The Role of Two-Dimensional Materials in Optical Chemical Sensing
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Two-dimensional (2D) materials, which are characterized by atomic-scale thickness and tunable electronic and optical properties, have emerged as promising components in optical chemical sensing technologies. Their large surface-to-volume ratio and unique photonic behaviors enable enhanced light–matter interactions, thereby significantly improving sensor sensitivity, selectivity, and stability. This review provides an overview of recent developments in the integration of 2D materials with key plasmonic sensing platforms, including surface plasmon resonance (SPR), localized SPR (LSPR), and surface-enhanced Raman scattering. Challenges inherent to each sensing modality are discussed, alongside how 2D materials address these issues through heterostructure assembly, surface functionalization, and hybrid nanostructure design. Recent advances in scalable synthesis and fabrication methods, especially solution-based processing, offer promising routes to improve compatibility with diverse substrates and device architectures. We highlight such fabrication strategies as potential enablers for flexible, scalable, and multifunctional sensor designs, thereby suggesting pathways for the translation of 2D materials from laboratory studies into practical chemical sensing applications.
Keywords:
Optical chemical sensing, Plasmonic sensor, Two-dimensional material, Chemical detection1. INTRODUCTION
Chemical sensors are essential tools for detecting specific substances in many areas, such as environmental monitoring [1,2], healthcare [3,4], industrial control [5], and security [6]. Their performance largely depends on their ability to balance sensitivity, selectivity, and speed, which are critical for real-world applications. As modern industries become more complex and concerns regarding the environment and public health increase, the demand for reliable, efficient, and easy-to-use chemical sensors has increased significantly. Chemical sensors operate by detecting changes in physical or chemical properties such as shifts in electrical resistance, light intensity, or chemical interactions with target analytes [7]. Recent studies have shown significant progress in the development of chemical sensors using two complementary strategies. One involves structural engineering approaches aimed at maximizing the active surface area to enhance sensor reactivity and sensitivity [8,9]. The second strategy emphasizes the integration of novel materials such as biopolymers and two-dimensional (2D) structures to boost performance and support miniaturization [10]. These strategies have been effectively demonstrated in recent reviews on nanofiber-based gas sensors and 2D material-integrated sensing platforms [11,12].
Traditional chemical sensors are primarily designed for detecting individual analytes, which limits their applicability in identifying complex mixtures and has led to the development of electronic nose (e-nose) systems [13,14]. These systems integrate arrays of broadly responsive sensors with sophisticated pattern recognition algorithms, enabling them to mimic the human sense of smell. E-noses do not identify one molecule at a time, but rather interpret the overall chemical patterns formed by mixtures. This unique capability enables e-noses to generate chemical "fingerprints" that represent complex mixtures [15]. Such approaches are useful for diverse applications, including pollution monitoring [16], food quality assessment [17], and disease diagnosis [18]. The broad responsiveness to volatile organic compounds (VOCs) and gases has made e-noses increasingly important tools for environmental protection and public health.
As shown in Fig. 1, chemical sensors can be classified into three main categories based on their sensing mechanisms: 1) chemiresistive sensors, 2) bio-based sensors, and 3) nanoplasmonic sensors. Each category presents unique strengths and challenges that affect its effectiveness and suitability for various real-world applications.
