively charged RNA while maintaining the Pn3m cubic structure, demonstrating how composition changes allow functionalization (here, nucleic-acid loading) without losing the desired phase. Similarly, adding lipids with photo-responsive groups or redox-active headgroups can endow the mesophase with new functionality while still self-assembling into the target nanostructure [39].
3.2 Polymer hybridization and stabilization
Another approach is to integrate polymers with lipid mesophases to improve their mechanical properties or impart responsiveness. Block copolymers can be used in two ways: (1) as stabilizers for dispersed lipid nanoparticles (cubosomes/hexosomes), and (2) as co-assembly components within the mesophase. In the first case, polymers like Pluronic F127 (a PEG–PPG copolymer) are commonly adsorbed on cubosome surfaces to prevent aggregation and provide a steric protective corona. These polymer-stabilized cubosomes remain stable in suspension and even under dilution, which is beneficial for making injectable sensor nanomaterials or thin-film coatings. In fact, cubosomes are often described as lipid nanoparticles with a polymer outer corona, reflecting this design. In the second approach, polymerizable or amphiphilic polymers can be mixed with lipids to form hybrid mesophases. For example, diacetylenic lipids can be polymerized in situ within a bilayer or cubic phase to “lock in” the structure. Cross-linking the lipid bilayer via polymerization yields a more rigid material that retains the nanostructure even under stress or dehydration. This can address stability issues for sensor deployment in the air or under flow. Researchers have successfully polymerized cubic phases to create nanoporous polymer matrices that still resemble the original lipid network. Another instance is grafting PEG chains onto lipids, producing “PEGylated” cubic phases that are more robust and less prone to protein fouling. Polymer-lipid hybrid mesophases thus combine the biofunctionality of lipids (biocompatibility, self-assembly) with the durability of polymers. These are promising for implantable or wearable biosensors where mechanical integrity is crucial.
3.3 Nanoparticle and additive inclusion
The incorporation of colloidal nanoparticles into lipid mesophases is an emerging strategy to modulate their structure or impart new functionalities (such as plasmonic or magnetic properties). Small hydrophilic nanoparticles can insert into the water channels or at the lipid/water interface. Their presence can stabilize or destabilize certain mesophases depending on concentration. For example, a study showed that adding 10 nm silica nanoparticles in low volume fraction expanded the pressure–temperature stability region of the cubic gyroid phase (QGII) in monoolein, effectively stabilizing the higher-curvature cubic phase over the lamellar phase [40]. The particles were thought to accumulate at domain boundaries, encouraging curvature towards cubic structures. However, at higher nanoparticle loading, the same system saw the cubic phase disappear in favor of lamellar or disordered phases, presumably because excess particles disrupted the lipid packing by forming a 'chain-of-pearls' aggregation that frustrated the ordered structure [41]. These results highlight that nanocomposites of lipids and particles need careful tuning: a small number of NPs can modulate mesophase curvature (potentially useful for increasing channel size or stability), but too many can break the periodic order. From a biosensing perspective, embedded nanoparticles can serve as signal transducers (e.g., fluorescent or Raman-active markers) or as anchoring points for biomolecules [42]. For instance, magnetic nanoparticles in a lipid mesophase could allow magnetic manipulation of the sensor matrix, and gold nanorods could enable plasmonic readouts or photothermal tuning of the phase [43]. The key is ensuring the additives do not abolish the beneficial mesophase structure. Researchers are also exploring biomolecule-loaded additives that integrate into mesophases to provide specific binding sites or signal amplification within the lipid matrix.
3.4 External stimuli control
One exciting aspect of lipid mesophases is their responsiveness to environmental conditions. This can be harnessed to create stimuli-responsive biosensors [44]. By design, mesophase transitions or structural changes can be triggered by changes in temperature, pH, light, or other stimuli. A straightforward example is thermally triggered phase transitions: certain lipid mixtures form a cubic phase at room temperature but transition to a hexagonal phase at ~50°C. If an analyte binding event produces heat, one could in principle detect that via a structural change. More relevant are chemical triggers: for example, pH-responsive lipids that change ionization state can cause a cubic-to-lamellar transition at a specific pH threshold. Embedding such lipids allows the mesophase to act as a pH sensor by structurally responding to acidity. Light-sensitive lipids (e.g., azobenzene-bearing amphiphiles) have been used to make light-triggerable mesophase systems. Upon UV or visible light, the photo-isomerization can alter the lipid’s shape (trans to cis) and drive a phase transition or a change in lattice parameter. This could enable optical switching of a sensor on and off, or a way to externally modulate the sensor’s calibration. Indeed, previous work demonstrated a light-activated release mechanism in a drug delivery context by incorporating a photo switchable lipid into a cubic phase, which upon illumination caused expulsion of the loaded drug molecules [45]. A similar idea could be applied to release a quenching agent or a signaling molecule in a biosensor in response to an optical cue. Finally, electrical or mechanical stimuli may also be applied: while lipid mesophases are not typically electro-active, an applied electric field might orient anisotropic mesophases or charged components within (for instance, aligning rod-like HII domains or causing electroosmotic flow in channels). Mechanically, the soft gel nature means they could deform under pressure – one could envision a pressure sensor where deformation of a mesophase changes its optical properties. These concepts remain exploratory, but the ability to externally control the sensor matrix offers opportunities for tunable and multi-modal biosensors.
