
Influence of Alumina Particles Addition in an Alkaline Silicate-Aluminate Electrolyte on Corrosion Properties of Plasma Electrolytic Oxidation Coatings on AZ31B Mg Alloy
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
This study investigates the electrochemical corrosion resistance of plasma electrolytic oxidation (PEO) coatings on AZ31B Mg alloy, focusing on the influence of Al2O3 particles addition into an alkaline silicate-aluminate electrolyte. PEO coatings were fabricated with varying Al2O3 concentrations from 0 to 10 g/L in the electrolyte and analyzed for thickness, surface morphology, and porosity. Electrochemical corrosion behavior was assessed via potentiodynamic polarization and electrochemical impedance spectroscopy (EIS). Although the thickness of PEO coatings increased with increasing Al2O3 concentration in the electrolyte, optimal corrosion resistance was achieved at 4 g/L of Al2O3, originating in the lowest porosity. This concentration yielded the highest corrosion potential and lowest corrosion current density, resulting in highest polarization resistance. EIS results corroborated these findings, confirming superior corrosion protection by PEO coating at the Al2O3 concentration of 4 g/L owing to a denser, more protective oxide layer.
Keywords:
Al2O3 particles addition, Plasma electrolytic oxidation, Corrosion resistance, AZ31B Mg alloy1. INTRODUCTION
Magnesium (Mg) alloys are increasingly used in industries where lightweight materials are required, such as the automotive, aerospace, and biomedical fields, owing to their low density and high strength-to-weight ratio [1]. Among commercial Mg alloys, the AZ31B Mg alloy is one of the best candidates for electronic and sensor materials because of its excellent formability and workability, remarkable mechanical properties, and good thermal and electrical conductivity [2,3]. However, a major limitation of Mg alloys is their poor corrosion resistance, which hinders their broader application, particularly in aggressive environments [4,5]. To overcome this challenge, researchers have focused on developing surface modification techniques to enhance the corrosion resistance of Mg alloys [1,6].
Plasma electrolytic oxidation (PEO) has emerged as an effective method for enhancing the surface properties of Mg alloys. PEO forms an oxide layer on the substrate surface through an electrochemical process under high voltage, leading to a thicker oxide layer that offers improved corrosion resistance, mechanical strength, and wear [7-10]. Oxide coatings formed in the PEO processes typically consist of magnesium oxide (MgO) and magnesium hydroxide (Mg(OH)2), which contribute to the enhanced corrosion resistance of the alloy [7-11].
In recent studies, the addition of ceramic particles, including alumina (Al2O3), to PEO electrolytes has been shown to further improve the performance of the oxide films. The incorporation of Al2O3, a stable and dense material, into the oxide layer has been found to significantly boost the corrosion resistance of Mg alloys [12,13]. Moreover, electrolytes based on silicate and aluminate have attracted attention because of their ability to generate more durable and protective oxide coatings. The synergy between these components and embedded ceramic particles further enhances the stability of the oxide layer, making it more effective in preventing corrosion [14,15].