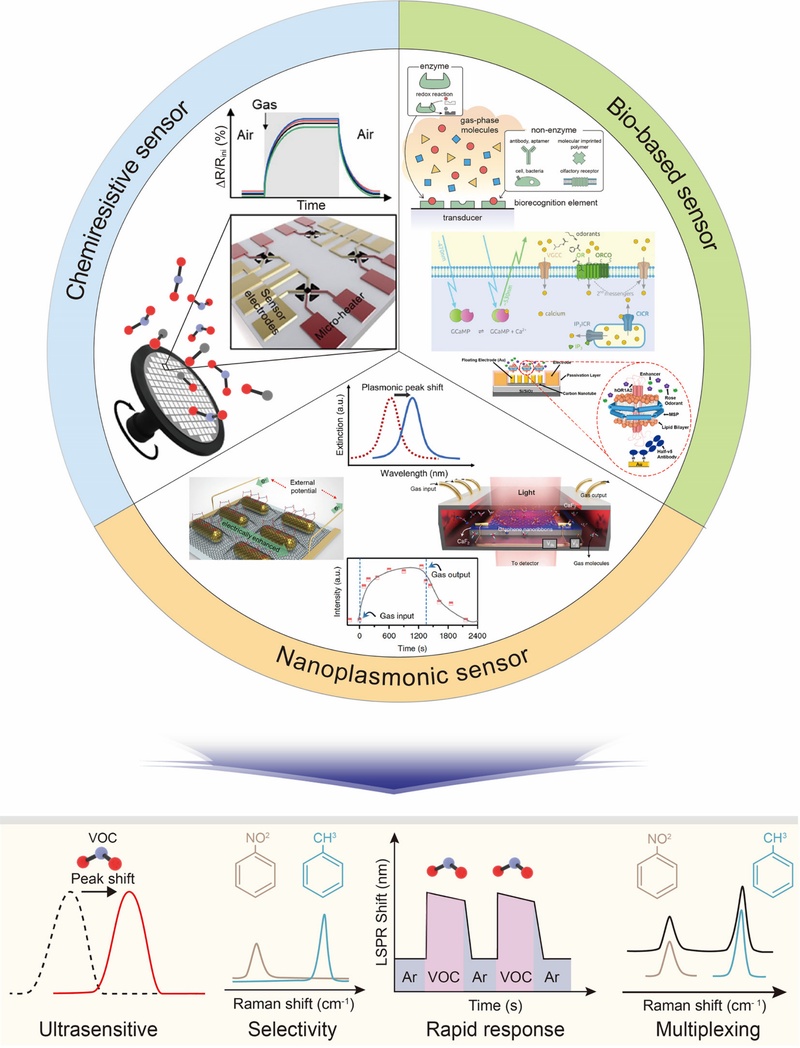
Introduction to chemical sensors and the advantages of nanoplasmonic sensors. Various chemical sensing platforms, including chemiresistive, bio-based, and nanoplasmonic sensor, are presented to highlight their distinct detection mechanisms and performance advantages for environmental pollutants and health-related biomarkers. Reprinted with permission from Refs. [20] and [27] Copyrights (2022, 2023) American Chemical Society; and Refs. [15], [21], and [22], Copyright (2019, 2014, 2018) Springer Nature.
Chemiresistive sensors, which are mostly based on semiconductor metal oxides, detect variations in electrical resistance caused by the modulation of the electron depletion layer when analyte molecules are adsorbed or desorbed. Chemiresistive sensors are highly suitable for both portable and large-scale applications owing to their small size, low production cost, and compatibility with scalable manufacturing methods such as screen printing [19,20]. However, their broad sensitivity to diverse chemical species often limits their selectivity. Methods including incorporating catalytic materials or selective filters have been proposed to address this issue, although such modifications may reduce the response speed and overall sensitivity [20]. In contrast, biosensors utilize biological recognition elements, including enzymes, antibodies, and whole cells, to achieve molecular specificity through selective binding [21,22]. Although bio-based systems were originally designed for liquid-phase analytes, technological advances have expanded their applicability to gas-phase sensing. For example, enzyme-based biosensors can selectively detect ethanol, which is a typical VOC [23]. The hydrated recognition layer helps to reduce humidity interference and ensures stable performance [23,24]. Furthermore, the spatial and temporal mapping of VOC emissions using gas-phase biosensors has demonstrated the potential for noninvasive health monitoring [24]. Despite these advantages, the practical implementation of bio-based sensors is limited by the inherent fragility of biological components that are susceptible to degradation under environmental stress, including temperature fluctuations, pH shifts, and organic solvents [25,26]. Given the persistent limitations of selectivity and stability observed in both chemiresistive and bio-based sensors, researchers have focused increasingly on nanoplasmonic sensors as promising alternatives owing to their enhanced surface sensitivity and signal amplification [25].