In implementing these strategies, it is common to combine multiple approaches (Fig. 2). For example, one might use a polymer-stabilized cubosome with a tuned lipid composition and embedded nanoparticles to achieve the desired stability, biorecognition, and signal transduction. A concrete case from recent literature involved creating a redox-active enzyme cubosome by doping a cubic phase with a ferrocene-tagged lipid (to shuttle electrons) and entrapping glucose oxidase for glucose sensing. This design drew on composition tuning, biomolecule loading, and resulted in a stable film on an electrode with efficient electron transfer – an outcome of multiple synergistic engineering steps. As another example, to detect a specific protein, one could incorporate a lipid-linked affinity ligand into the mesophase and also include fluorescence reporter nanoparticles in the aqueous channels. The possibilities are expansive, and we are only beginning to explore the combinatorial design space of these soft, nanostructured materials. In the future, rational design aided by computational modeling or machine learning may guide the selection of lipid mixtures and additives to achieve a target phase and function. Indeed, the pharmaceutical field has started using AI to optimize lipid nanoparticle formulations for drug delivery, and similar techniques could be adapted to optimize lipid mesophases for sensing. The next section will illustrate how these engineered lipid mesophases are being applied in diverse biosensing modalities.
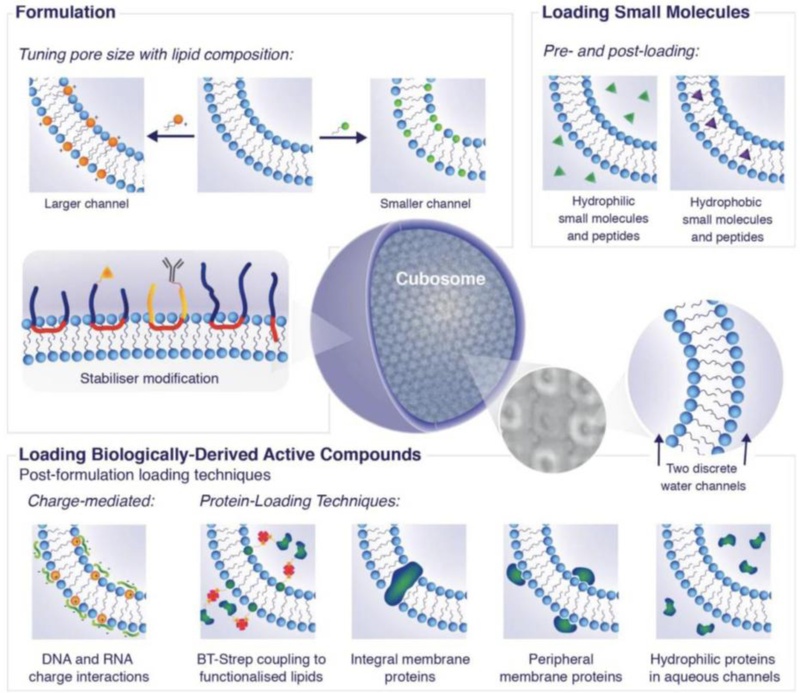
Design and engineering of cubic phase lipid nanoparticles for biomedical applications (schematic overview). This diagram summarizes approaches to tailor cubic phase lipid nanoparticles (cubosomes) via formulation and loading strategies. Formulation tuning: The lipid composition can be altered (varying tail length, saturation, headgroup charge/size) to adjust phase behavior and pore size. The choice of stabilizer (polymer or particulate corona) can target specific cells or add functionality. Molecule loading methods: Cubosomes can be loaded with a wide variety of cargo: small hydrophobic molecules partition into the lipid bilayer, while hydrophilic molecules reside in water channels. Bio-macromolecules can be incorporated either during formation (co-lyophilization or premixing) or post-formation (incubation). Specialized methods allow insertion of membrane proteins (integral proteins embedded in the lipid bilayer) or attachment of targeting ligands on the particle surface. Charged polymers and adapter molecules (e.g. biotin-streptavidin linkers) facilitate functionalization with enzymes, antibodies, DNA, etc. This engineered modularity enables the design of cubosome-based biosensors customized for different analytes – for instance, an enzyme-loaded cubosome for metabolite detection, or a cubosome displaying aptamers for pathogen detection. Adapted from Ref. [46].
4. APPLICATIONS IN BIOSENSING
Lipid mesophases have enabled a variety of biosensing approaches, taking advantage of their biocompatible nano-architecture to interface biological recognition events with transducers. Here, we survey major categories of biosensors utilizing lipid mesophases, highlighting representative examples.
4.1 Electrochemical Biosensors
Electrochemical transducers are widely used in biosensors due to their sensitivity and ease of miniaturization. Lipid mesophases, especially the cubic phase, have found a natural fit in electrochemical enzyme biosensors, where they are typically formed directly on electrode surfaces as hydrated, porous films rather than acting as insulating layers. In such sensors, enzymes embedded within the lipid matrix convert the target analyte into electroactive products, while the interconnected aqueous channels of the mesophase enable efficient ion and analyte transport to the electrode. The resulting current or potential change is then measured, either through direct diffusion of electroactive species (e.g., H2O2) or via embedded redox mediators that facilitate electron transfer.
Traditionally, enzymes are immobilized on electrodes by cross-linking or entrapment in hydrogels, but these methods can impair enzyme activity or leach out over time. By contrast, the lipid cubic phase offers a nanoporous, biomembrane-mimetic scaffold that hosts enzymes at the electrode interface while maintaining them in a native-like aqueous/membrane environment. Early work in the 1990s showed that enzymes such as glucose oxidase, when incorporated into a glycerol monooleate cubic phase on an electrode, retained activity and enabled the detection of glucose, lactate, and other metabolites via amperometry, with H2O2 generated by the enzymatic reaction serving as the electroactive signal [46]. This concept of an “enzyme in a cubic lipid matrix” has since evolved into more advanced designs.
A state-of-the-art example is a glucose biosensor based on a ferrocene-doped cubic phase film. In this 2023 study by Liu et al., monoolein cubic phase was doped with a small fraction of a ferrocene-functionalized amphiphile (Fc12), which acts as a redox mediator shuttling electrons from glucose oxidase (GOx) to the electrode. GOx was co-encapsulated in the cubic phase. The bicontinuous lipid matrix held GOx in proximity to the electrode and Fc mediator, while the interconnected water channels allowed glucose to diffuse in and H2O2 to diffuse to the electrode. This resulted in a stable, reagent-less sensor for glucose with a linear response in the 2–17 mM range (covering physiologically relevant glucose concentrations). SAXS confirmed that the cubic Pn3m structure was preserved even with the ferrocene and enzyme present. The sensor achieved a KM close to that of free GOx, indicating the enzyme remained functional in the lipid environment. Fig. 3 illustrates this design: essentially a hybrid lipid/enzyme electrochemical interface where the lipid mesophase provides a biomembrane mimic and a nanowire network for electron transfer via the embedded redox shuttle.
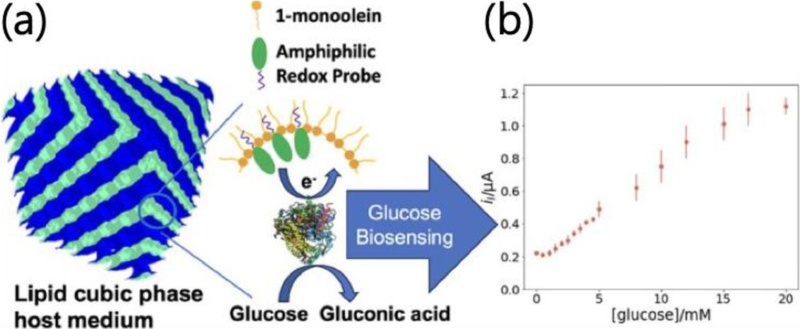