In this study, oxide coatings were fabricated on an AZ31B Mg alloy using the PEO method in an alkaline silicate-aluminate electrolyte containing Al2O3 particles as an additive. The effect of Al2O3 particles concentration on the corrosion behavior of Mg alloys was systematically investigated. The surface morphology and porosity of the coatings were analyzed. The corrosion resistance was evaluated using potentiodynamic polarization tests and electrochemical impedance spectroscopy (EIS), with a focus on applications in packaging and as reference-sensing elements in Mg-based corrosion sensors.
2. EXPERIMENTAL
A commercially available AZ31B Mg alloy (Al 2.5–3.5 wt%, Zn 0.6–1.4 wt%, Mn 0.2–1.0 wt%, Mg balance) was used as substrate. Prior to the experiments, AZ31B Mg alloy plates with a size of 25 × 25 × 1 mm3 were mechanically polished using #600, #1000 and #1500 SiC abrasive papers. To eliminate surface contaminants, the polished substrates were ultrasonically cleaned in acetone, followed by rinsing with distilled water. The alkaline silicate-aluminate solution composed of 1 g/L NaOH, 4 g/L NaAlO2, and 12 g/L Na2SiO3·9H2O in distilled water was used as the base electrolyte. The electrolyte for the PEO process was prepared from the base electrolyte with addition of 0, 2, 4, 6, 8, and 10 g/L Al2O3 particles (ca. ~ 1 μm). A direct current (DC) constant voltage mode was employed for the PEO treatment. The details of experiments are largely similar to those in the literature [1,16]. The PEO treatment was performed for 30 s with a constant DC voltage of 270 V.
Surface analysis was conducted using a field emission scanning electron microscopy (FE-SEM, Hitachi S4200). The FE-SEM facilitated the investigation of both the surface and cross-sectional morphologies of PEO coatings. Surface porosity was quantified from surface morphological data via an image analysis software. The PEO coating thickness was obtained from cross-sectional FE-SEM images. An energy-dispersive X-ray spectrometer (EDS, Horiba Inc.) was used to investigate the chemical composition of PEO coatings on the AZ31B Mg alloy.
All electrochemical experiments were conducted at room temperature using a conventional three-electrode system cell. The PEO-coated AZ31B Mg alloy with an exposed area of 1 cm2 was used as the working electrode. A platinum (Pt) plate and saturated silver/silver chloride (Ag/AgCl) were used as the counter and reference electrodes, respectively. Using the above experimental materials and instruments, potentiodynamic polarization and EIS tests were performed to analyze the corrosion resistance in Ringer’s solution [17]. The polarization curve representing the corrosion potential and corrosion current density values was obtained by forcibly corroding the PEO-coated alloy using the three-electrode measurement method. This curve was analyzed using the Tafel extrapolation method to obtain the polarization resistance and anode/cathode Tafel values [18]. The EIS test is a method for analyzing impedance by applying a sinusoidal signal over a broad frequency range (from high to low) to a test specimen and measuring the amplitude and phase shift in the resulting response. A Nyquist plot was obtained from the measurement results [19].
3. RESULTS AND DISCUSSIONS