Nanoplasmonic sensors, including surface plasmon resonance (SPR), localized SPR (LSPR), and surface-enhanced Raman scattering (SERS), have drawn considerable attention because of their high sensitivity and ability to detect chemical species at the nanoscale. SPR sensors detect variations in the refractive index that occur when light interacts with a thin metallic surface, typically made of gold or silver [27-29]. SPR is well suited for real-time monitoring, particularly for liquid or gas-phase samples, making it a powerful tool for biosensing and environmental monitoring [30]. However, the SPR configuration has some limitations. The sensing setup requires precise optical alignment and bulky components, which complicate miniaturization [31]. In addition, SPR systems are highly sensitive to changes in the bulk refractive index, making the measurements vulnerable to environmental fluctuations such as temperature or pressure variations [32]. Moreover, the relatively long decay length of the evanescent field (typically 100–400 nm) limits the detection of fine-scale molecular events that occur close to the surface [33]. Conformational transitions and minor interactions between biomolecules often fall below the sensitivity threshold because the signal is averaged across the larger sensing volume [33]. In recent studies, alternative approaches have been actively pursued to overcome such limitations, with particular emphasis on enhancing the sensitivity and enabling sensor miniaturization by integrating optical-fiber technology [32,34].
Compared with SPR, LSPR sensors utilize metal nanostructures (e.g., nanoparticles, nanorods, hollow nanospheres, and nanodisks) that interact with light at the nanoscale. These interactions generate a localized electromagnetic field with a much shorter decay length (typically 10–30 nm) [35], making LSPR more sensitive to surface-bound analytes and less responsive to changes in the bulk refractive index. Consequently, LSPR devices provide improved measurement stability and are less affected by environmental fluctuations, enabling the detection of very small amounts of analytes, often at the parts per billion (ppb) level [36]. LSPR sensors can be fabricated in compact formats and produce visible color changes in response to analyte binding, which are often observable without sophisticated equipment [31]. The combination of these structural and optical advantages enables their effective deployment in real-time applications, including diagnostics [37], food safety [38], and environmental monitoring [39].
SERS amplifies Raman signals by utilizing the strong electromagnetic field generated at the surface of nanostructured metal materials [40,41]. When analytes are adsorbed onto these metal surfaces, localized surface plasmons enhance the electromagnetic field, significantly boosting the Raman signal. Signal enhancement occurs because of the interaction between the incident light and vibrational modes of the molecules, which shifts the frequency of the scattered light. The resulting spectral shift reveals a distinct vibrational profile that enables the precise identification of molecular species [42-44]. The high sensitivity and specificity of SERS make it ideal for chemical and biological sensing [42]. Although SPR and LSPR offer highly sensitive detection based on refractive index changes, SERS provides molecular-specific information via vibrational modes, offering a complementary approach to comprehensive analysis. A major advantage of nanoplasmonic sensors is their compatibility with integrated platforms, including lab-on-a-chip systems [45]. However, LSPR and SERS devices may experience limitations depending on the size and shape of the nanoparticles or substrate [42]. For example, elongated nanostructures, notably nanorods, exhibit increased responsiveness to changes in the bulk refractive index owing to their high aspect ratios. However, this improvement often comes at the cost of reduced surface sensitivity owing to the extended decay length of the localized electromagnetic field [46]. Accordingly, recent studies have focused on optimizing the nanostructure geometry and incorporating functional materials into the surface layers to enhance the molecular affinity or optical responsiveness [47-52].