Electrochemical biosensor using a redox-active lipid cubic phase. Schematic (a) of a cubic phase film on an electrode, composed of monoolein lipid with an added ferrocene-derived lipid (red stars) that mediates electron transfer. Enzyme (green) is immobilized in the lipid matrix and catalyzes conversion of analyte (e.g., glucose) to a product that reduces the ferrocene mediator. The mediator shuttles electrons to the electrode, generating a current proportional to analyte concentration. The cubic phase’s 3D nanostructure allows efficient diffusion of glucose and mediator through its water channels and intimate contact of enzyme with the electrode surface without need for harsh chemical linking. (b) Typical calibration curve (current vs glucose concentration) showing a linear response range suitable for physiological sensing. Such a design demonstrates the synergy of lipid mesophases and electrochemistry, achieving high enzyme loading, preserved activity, and enhanced electron transport in a stable configuration. Adapted from Ref. [47].
Beyond glucose sensors, similar lipid cubic phase systems have been employed for detecting lactate, urea, cholesterol, and other substrates by choosing appropriate oxidase or dehydrogenase enzymes [48]. A notable advantage is the “third-generation” biosensor configuration, where the enzyme’s electrons are directly transferred to the electrode rather than relying on oxygen/hydrogen peroxide detection [49]. The cubic phase can facilitate this direct electron transfer (DET) by positioning redox cofactors or prosthetic groups in proximity to the electrode. For example, cellobiose dehydrogenase, a redox enzyme capable of DET, was incorporated into a lipid cubic film and showed improved catalytic current and stability versus a traditional adsorbed enzyme electrode [50]. In that 2019 study, the mesophase provided a protective, hydrated environment that prolonged enzyme activity and allowed repeated measurements with minimal signal drift.
Another area is electrochemical impedance biosensors, where changes in an electrode-immobilized lipid mesophase modulate the impedance. For instance, a free-standing lipid cubic film with embedded ion-channel proteins was demonstrated as an impedance-based sensor for channel blockers [51]. The lipid mesophase film acted as a robust “black lipid membrane” alternative, and the binding of analytes that block the ion channels led to measurable impedance changes. This highlights that lipid mesophases can incorporate not just enzymes but also membrane proteins directly, enabling biomimetic sensors for toxins, neurotransmitters, or other ligands that interact with ion channels or GPCRs. The stability of the cubic phase was key to support the protein function in a free-standing configuration, which would be challenging with a planar bilayer.
It is worth noting that many electrochemical biosensors using lipid mesophases are geared towards point-of-care diagnostics [52]. The stability and self-contained nature of these lipid films make them suitable for portable devices like test strips or wearable sensors. For example, envision a disposable electrode strip coated with an enzyme-loaded cubic phase that can detect metabolites in a drop of blood. Coupled with miniaturized electronics and wireless readouts, such a system could transmit results to a smartphone for real-time health monitoring [53,54]. Indeed, the integration of biosensors with wearable technology is a major trend. While not all current wearable sensors use lipid mesophases, the concept is gaining traction – e.g., a recent wearable sweat sensor combined an electrochemical enzymatic assay with a colorimetric readout for digital measurement of glucose and lactate in perspiration. In the future, lipid mesophase-based sensing elements could be incorporated into such wearable platforms to take advantage of their biocompatibility and high performance. For instance, a flexible microfluidic patch could contain a lipid cubic phase with enzymes to continuously measure sweat metabolites. The challenge of operational stability under continuous use will need to be addressed, but progress in polymer-lipid hybrids and solid-supported mesophase films are promising in this regard.
4.2 Optical and Affinity-Based Sensors
Liquid crystals are well-known for their optical properties, and lipid mesophases can be leveraged in optical biosensing techniques. The general principle in many optical LC biosensors is that an analyte binding event perturbs the orientational order or periodic structure of the liquid crystal, leading to an optical signal change [55]. Thermotropic liquid crystals have been extensively studied in this context [56], but lipid lyotropic phases offer some distinct opportunities. For instance, photonic crystal sensors have been envisioned using the periodic cubic phase lattice – a periodicity of ~10 nm is too small for photonic bandgaps in visible range, but it could influence Förster resonance energy transfer (FRET) or diffraction of X-rays for high-resolution sensing [57,58]. More practically, the transparency of cubic phases means one can incorporate optical reporters (dyes, quantum dots) inside and read changes in their emission or absorption when analytes interact.
One example of an optical biosensor utilizing a lipid mesophase involved a quartz crystal microbalance with dissipation (QCM-D) and fluorescent readout to detect a bacterial toxin [59]. Here, phytantriol cubic phase particles were functionalized with a glycolipid that specifically binds cholera toxin B [60]. These functional cubosomes were then tethered to a QCM sensor surface via a biotin-NeutrAvidin linkage. When cholera toxin was introduced, it bound to GM1 on the cubosome, increasing the mass (detected by QCM frequency shift) and could also be confirmed by fluorescence since the toxin was labeled. The cubosome provided a fluid, 3D membrane environment presenting the glycolipid receptor, resulting in very low non-specific binding and a strong specific signal. This can be seen as an affinity biosensor, where the affinity element (GM1) is embedded in a lipid mesophase, and the binding is transduced by both acoustic (QCM) and optical means. The success of this approach suggests that other glycolipids or membrane receptors could be similarly integrated into mesophases to capture pathogens or biomarkers [64]. For example, a sialic-acid-containing lipid could be used in a mesophase to capture influenza virus hemagglutinin, or an aptamer-functionalized lipid could bind a specific protein target.