Fig. 1 shows the variation in thickness of the PEO coating layer on the AZ31B Mg alloy with respect to the Al2O3 particle concentration in the alkaline silicate-aluminate electrolyte. The inserted figures show the cross-sectional FE-SEM images of the PEO coating layer for Al2O3 particles concentrations of 0 and 10 g/L. The thickness of the PEO coating layer increased from 0.744 to 1.84 μm as Al2O3 particles content increased from 0 to 10 g/L. From the EDS examination, PEO coatings prepared with 0, 2, 4, 6, 8, and 10 g/L Al2O3 particles added to the electrolyte revealed Al contents of 4.73, 4.87, 4.91, 4.98, 5.04, and 5.09 at%, respectively. Without Al2O3 particles addition, the Al concentration of 4.73 at% in the PEO coating resulted from the presence of NaAlO2 in the base electrolyte. The concentration of Al in the PEO coating increased with increasing Al2O3 particles content in the electrolyte, indicating that the Al2O3 particles were successfully incorporated into the PEO coating. When oxide particles were added and dispersed in aqueous solutions, they could not be dissolved in the solution but were negatively charged on the surface [20]. Thus, the negatively surface-charged Al2O3 particles could move toward the AZ31B substrate, an anodic electrode, under a DC bias. Under plasma arcs, the melting of Al2O3 particles close to the substrate and the fluctuating molten oxide on the substrate surface might occur, giving rise to the reaction of Al2O3 particles with the surface of the substrate. Therefore, an increase in the concentration of Al2O3 particles in the electrolyte enhanced the reaction near the substrate, resulting in an increase in the thickness of the PEO coating layer.
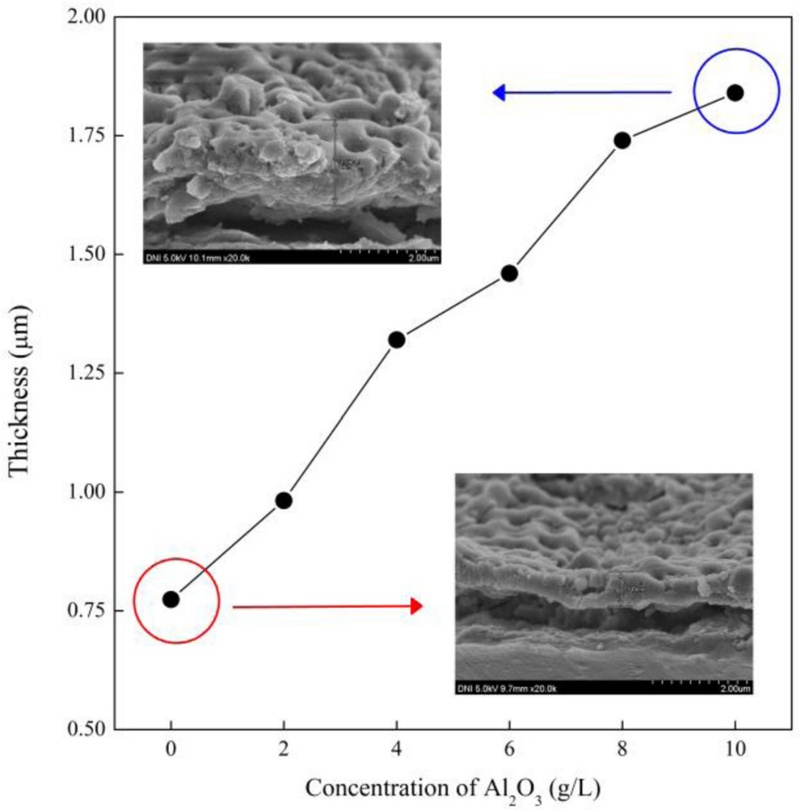
Thickness variation of PEO coating layer on AZ31B Mg alloy with respect to Al2O3 particles concentration in the electrolyte. The inset is the cross-sectional FE-SEM images for PEO coating layer without and with the addition of Al2O3 particles.
The morphology of PEO-coated surfaces fabricated with varying Al2O3 concentrations from 0 to 10 g/L were investigated using FE-SEM images, as shown in Fig. 2. Quantitatively, the porosity of PEO coatings at Al2O3 concentrations of 0, 2, 4, 6, 8, and 10 g/L were 8.348, 7.128, 6.070, 7.626, 8.624, and 10.613%, respectively. It was evident that as the Al2O3 concentration in the electrolyte increased from 0 to 4 g/L, the porosity decreased. The lowest porosity was observed in samples with 4 g/L of additive, with porosity beginning to increase at 6 g/L. During plasma arc generation, the substrate surface, native oxide layer, and previously formed oxide film became ionized. These ionized species then combined with oxygen ions from the electrolyte and other ions of the additive components, leading to the formation of pores at the ionized sites and the filling of other pores. This continuous process of oxide film formation and regeneration resulted in an increase in thickness. For a sample containing at 4 g/L of Al2O3, this process yielded a surface with the lowest porosity. Beyond this concentration, the additional additive caused the oxide film to form with increasing porosity.
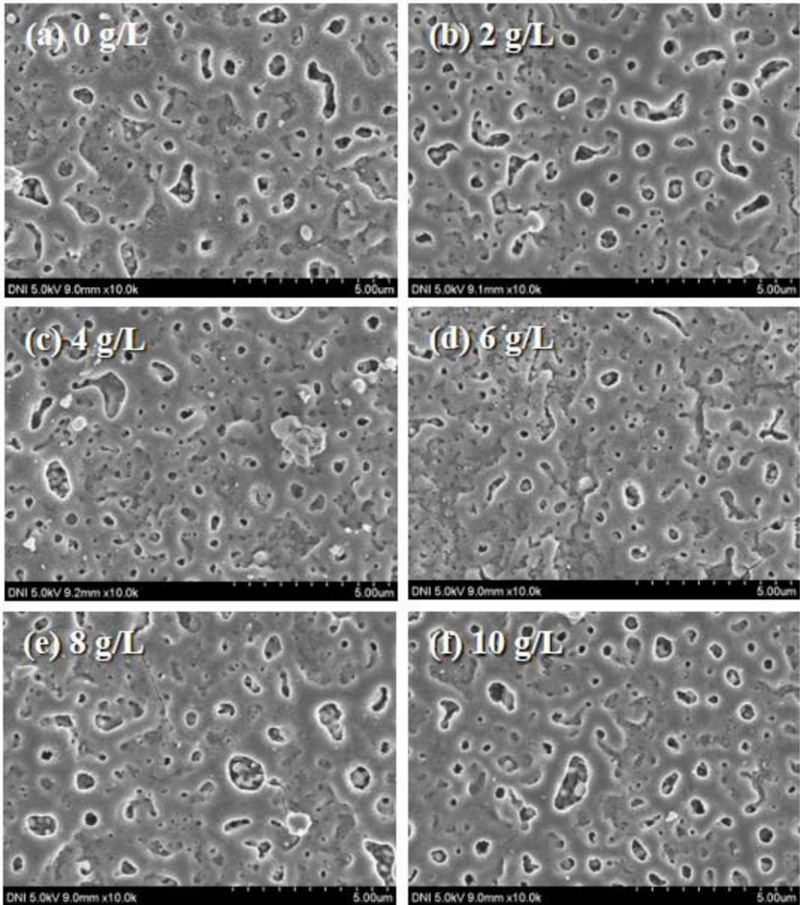
FE-SEM images of the PEO-coated surfaces with (a) 0 g/L, (b) 2 g/L, (c) 4 g/L, (d) 6 g/L, (e) 8 g/L, and (f) 10 g/L of Al2O3 concentration.
Polarization curves were obtained for PEO-coated AZ31B Mg alloy specimens by applying forced corrosion using a conventional three-electrode electrochemical cell. These curves provided the corrosion potential (Ecorr) and corrosion current density (Icorr) values, which were subsequently analyzed using the Tafel extrapolation method to determine the polarization resistance (Rp) and anodic and cathodic Tafel slopes (βa and βc). Fig. 3 shows the potentiodynamic polarization curves of PEO coatings immersed in Ringer’s solution. Corrosion-related parameters, including Ecorr, Icorr, and Tafel constants (βa and βc), were derived from these curves via Tafel extrapolation. The corresponding values are summarized in Table 1. The Rp was calculated using the Stern-Geary equation [21].