2D materials, including graphene, transition metal dichalcogenides (TMDs), black phosphorus, and MXenes, have attracted substantial attention for chemical sensing because of their atomically thin structures and large surface-area-to-volume ratios [53,54]. A high density of exposed active sites allows for exceptional responsiveness to molecular-level interactions. The optical and electronic properties can be finely tuned by adjusting the composition, layer thickness, or surface functionalization, thereby enabling broad application potential [55,56]. Their high electrical conductivities, chemical stabilities, and straightforward integration onto various substrates have led to their increasing use in sensor interfaces and platforms [57]. The strong light–matter interactions observed in many 2D systems further contribute to signal amplification when paired with plasmonic nanostructures such as metal nanoparticles or patterned films [58]. The combination of an ultrathin morphology and tunable photonic behavior makes 2D materials promising candidates for next-generation optical chemical sensing.
Herein, we present the integration of 2D materials with optical chemical sensors as a promising approach to overcome the current limitations of sensitivity, selectivity, and device miniaturization. To establish the basis for this discussion, the key optical sensing mechanisms of SPR, LSPR, and SERS are examined. The inherent challenges associated with each sensing technique are identified, followed by a discussion of how the unique electronic and photonic properties of 2D materials, such as tunable band structures, large surface areas, and strong light–matter interactions, can enhance the sensor performance. Recent advances in the incorporation of these materials into various optical sensor platforms have demonstrated improvements in resonance tuning, signal amplification, and molecular recognition. This comprehensive analysis aims to offer new perspectives on the synergistic combination of 2D materials and optical sensors to guide future research and applications.
2. CLASSIFICATION OF OPTICAL CHEMICAL SENSORS: SPR, LSPR, AND SERS MECHANISMS
2.1 SPR
SPR sensors detect changes in the refractive index at the interface between a thin metal film and dielectric medium [27-29]. Surface plasmons are collective oscillations of free electrons on the surface of a noble metal, and their excitation forms the basis of the SPR sensing mechanism. When p-polarized light enters the system under the condition of total internal reflection, an evanescent electromagnetic field is generated along the metal–dielectric boundary. Resonance occurs when the in-plane momentum of the evanescent field matches that of the surface plasmons, causing a pronounced decrease in the reflected light intensity, known as the resonance dip, as shown in Fig. 2 (a). The position of the resonance dip is highly sensitive to the optical properties of the surrounding environment. The evanescent field typically penetrates only a few hundred nanometers into the adjacent medium, enabling the detection of subtle molecular interactions near the sensor surface. The adsorption of analyte molecules onto a functionalized surface changes the local refractive index and shifts the resonance angle or wavelength. The measurement of these shifts allows real-time sensing without chemical labels. Fig. 2 (b) illustrates a typical Kretschmann-configuration-based SPR sensor setup, where a gold film (~50 nm) is deposited onto a glass prism to support surface plasmon excitation [59]. When p-polarized light is incident at a specific angle, an evanescent wave is generated at the metal–dielectric interface, coupling with surface plasmons and resulting in a sharp dip in the reflected light intensity. The position of this resonance angle is highly sensitive to changes in the refractive index within a sub-500 nm detection depth above the gold surface. As shown in the accompanying graph, the binding events near the sensor surface cause a measurable shift in the resonance angle, providing a real-time, label-free method for detecting molecular interactions.