Another optical strategy is using liquid-crystal droplet sensors, which has been demonstrated with droplets of chromonic liquid crystals and could be extended to lipid-based smectic or hexagonal droplets [61]. The idea is that binding of analyte at the droplet interface changes the anchoring of the LC, switching the droplet’s internal ordering and thereby its appearance between bright and dark under polarized light [62]. While most such studies used thermotropic LCs [63], one can imagine using lamellar phase multilamellar vesicles that change optical properties upon analyte binding. In fact, colorimetric liposome-based assays exist, although they rely on aggregation-induced color change rather than LC orientation [64].
In terms of fluorescence-based sensors, lipid mesophases have been used to host fluorescent proteins or organic dyes that report on local chemical conditions. For instance, pH-sensitive fluorescent molecules entrapped in a cubic phase can act as a sensor for enzymatic reactions that produce or consume protons: the mesophase confines the dye and perhaps the enzyme together, allowing FRET or intensity changes to be localized and enhanced [11]. One study integrated bacteriorhodopsin into a cubic phase and monitored its absorption spectrum shifts as a function of membrane state, effectively creating an optical sensor for membrane perturbations [10]. Although that was more of a bioelectronic device demonstration, it indicates the feasibility of optical readouts from proteins in mesophases.
Portable and point-of-care optical sensors can also benefit from lipid mesophases. A concrete development in recent literature is an electrophoretic colorimetric biosensor for sweat metabolites, where a color change indicates analyte level in a user-readable way [65]. While that device did not employ lipid mesophases, it underscores the trend of integrating optical signal transduction for portable diagnostics. Lipid mesophases could be incorporated as sensing elements in such devices – for example, a small window in a microfluidic device could contain a lipid LC that changes color or transparency when the target analyte is present [66]. Because lipid mesophases are soft and can be made biocompatible, one could envision sticking such a device on skin or a wound to optically monitor biomarkers.
4.3 Affinity Biosensors (DNA, Protein, Virus Detection)
Affinity biosensors rely on selective binding between a target and a capture molecule. Lipid mesophases can serve as novel platforms for presenting capture molecules in a more biomimetic and effective manner. One approach is to incorporate biorecognition lipids or conjugates into the mesophase. We saw an example with GM1 for cholera toxin [64]. Similarly, biotinylated lipids can be used to attach streptavidin-conjugated antibodies within a mesophase, essentially creating an antibody-decorated lipid matrix. This was demonstrated in a simplified form where a biotinylated cubic phase was used to concentrate a biotin-binding protein (avidin) which could then capture a biotinylated target – a strategy that can be generalized to many ligand-receptor pairs by using the appropriate lipid conjugates.
DNA and RNA sensors could also benefit from lipid mesophases. One can functionalize lipids with single-stranded DNA and mix a small percentage of these into a bulk lipid mesophase. The result is a nanostructured matrix with DNA probes protruding into the aqueous channels. If the complementary DNA or an aptamer target is present, it will hybridize or bind within the mesophase. Because the mesophase confines the probes at high local concentration and in a 3D network, binding events might be transduced by changes in the matrix. There have been reports of liposome-based DNA sensors where the binding of target causes liposome aggregation or content release [67]. By extension, a cubic phase could be used to improve stability and possibly allow continuous monitoring. In one design, a cubic phase containing embedded DNA was envisioned as in situ hybridization matrix: when the target DNA enters, it base-pairs with the probe, and because the probes were linked to a quencher, the displacement of a reporter strand could generate fluorescence [68]. This concept merges the ideas of solid-phase DNA biosensors with the advantages of a solution-phase reaction environment provided by the mesophase.
For virus detection, lipid mesophases can mimic the host cell surface that viruses bind to [69]. Many viruses initially attach to specific lipid or protein receptors on cell membranes. By incorporating those receptors into a mesophase, one can create a virus trap that generates a signal upon binding. For example, certain cholesterol-containing glycolipids serve as entry points for viruses like polyomavirus; embedding such glycolipids in a supported cubic phase coating on an optical sensor could capture viruses from a sample [70]. A Swiss research group proposed a cubic-phase-based sensor to detect whole pathogens by the principle of them getting trapped in the nanochannels and causing a measurable change in optical or electrochemical response [71]. While this is still a nascent idea, the approach could yield rapid, cost-effective pathogen detectors that do not rely on antibodies. Instead, the virus is recognized by the lipid matrix itself. A practical example is a reported bird flu virus sensor that used a lipid-coated surface to capture virus particles and achieved detection in minutes, though that utilized a supported bilayer rather than a non-lamellar phase [29]. The incorporation of more complex phases like cubic or hexagonal might further enhance sensitivity by increasing surface area and binding capacity.