The potentiodynamic polarization parameters of the PEO-coated AZ31B Mg alloy within electrolytes containing different concentrations of Al2O3 particles.
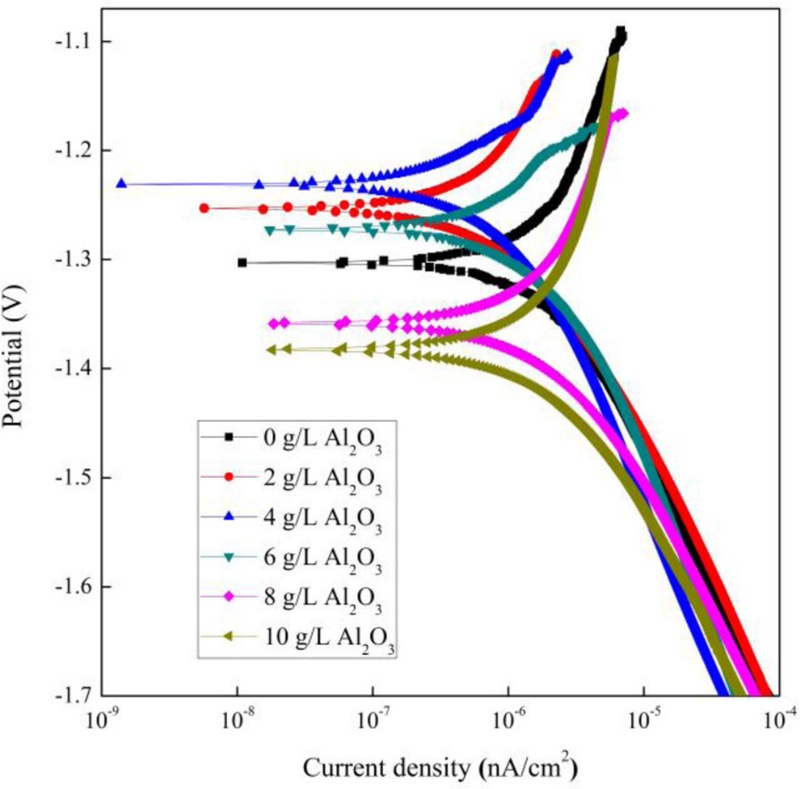
Potentiodynamic polarization curve of the PEO-coated AZ31B Mg alloy within electrolytes containing various Al2O3 particles concentrations.
| (1) |
Among the samples, the specimen with 4 g/L of Al2O3 exhibited the most favorable corrosion resistance. A progressive enhancement in corrosion resistance was observed with increasing Al2O3 concentration up to 4 g/L; however, a decline was noted at 6 g/L. Tafel analysis further confirmed that the 4 g/L Al2O3 sample exhibited the highest Ecorr and Rp values among all tested conditions. Specifically, the sample with 4 g/L Al2O3 showed an Ecorr of −1.2219 V, indicating highest corrosion potential compared to the other specimens. This improvement was attributed to the reduced porosity of the oxide layer, which minimized Mg dissolution. In addition, the Icorr value for this sample was 3.158 × 10-8 A/cm2, the lowest among the group, suggesting that the PEO coating effectively insulated the substrate from the corrosive environment. The corresponding Rp was measured at 7.72 × 105 Ω, the highest value among the specimens, indicating superior corrosion protection provided by the PEO coating with 4 g/L of Al2O3 [22].
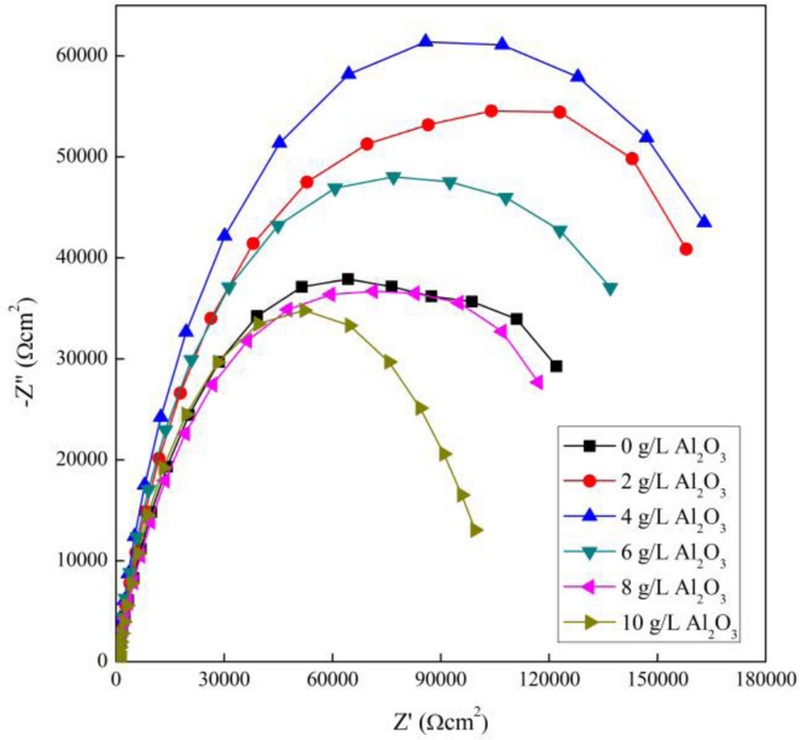
Nyquist plots of the PEO-coated AZ31B Mg alloy within electrolytes containing various Al2O3 particles concentrations.
EIS was used to analyze impedance by measuring changes in amplitude and phase in response to a sinusoidal signal applied over a wide frequency range, typically from high to low frequencies. Nyquist plots were obtained as a result of the EIS measurements with various concentrations of Al2O3. In the Nyquist plot, the charge-transfer resistance, represented by the diameter of the semicircle in the high- to mid-frequency range, was highest for the sample containing 4 g/L of Al2O3. In the high-frequency region, these plots exhibited depressed semicircles, indicative of capacitive behavior. The radius of these semicircles was directly correlated with corrosion resistance; a larger radius corresponds to enhanced corrosion protection [22]. Notably, the PEO coating formed with 4 g/L of Al2O3 exhibited the largest semicircle radius among all tested concentrations, indicating superior corrosion resistance.