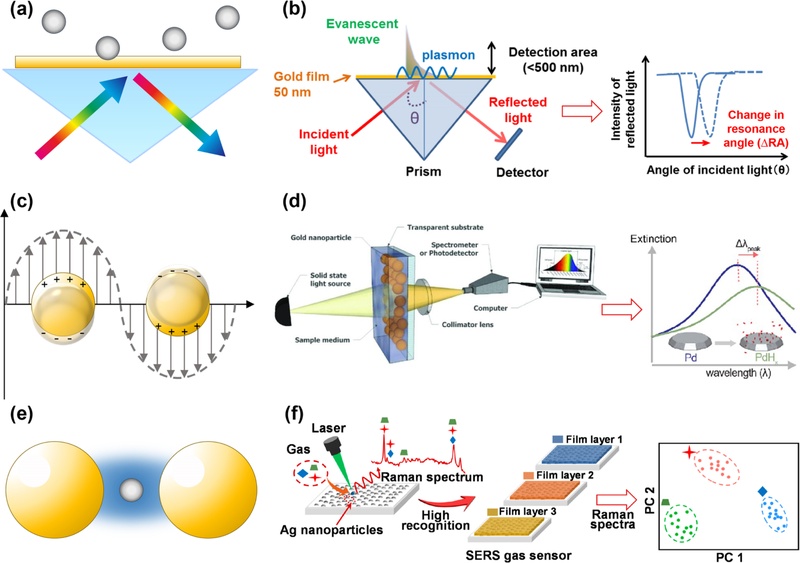
Overview of types and sensing mechanisms of optical-based chemical sensors. (a) Schematic of the surface plasmon resonance (SPR) principle. (b) Typical SPR-based chemical sensing measurement setup and representative refractive index changes within a detection area (<500 nm) with the change in the resonance angle (RA). Reprinted with permission from Ref. [59] under a CC BY 3.0 license, Copyright (2014) MDPI. (c) Schematic of the localized SPR (LSPR) principle. (d) Typical LSPR-based chemical sensing measurement setup and representative extinction spectra. Reprinted with permission from Ref. [61] under a CC BY 4.0 license, Copyright (2018) MDPI; and from Ref. [80] under a CC BY 4.0 license, Copyright (2021) American Chemical Society. (e) Schematic of the surface-enhanced Raman scattering (SERS) mechanism. (f) Typical LSPR-based chemical sensing measurement setup and capability of the SERS sensor to enable label-free simultaneous detection of multiple target species within mixed samples. Reprinted with permission from Ref. [63] under a CC BY 4.0 license, Copyright (2021) MDPI.
2.2. LSPR
LSPR sensors operate based on the interaction between incident light and the collective oscillations of electrons confined within metal nanostructures, typically gold or silver nanoparticles. When the nanoparticle size is comparable to or smaller than the wavelength of light, the electromagnetic field becomes strongly confined near the particle surface, producing a localized resonance effect, as illustrated in Fig. 2 (c) [35,36]. The detection relies on changes in the local refractive index near the nanoparticle surface. Analyte molecules bind to functional groups on the nanostructures and modify the dielectric environment [60]. The binding process alters the electron density and polarizability of the nanoparticles, leading to shifts in the LSPR frequency and corresponding changes in the extinction spectrum. The spectral shift, measured as changes in the peak wavelength or intensity, correlates directly with the analyte concentration, enabling sensitive detection [47]. Fig. 2 (d) shows a typical experimental setup in which a solid-state light source illuminates a transparent substrate coated with gold nanoparticles [61]. The light transmitted through the sample passes through a collimating lens before reaching a spectrometer or photodetector that records the extinction spectrum. The spectrum reveals characteristic absorption peaks that shift upon analyte binding, providing quantitative information regarding the surrounding environment. This setup enables rapid, label-free sensing and offers straightforward potential for miniaturization. These properties render LSPR sensors valuable for applications requiring portable real-time detection.
2.3. SERS
SERS significantly amplifies Raman signals by utilizing nanoplasmonic structures, typically metallic nanoparticles. When incident light excites the nanoparticles, a highly localized electromagnetic field is generated near their surfaces, greatly enhancing the Raman scattering from nearby adsorbed molecules (Fig. 2 (e)) [42-44]. Regions of intense electromagnetic enhancement, known as “hotspots,” often occur with sharp features or narrow gaps between nanoparticles, serving as primary sites for signal amplification [62]. The sensing mechanism depends on the changes in the local dielectric environment caused by analyte binding. As the molecules are adsorbed onto the plasmonic surface, the interactions modify the electronic structure and dielectric properties, resulting in measurable changes in the Raman spectrum. These changes manifest as variations in the peak intensity, position, or bandwidth, providing a molecular fingerprint with high specificity and sensitivity. Because of its strong enhancement effect, SERS can achieve detection limits down to the single-molecule level, making it exceptionally powerful for trace analyses [44]. Fig. 2 (f) depicts a typical SERS sensing setup, in which a laser beam irradiates a substrate coated with metal nanoparticles [63]. The scattered Raman signals are collected and analyzed to generate spectra that enable the precise identification of chemical species, even at very low concentrations. Multivariate analysis techniques can further distinguish complex mixtures by comparing spectral patterns, thereby enhancing the recognition accuracy. Strong signal enhancement, combined with molecular specificity, allows SERS systems to complement the detection principles of SPR and LSPR, thus forming a versatile and comprehensive toolkit for optical chemical sensing.