In summary, affinity-based sensing with lipid mesophases leverages the ability to present biorecognition elements in a fluid, native-like environment. The fluidity ensures that binding can occur with minimal steric hindrance. Additionally, the multi-modal nature of mesophases means that one binding event could potentially be read out in multiple ways, increasing robustness. As these systems are developed, we expect to see lipid mesophases enabling detection of a broad array of biomolecules - from small toxins up to whole viruses - via selective affinity interactions.
4.4 Portable and Integrated Sensors
Many applications demand sensors that are not only sensitive and selective but also portable, inexpensive, and able to operate in real-world conditions. Lipid mesophase-based sensors are being adapted to meet these needs. The soft and biocompatible nature of lipid materials makes them attractive for implantable biosensors. There is ongoing research on integrating lipid-sensing layers with flexible electronics and microfluidics. One example is the use of 3D printing to pattern lipid mesophases into device architectures. Semi-solid extrusion of a lipid cubic phase ink has been demonstrated to create 3D structures that retain the mesophase; while that was for drug delivery, similar techniques could print micro-patterned sensor arrays with different lipid formulations for multiplexed detection [72].
Another avenue is coupling lipid mesophases with MEMS/NEMS devices. For instance, imagine a microcantilever coated with a lipid film - when analytes insert or bind, the surface stress changes and the cantilever bends. The lipid coating could be lamellar or non-lamellar; the key is that it provides a biologically receptive interface on the otherwise inert microdevice. This concept has been applied with lipid bilayers on cantilevers to detect protein-lipid interactions; extending it to cubic phases could increase the effect.
As portable sensors move towards multi-analyte and continuous monitoring, multi-functional lipid mesophases might be developed. These would incorporate several sensing chemistries in one matrix - for example, a single cubic phase could host both a glucose oxidase and a lactate oxidase, each linked to a different mediator or optical reporter, thus detecting two metabolites concurrently. The advantage of the mesophase is that it can compartmentalize functions in the same physical space while sharing the same transducer. This could simplify device design.
It is also worth noting that from a manufacturability standpoint, lipid mesophase sensors can be amenable to scaling and preservation. Techniques like lyophilization can turn a lipid cubic phase with biomolecules into a dry powder that is stable for storage; upon rehydration, it re-forms the mesophase and becomes active [13]. This could lead to field-deployable kits: for example, a cartridge with a dry lipid/enzyme mixture that is stable at ambient temperature, which, when a drop of sample is added, forms a sensing gel ready to measure. The robust self-assembly of lipids is a huge benefit here - the detailed nanostructure forms automatically, no precise nanofabrication needed, which keeps costs low.
In closing this section, lipid mesophases are enabling biosensors across a spectrum of methods: electrochemical, optical, and affinity-based. Each exploits different attributes of these fascinating soft materials. The applications demonstrated so far are proof-of-concept, but they point toward real devices in the near future that could, for instance, allow patients to continuously monitor health markers using a biocompatible interface, or enable on-site detection of pathogens with a simple dipstick whose specificity comes from lipid-encoded biomimicry. The next section will discuss the challenges that must be addressed to fully realize this potential, and the opportunities on the horizon – including how advanced tools like artificial intelligence might accelerate development in this interdisciplinary field.
5. CHALLENGES AND OPPORTUNITIES
Engineering lipid mesophases for biosensors faces several key hurdles. Reproducibility in phase formation is sensitive to composition, hydration, and processing, highlighting the need for scalable methods such as microfluidic mixing. Integration with devices remains challenging since soft, hydrated phases must interface with rigid electronics; hybrid supports like nanoporous membranes or polymer coatings provide stability. Stability and shelf-life are limited by dehydration and biomolecule degradation, but strategies such as sugar stabilizers, lyophilization, and extremophile enzymes offer solutions. In complex media, biofouling and nonspecific adsorption can disrupt performance, necessitating antifouling strategies like PEGylation. Regulatory and biocompatibility issues must also be addressed, particularly for implantable devices. On the opportunity side, AI-guided design promises to accelerate lipid formulation and signal interpretation, while bio-inspired lipids and exosome-mimetic systems may unlock new sensing capabilities.
6. CONCLUSIONS
Lipid mesophases enable functional biomimicry, offering environments that preserve biomolecule activity and enhance analyte recognition. The field’s future lies in integration and miniaturization, where lipid sensors merge with flexible and wearable electronics. Multi-modal and responsive sensing strategies, combined with computational design, will further expand capabilities. Overall, engineered lipid mesophases are advancing from conceptual demonstrations to practical biosensors, promising devices that combine sensitivity, selectivity, and compatibility with biological systems.
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. RS-2023-00209955). This work was partially supported by Korea Institute of Science and Technology (26E0141). This work was also supported by Korea Institute of Planning and Evaluation for Technology in Food, Agriculture and Forestry (IPET) through High-Risk Animal infectious Disease Control Technology Development Program, funded by Ministry of Agriculture, Food and Rural Affairs (MAFRA)(RS-2024-00396818). This work was supported by the Technology Innovation Program for Materials and Components (RS-2025-25409767) funded by the Ministry of Trade, Industry and Energy (MOTIE).
REFERENCES
-
A.P.F. Turner, I. Karube, G.S. Wilson, Biosensors: Fundamentals and Applications, 1st ed., Oxford University Press, Oxford, 1987.
[https://doi.org/10.1016/S0003-2670(00)85361-1]