4. CONCLUSIONS
This study demonstrated that the incorporation of Al2O3 particles into PEO electrolyte enhanced the corrosion resistance of AZ31B Mg alloy. Comprehensive analyses of the coating morphology, porosity, and electrochemical behavior revealed that an Al2O3 concentration of 4 g/L yielded the optimal performance. At this concentration, the PEO coating exhibited the lowest porosity, indicating a denser and more uniform protective layer. Electrochemical evaluations, including potentiodynamic polarization and EIS, consistently confirmed the superior corrosion resistance of the PEO coating with 4 g/L Al2O3 coating. This was evidenced by a more noble corrosion potential, reduced corrosion current density, and significantly increased polarization and charge-transfer resistances. The enhanced protective performance is attributed to the synergistic effect of the decreased porosity and stable incorporation of Al2O3 particles within the oxide matrix, which collectively insulated the substrate from the corrosive environment. These findings provide valuable guidance for the design of corrosion-resistant Mg alloys and highlight the potential of customized PEO treatments for advanced surface engineering applications. Nevertheless, for practical device applications, further investigations on the corrosion behavior of PEO coatings with reactively incorporated Al2O3 particles on AZ31B Mg alloys in aggressive environments are needed from a reliability point of view.
Acknowledgments
This work was supported by research grants from Daegu Catholic University in 2025.
References
-
D.K. Lee, Y.-S. Sohn, Electrochemical Corrosion Properties of Plasma Electrolytic Oxidation Coatings on AZ31 Mg Alloy in Sodium Silicate-based Electrolyte Containing NaAlO2, J. Sens. Sci. Technol. 33 (2024) 453–457.
[https://doi.org/10.46670/JSST.2024.33.6.453]