3. ENHANCING SPR SENSOR PERFORMANCE WITH PLASMONIC RESONANCE ENGINEERING
A typical SPR sensor uses a Kretschmann configuration consisting of a high-refractive-index prism coated with a thin metal film, often gold or silver. The incident light experiences total internal reflection at the prism interface, generating an evanescent wave that excites the surface plasmons at the metal–dielectric interface [64]. The resonance condition, identified by a sharp decrease in the reflected light intensity, shifts sensitively with changes in the refractive index near the metal surface caused by analyte binding. Prism-based systems enable real-time, label-free detection with high specificity and a rapid response and are widely applied in biosensing and environmental monitoring [65]. However, limitations include the large equipment size [66,67], reduced sensitivity to small molecules owing to long evanescent field decay lengths, and susceptibility to environmental factors.
To overcome these limitations, research has advanced towards multilayer material integration and structural optimization [68,69]. For example, Aliqab et al. performed numerical simulations of SPR sensors by integrating phase-change materials and hafnium dioxide (HfO2) [70]. Their proposed multilayer structure combined a silver layer or nanoparticles to provide strong plasmonic resonance, the phase-change material Ge2Sb2Te5 (GST) for dynamic optical tuning through reversible phase transitions, and HfO2 to enhance thermal stability and dielectric control (Fig. 3 (a)) [70]. Simulations of the analyte–Ag–Si–HfO2–Si–GST–Si configuration revealed twenty resonance modes within an analyte refractive index range of 1.2–2.4, achieving a high sensitivity of 1240 nm per refractive index unit (nm/RIU). This sensitivity level is suitable for detecting various biomolecules, such as glucose and hemoglobin.
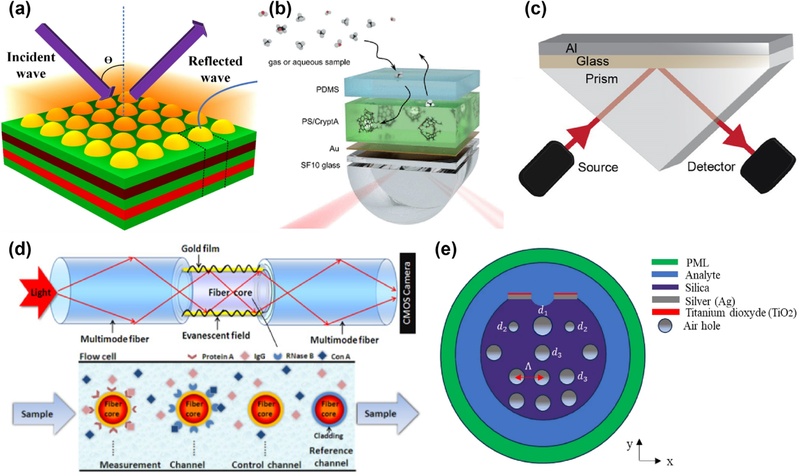
Strategies for performance enhancement and miniaturization of SPR sensors. (a) Three-dimensional schematic of the SPR sensor used in numerical simulations, incorporating phase-change material (GST) and HfO2. Reprinted with permission from Ref. [70], Copyright (2023) Springer Nature. (b) Methane sensor employing a sensing layer based on polystyrene polymer doped with cryptophane A (CryptA). Reprinted with permission from Ref. [71], Copyright (2024) American Chemical Society. (c) Approach to using Al as an alternative metal layer in SPR sensors. Reprinted with permission from Ref. [72], Copyright (2020) American Chemical Society. (d) Compact multichannel SPR biosensor developed based on a fiber-optic bundle architecture, with each channel functionalized to detect different target molecules selectively. Reprinted with permission from Ref. [74], Copyright (2015) Optica Publishing Group. (e) Cross-sectional view of the proposed dual-core PCF sensor. Reprinted with permission from Ref. [67] under a CC BY 4.0 license, Copyright (2024) Springer Nature.