-
T. Kim, I. Park, Skin-interfaced wearable biosensors: A mini-review, J. Sens. Sci. Technol. 31 (2022) 71–78.
[https://doi.org/10.46670/JSST.2022.31.2.71]

-
Y. Jeong, E.-K. Lim, Exosome-based diagnostics: Emerging tools for early and non-invasive disease detection, J. Sens. Sci. Technol. 34 (2025) 410–422.
[https://doi.org/10.46670/JSST.2025.34.5.410]

-
J.W. Park, D.H. Park, J.H. Kim, Self-assembled nanostructures for high-performance sensor applications, J. Sens. Sci. Technol. 34 (2025) 423–431.
[https://doi.org/10.46670/JSST.2025.34.5.423]

-
H.H. Jung, Nanostructured ZnO interfaces for highly sensitive and selective dopamine sensing, J. Sens. Sci. Technol. 34 (2025) 526–531.
[https://doi.org/10.46670/JSST.2025.34.5.526]

-
J. Song, I. Jung, J. Yang, H. Kim, Non-invasive glucose measurement using electric field and temperature sensor, J. Sens. Sci. Technol. 34 (2025) 549–554.
[https://doi.org/10.46670/JSST.2025.34.5.549]

-
W. Heo, Q. Wang, S. Choi, J. Shin, S.J. Park, S. Park, et al., Lab-in-a-cartridge for real-time detection of tuberculosis via precise measurement of urinary lipoarabinomannan, Nat. Commun. 16 (2025) 10299.
[https://doi.org/10.1038/s41467-025-65217-w]

-
A. Sassolas, L.J. Blum, B.D. Leca-Bouvier, Immobilization strategies to develop enzymatic biosensors, Biotechnol. Adv. 30 (2012) 489–511.
[https://doi.org/10.1016/j.biotechadv.2011.09.003]

-
M. Zatloukalova, L. Poltorak, R. Bilewicz, J. Vacek, Lipid-based liquid crystalline materials in electrochemical sensing and nanocarrier technology, Microchim. Acta 190 (2023) 187.
[https://doi.org/10.1007/s00604-023-05727-w]

-
E.M. Landau, J.P. Rosenbusch, Lipidic cubic phases: a novel concept for the crystallization of membrane proteins, Proc. Natl. Acad. Sci. U.S.A. 93 (1996) 14532–14535.
[https://doi.org/10.1073/pnas.93.25.14532]

-
J.J. Vallooran, S. Handschin, S.M. Pillai, B.N. Vetter, S. Rusch, H.P. Beck, et al., Lipidic cubic phases as a versatile platform for the encapsulation and delivery of biocatalysts, Adv. Funct. Mater. 26 (2016) 627–645.
[https://doi.org/10.1002/adfm.201503428]

-
V. Luzzati, P.A. Spegt, Polymorphism of lipids, Nature 215 (1967) 701–704.
[https://doi.org/10.1038/215701a0]

-
R. Mezzenga, J M. Seddon, C.J. Drummond, B.J. Boyd, G.E. Schröder-Turk, L. Sagalowicz, Nature-inspired design and application of lipidic lyotropic liquid crystals, Adv. Mater. 31 (2019) 1900818.
[https://doi.org/10.1002/adma.201900818]

- J.N. Israelachvili, Intermolecular and Surface Forces, 3rd ed., Academic Press, San Diego, 2011.
- S. Hyde, S. Andersson, K. Larsson, The Language of Shape: The Role of Curvature in Condensed Matter, Elsevier, 1997.
-
A.M. Seddon, P. Curnow, P.J. Booth, Membrane proteins, lipids and detergents: Not just a soap opera, Biochim. Biophys. Acta 1666 (2004) 105–117.
[https://doi.org/10.1016/j.bbamem.2004.04.011]

- H.T. Tien, A. Ottova-Leitmannova, Planar Lipid Bilayers (BLM’s) and Their Applications, Elsevier, Amsterdam, 2003.
-
H. Qiu, M. Caffrey, The phase diagram of the monoolein/water system: Metastability and equilibrium aspects, Biomaterials 21 (2000) 223–234.
[https://doi.org/10.1016/S0142-9612(99)00126-X]

- A. Angelova, B. Angelov, S. Lesieur, R. Mutafchieva, R.P. O'Sullivan, Bicontinuous lipidic cubic phase nanostructures for bioactive molecule encapsulation and delivery, Curr. Opin. Colloid Interface Sci. 18 (2013) 464–475.
- E. Nazaruk, M. Sacha, R. Bilewicz, Enzymatic kinetics in liquid crystalline phases: coulo- and amperometric studies, Langmuir (2008) 12413–12419.
-
J. Barauskas, T. Landh, Phase behavior of the phytantriol/water system, Langmuir 19 (2003) 9562–9565.
[https://doi.org/10.1021/la0350812]

-
D.M. Anderson, S.M. Gruner, S. Leibler, Geometrical aspects of the frustration in the cubic phases of lyotropic liquid crystals, Proc. Natl. Acad. Sci. U.S.A. 85 (1988) 5364–5368.
[https://doi.org/10.1073/pnas.85.15.5364]

-
J. Briggs, H. Chung, M. Caffrey, The temperature–composition phase diagram and mesophase structure characterization of monoolein/water system, J. Phys. II 6 (1996) 723–751.
[https://doi.org/10.1051/jp2:1996208]

-
J. Barauskas, M. Johnsson, F. Tiberg, Self-assembled lipid superstructures: Beyond vesicles and liposomes, Nano Lett. 5 (2005) 1615–1619.
[https://doi.org/10.1021/nl050678i]

-
R. Negrini, R. Mezzenga, pH-responsive lyotropic liquid crystals for controlled drug delivery, Langmuir 27 (2011) 5296–5303.
[https://doi.org/10.1021/la200591u]

-
A.A. Brian, H.M. McConnell, Allogeneic stimulation of cytotoxic T cells by supported planar membranes, Proc. Natl. Acad. Sci. U.S.A. 81 (1984) 6159–6163.
[https://doi.org/10.1073/pnas.81.19.6159]

-
E. Sackmann, Supported membranes: Scientific and practical applications, Science 271 (1996) 43–48.
[https://doi.org/10.1126/science.271.5245.43]

-
E.T. Castellana, P.S. Cremer, Solid supported lipid bilayers: From biophysical studies to sensor design, Surf. Sci. Rep. 61 (2006) 429–444.
[https://doi.org/10.1016/j.surfrep.2006.06.001]