-
J. Tan, S. Ramakrishna, Applications of magnesium and its alloys: a review, Appl. Sci. 11 (2021) 6861.
[https://doi.org/10.3390/app11156861]

-
S. Wang, Z. Yan, H. Zhang, X. He, P. Dong, W. Wang, et al., Acoustic and thermal energy evolution of AZ31B magnesium alloy under static tensile deformation, J. Mater. Res. Technol. 20 (2022) 1645–1658.
[https://doi.org/10.1016/j.jmrt.2022.07.169]

-
J. Zhang, S. Hou, M. Zhang, S. Zhang, W. Li, Corrosion resistance and biocompatibility of silica coatings on AZ31 magnesium alloy via magnetron sputtering, Mater. Today Commun. 41 (2024) 110890.
[https://doi.org/10.1016/j.mtcomm.2024.110890]

-
L. Yan, G.-L. Song, P. Wu, Y. Zhu, D. Zheng, A chloride-sensitive corrosion sensor and protector made of an optimized Mg-Al alloy for reinforcing steel, Mater. Des. 210 (2021) 110028.
[https://doi.org/10.1016/j.matdes.2021.110028]

-
J. Kloiber, V. Anetsberger, U. Schultheiß, H. Hornberger, High quality surfaces of magnesium alloy AZ31 by adjusting appropriate electropolishing parameters, Electrochim. Acta 513 (2025) 145547.
[https://doi.org/10.1016/j.electacta.2024.145547]

-
H. Fan, K. Liang, R. Bai, C. Wang, J. Zhang, L. Wang, Enhanced corrosion resistance of PEO coating on AZ31B Mg alloys with delayed corrosion and slow diffusion, Surf. Coat. Technol. 496 (2025) 131630.
[https://doi.org/10.1016/j.surfcoat.2024.131630]

-
V. Khalili, H. Ghaleh, H.N. Asl, D. Ege, B. Dikici, M. Kaseem, et al., Assessing the properties of biodegradable magnesium alloy AZ31 protected by a polymer layer on a plasma electrolytic oxidized (PEO) surface, Surf. Coat. Technol. 487 (2024) 131002.
[https://doi.org/10.1016/j.surfcoat.2024.131002]

-
R.O. Hussein, D.O. Northwood, X. Nie, The effect of processing parameters and substrate composition on the corrosion resistance of plasma electrolytic oxidation (PEO) coated magnesium alloys, Surf. Coat. Technol. 237 (2013) 357–368.
[https://doi.org/10.1016/j.surfcoat.2013.09.021]