-
G. Lindblom, L. Rilfors, Cubic phases and isotropic structures formed by membrane lipids—possible biological relevance, Biochim. Biophys. Acta 988 (1989) 221–256.
[https://doi.org/10.1016/0304-4157(89)90020-8]

- J.Y.T. Chong, X. Mulet, L.J. Waddington, B.J. Boyd, C.J. Drummond, Steric stabilization of self-assembled cubic lyotropic liquid crystalline nanoparticles, Soft Matter 5 (2009) 3506–3519.
-
P. Mariani, V. Luzzati, H. Delacroix, Cubic phases of lipid-containing systems: Structure analysis and biological implications, J. Mol. Biol. 204 (1988) 165–189.
[https://doi.org/10.1016/0022-2836(88)90607-9]

-
J.M. Seddon, R.H. Templer, Polymorphism of lipid–water systems, in Structure and Dynamics of Membranes, In: R. Lipowsky, E. Sackmann (Eds.), Handbook of Biological Physics: Structure and Dynamics of Membranes – From Cells to Vesicles, Elsevier, Amsterdam, 1995, 97–160.
[https://doi.org/10.1016/S1383-8121(06)80020-5]

-
J.M. Seddon, Structure of the inverted hexagonal (HII) phase, and non-lamellar phase transitions of lipids, Biochim. Biophys. Acta 1031 (1990) 1–69.
[https://doi.org/10.1016/0304-4157(90)90002-T]

-
R. Koynova, M. Caffrey, Phases and phase transitions of the phosphatidylcholines, Biochim. Biophys. Acta 1376 (1998) 91–145.
[https://doi.org/10.1016/S0304-4157(98)00006-9]

-
J.N. Israelachvili, D.J. Mitchell, B.W. Ninham, Theory of self-assembly of hydrocarbon amphiphiles into micelles and bilayers, J. Chem. Soc., Faraday Trans. 2 72 (1976) 1525–1568.
[https://doi.org/10.1039/f29767201525]

-
H. Kim, Z. Song, C. Leal, Super-swelled lyotropic single crystals, Proc. Natl. Acad. Sci. U.S.A. 114 (2017) 10834–10839.
[https://doi.org/10.1073/pnas.1710774114]

-
G. Son, J. Song, J. Park, H.N. Kim, H. Kim, Fusogenic lipid nanoparticles for rapid delivery of large therapeutic molecules to exosomes, Nat. Commun. 16 (2025) 4799.
[https://doi.org/10.1038/s41467-025-59489-5]

-
W.K. Fong, T.L. Hanley, B.E. Thierry, T.W. Kee, B.J. Boyd, Photocontrol of nanostructure in a reversibly photoswitchable lipidic cubic phase, Langmuir 26 (2010) 6136–6139.
[https://doi.org/10.1021/la100644s]

-
H.M.G. Barriga, A.I.I. Tyler, N.J. Brooks, J.M. Seddon, Pressure–temperature phase behavior of swollen bicontinuous cubic membranes, Soft Matter 11 (2015) 600–607.
[https://doi.org/10.1039/C4SM02343A]

- R. Negrini, W.K. Fong, B.J. Boyd, R. Mezzenga, Tuning the phase behavior of lyotropic liquid crystals with silica nanoparticles, Langmuir 30 (2014) 3917–3920.
-
D. Sivadasan, M.H. Sultan, S.S. Alqahtani, S. Javed, Cubosomes in drug delivery—A comprehensive review on their structural components, preparation techniques and therapeutic applications, Biomedicines 11 (2023) 1114.
[https://doi.org/10.3390/biomedicines11041114]

-
W.K. Fong, B.E. Thierry, R.F. Tabor, T.L. Hanley, B.J. Boyd, Photothermal control of the structure of lipid-based liquid crystals using gold nanorods, Langmuir 28 (2012) 14450–14460.
[https://doi.org/10.1021/la302901q]

- C.V. Kulkarni, Nanostructural studies on monoolein-based stimuli-responsive lipidic liquid crystalline systems, Curr. Opin. Colloid Interface Sci. 16 (2011) 337–344.
-
B.E. Jones, E.A. Kelly, N. Cowieson, G. Divitini, R.C. Evans, Light-responsive molecular release from cubosomes using swell–squeeze lattice control, J. Am. Chem. Soc. 144 (2022) 19532–19541.
[https://doi.org/10.1021/jacs.2c08583]

-
H.M.G. Barriga, M.N. Holme, M.M. Stevens, Cubosomes: The next generation of smart lipid nanoparticles?, Angew. Chem. Int. Ed. 58 (2019) 2958–2978.
[https://doi.org/10.1002/anie.201804067]

- S.S. Razola, P. Wadsten-Hindrichsen, A. Nylander, A.M. Johansson, A.E.G. Persson, R. Bilewicz, Microperoxidase-11 in cubic phases: structure and electrochemistry, Langmuir 18 (2002) 7112–7116.
-
W. Liu, S.E. Lewis, M. Di Lorenzo, A.M. Squires, Development of redox-active lyotropic lipid cubic phases for biosensing platforms, Langmuir 40 (2023) 170–178.
[https://doi.org/10.1021/acs.langmuir.3c02307]

- R. Bilewicz, E. Nazaruk, Recent developments in the use of lipid cubic phases for bioelectrochemical applications, Electroanalysis 25 (2013) 1339–1350.
- E. Nazaruk, B. Lindholm-Sethson, R. Bilewicz, Liquid crystalline cubic phase as a matrix for direct electron transfer of cellobiose dehydrogenase, Electrochem. Commun. 11 (2009) 194–197.
-
G. Valincius, T. Meškauskas, F. Ivanauskas, Electrochemical impedance spectroscopy of tethered bilayer membranes, Langmuir 28 (2012) 977–990.
[https://doi.org/10.1021/la204054g]

-
J.R. Windmiller, J. Wang, Wearable electrochemical sensors and biosensors: A review, Electroanalysis 25 (2013) 29–46.
[https://doi.org/10.1002/elan.201200349]

-
Y.H. Lee, M. Jang, M.Y. Lee, O.Y. Kweon, J.H. Oh, Flexible Field-Effect Transistor-Type Sensors Based on Conjugated Molecules, Chem 3 (2017) 724–763.
[https://doi.org/10.1016/j.chempr.2017.10.005]

-
Y.H. Lee, O.Y. Kweon, H. Kim, J.H. Yoo, S.G. Han, J.H. Oh, Recent advances in organic sensors for health self-monitoring systems, J. Mater. Chem. C 6 (2018) 8569–8612.
[https://doi.org/10.1039/C8TC02230E]

-
J.P. Lagerwall, G. Scalia, A new era for liquid crystal research: Applications of liquid crystals in soft matter nano-, bio- and microtechnology, Curr. Appl. Phys. 12 (2012) 1387–1412.
[https://doi.org/10.1016/j.cap.2012.03.019]

-
R.J. Carlton, J.T. Hunter, D.S. Miller, R. Abbasi, P.C. Mushenheim, L.N. Tan, et al., Chemical and biological sensing using liquid crystals, Langmuir 1 (2013) 29–51.
[https://doi.org/10.1080/21680396.2013.769310]

- P. Sarkar, A.M. Squires, A.J.M. Valner, Modulation of FRET in lipidic cubic phases, J. Phys. Chem. B 121 (2017) 534–540.
- S.T. Hyde, S. Andersson, K. Larsson, Z. Blum, T. Landh, S. Lidin, et al., The Language of Shap e: The Role of Geometry in Physical and Biological Systems, Elsevier, Amsterdam, 1997.
- J. Barauskas, C. Cervin, M. Jankunec, M. Spandyreva, K. Ribokaite, F. Tiberg, et al., Interactions of cholera toxin B-subunit with GM1-decorated lipid nanotubes and cubic phases, Nano Lett. 10 (2010) 4516–4522.
- M. Tylman, J. Barauskas, F. Tiberg, T. Nylander, Cholera toxin binding to GM1-containing lipid membranes, J. Pharm. Biomed. Anal. 55 (2011) 301–306.
- J.P. Skinner, S.J. Woltman, G.P. Crawford, Liquid crystal droplets for biosensing, Langmuir 26 (2010) 2196–2201.
- T. Turiv, I. Lazo, O.D. Lavrentovich, Effect of bacterial motion on chromonic liquid crystal droplets, Soft Matter 9 (2013) 5136–5141.
-
I.-H. Lin, D.S. Miller, P.J. Bertics, C.J. Murphy, J.J. de Pablo, N.L. Abbott, Endotoxin-induced structural transformations in liquid crystalline droplets, Science 332 (2011) 1297–1300.
[https://doi.org/10.1126/science.1195639]

- S. Kolusheva, R. Jelinek, Colorimetric liposome bioassays: no longer "in the red", Drug Discov. Today 7 (2002) 216–217.
-
A. Koh, D. Kang, Y. Xue, S. Lee, R.M. Pielak, J. Kim, et al., A soft, wearable microfluidic device for the capture, storage, and colorimetric sensing of sweat, Sci. Transl. Med. 8 (2016) 366ra165.
[https://doi.org/10.1126/scitranslmed.aaf2593]

-
C.E. Stanley, E. Fodor, I. Grotjohann, T. Gonen, R.H. Templer, O. Ces, A microfluidic platform for scaling lipidic cubic phases, Chem. Commun. 46 (2010) 1620–1622.
[https://doi.org/10.1039/b924897h]

-
E. Katz, I. Willner, Integrated nanoparticle-biomolecule hybrid systems: synthesis, properties, and applications, Angew. Chem. Int. Ed. 43 (2004) 6042–6108.
[https://doi.org/10.1002/anie.200400651]

- N.M. Soolaman, M.A. Peterson, S.R. Niezgoda, T.M. Ferreira, H. Yu, Lipid-DNA assembly for hybridization sensing, J. Phys. Chem. B 122 (2018) 2894–2902.
-
M. Marsh, A. Helenius, Virus entry: open sesame, Cell 124 (2006) 729–740.
[https://doi.org/10.1016/j.cell.2006.02.007]

-
B. Tsai, J.M. Gilbert, T. Stehle, W. Lencer, T.L. Benjamin, T.A. Rapoport, Gangliosides are receptors for murine polyoma virus and SV40, EMBO J. 22 (2003) 4346–4355.
[https://doi.org/10.1093/emboj/cdg439]

-
S. Bolisetty, R. Mezzenga, Amyloid-carbon hybrid membranes for universal water purification, Nat. Nanotechnol. 11 (2016) 365–371.
[https://doi.org/10.1038/nnano.2015.310]

-
M. Porras-Gomez, C. Leal, Lipid-based liquid crystalline films and solutions for the delivery of cargo to cells, Liq. Cryst. Rev. 7 (2019) 167–182.
[https://doi.org/10.1080/21680396.2019.1666752]


Hojun Kim is a Senior Researcher at the Korea Institute of Science and Technology (KIST), where he leads research on lipid membrane engineering for biosensors and gene therapy. He received his B.S. degree in Materials Science and Engineering from KAIST in 2012 and his Ph.D. in Materials Science and Engineering from the University of Illinois at Urbana-Champaign in 2017, under the supervision of Prof. Cecilia Leal. He subsequently conducted postdoctoral research at UIUC before joining KIST in 2018 as a Research Scientist, and has served as Senior Researcher since 2020. His research interests span vesicle transport in extracellular matrices, super-swelling lyotropic phases, mRNA carriers, onsite colloidal biosensors for infectious disease screening, and transdermal delivery systems. He has received multiple awards, including the Racheff-Intel Award (2017) and the PSK-Innox Young Investigator Award (2022).