-
R. Ji, S. Wang, X. Zhao, Y. Zou, T. Zhang, X. Qian, et al., Enhanced wear resistance, corrosion behavior, and thermal management in magnesium alloys with PEO coatings, Surf. Coat. Technol. 494 (2024) 131438.
[https://doi.org/10.1016/j.surfcoat.2024.131438]

-
D. Zhang, J. Tan, H. Du, S. Qian, X. Liu, Comparison study of Mg (OH)2, Mg-Fe LDH, and FeOOH coatings on PEO-treated Mg alloy in anticorrosion and biocompatibility, Appl. Clay Sci. 225 (2022) 106535.
[https://doi.org/10.1016/j.clay.2022.106535]

-
E. Lopez-Martinez, M. Mohedano, E. Matykina, R. Arrabal, PEO of additively manufactured Al10SiMg alloy: Effects of α-Al2O3 particles on energy consumption and wear behaviour, J. Mater. Res. Technol. 31 (2024) 1786–1976.
[https://doi.org/10.1016/j.jmrt.2024.06.171]

-
Y. Li, C. Li, Q. Wang, X. Zhao, T. Zhang, F. Wang, Enhancing wear and corrosion resistance of magnesium alloys through in-situ Al2O3 doping for plasma electrolytic oxidation coating, Mater. Today Commun. 38 (2024) 108234.
[https://doi.org/10.1016/j.mtcomm.2024.108234]

-
T. Wu, C. Blawert, M. Serdechnova, P. Karlova, G. Dovzhenko, D.C.F. Wieland, et al., Role of phosphate, silicate and aluminate in the electrolytes on PEO coating formation and properties of coated Ti6Al4V alloy, Appl. Surf. Sci. 595 (2022) 153523.
[https://doi.org/10.1016/j.apsusc.2022.153523]

-
C. Yang, P. Chen, W. Wu, L. Sheng, Y. Zheng, P.K. Chu, A Review of Corrosion-Resistant PEO Coating on Mg Alloy, Coatings 14 (2024) 451.
[https://doi.org/10.3390/coatings14040451]

-
K.D. Moon, D.K. Lee, Effects of the sodium borate additions in NaOH-Na2SiO3 electrolytes on the surface and corrosion properties of plasma electrolytic oxidation coatings on AZ31 Mg alloy, Mol. Cryst. Liq. Cryst. 735 (2022) 2–8.
[https://doi.org/10.1080/15421406.2021.1972221]

-
M. Jamesh, S. Kumar, T.S.N. Sankara Narayanan, Corrosion behavior of commercially pure Mg and ZM21 Mg alloy in Ringer’s solution – Long term evaluation by EIS, Corros. Sci. 53 (2011) 645–654.
[https://doi.org/10.1016/j.corsci.2010.10.011]

-
X.L. Zhang, Z.H. Jiang, Z.P. Yao, Y. Song, Z.D. Wu, Effects of scan rate on the potentiodynamic polarization curve obtained to determine the Tafel slopes and corrosion current density, Corros. Sci. 51 (2009) 581–587.
[https://doi.org/10.1016/j.corsci.2008.12.005]

-
Z. Shahri, S.R. Allahkaram, R. Soltani, H. Jafari, Electrochemical analysis on localized corrosion of PEO/magnesium oxide coating, J. Alloys Compd. 1003 (2024) 175572.
[https://doi.org/10.1016/j.jallcom.2024.175572]

-
K.M. Lee, K.R. Shin, S. Namgung, B. Yoo, D.H. Shin, Electrochemical response of ZrO2-incorporated oxide layer on AZ91 Mg alloy processed by plasma electrolytic oxidation, Surf. Coat. Technol. 205 (2011) 3779–3784.
[https://doi.org/10.1016/j.surfcoat.2011.01.033]

-
M. Stern, A.L. Geary, Electrochemical polarization: I. A theoretical analysis of the shape of polarization curves, J. Electrochem. Soc. 104 (1957) 56–63.
[https://doi.org/10.1149/1.2428496]

-
A. Rudawska, E. Jacniacka, Analysis for determining surface free energy uncertainty by the Owen–Wendt method, Int. J. Adhes. Adhes. 29 (2009) 451–457.
[https://doi.org/10.1016/j.ijadhadh.2008.09.008]