Efforts to improve SPR sensor performance have also focused on the integration of selective polymer sensing layers. Polymers doped with target-specific molecules, such as polystyrene combined with methane-selective cryptophane A (CryptA), show enhanced stability and selectivity (Fig. 3 (b)) [71]. CryptA possesses a unique cage-like molecular structure that enables the selective encapsulation of small nonpolar molecules such as methane, providing high affinity through size and shape complementarity. Molecular recognition by the polymer film enables preferential methane binding despite the presence of other gases, enhancing the sensor selectivity. In addition, polystyrene acts as a stable matrix that uniformly disperses CryptA whereas a polydimethylsiloxane protective layer shields the sensing film from environmental fluctuations and enhances the signal stability. The optimization of the polymer layer thickness balances the sensitivity and response time by controlling the evanescent field penetration and analyte diffusion rates. The strategy demonstrated accurate and reproducible methane detection in both the gas and aqueous phases, highlighting selective polymer films as promising functional interfaces in SPR sensing systems.
Research on alternative metal layers for SPR sensors has gained momentum with the aim of improving the sensitivity, stability, and cost-effectiveness. Aluminum stands out because of its high electron density, broad plasmonic response range, and formation of a self-limiting oxide layer (Al2O3), which enhances the durability and corrosion resistance. In addition, the low cost of aluminum has enhanced its appeal for commercial applications. Lambert et al. applied aluminum as a metal layer in SPR biosensors (Fig. 3 (c)) [72]. Finite-difference time-domain (FDTD) simulations optimized the thickness of the aluminum layer to 15 nm, which was then deposited onto glass slides via electron-beam evaporation. Experimental measurements revealed a sensitivity of 70,041 IU/RIU for aluminum sensors, which was approximately 13.9% higher than that of gold, and a wider linear detection range. Stability tests conducted with 24-h immersion in a 10× PBS solution confirmed negligible changes in the surface morphology or spectral response, confirming the effective corrosion resistance conferred by the Al2O3 layer. The combined attributes of high plasmonic performance, excellent environmental stability, and antifouling capability position aluminum as a promising and cost-effective candidate for next-generation SPR sensors.
Following advancements in material engineering and structural optimization to enhance prism-based SPR sensors, fiber-optic SPR sensors have emerged as a promising solution to overcome challenges related to device size and portability [66,73]. In particular, transmissive fiber-optic SPR sensors employ a configuration similar to the Kretschmann setup, consisting of a light source, a fiber-based sensing region coated with a thin metal film, and a detector [66]. Light propagates through the fiber core, exciting the surface plasmons at the metal–dielectric interface within the sensing region. The resonance shows high sensitivity to local refractive index variations near the metal surface, which change upon analyte binding to the functionalized layers [66,74]. Consequently, fluctuations in the SPR absorption allow for the label-free detection and quantification of analytes in real time. Fig. 3 (d) shows a multichannel fiber-optic SPR sensor [74]. The fiberoptic bundle, functionalized with distinct biomolecular receptors, allows simultaneous detection of multiple analytes. This design includes channels modified with staphylococcal protein A and ribonuclease B (RNase B) for selectively binding immunoglobulin G (IgG) and concanavalin A (Con A), respectively. An unmodified reference channel compensates for environmental fluctuations and bulk refractive index changes, thus enhancing the measurement accuracy. A quantitative analysis showed a linear correlation between the signal intensity and analyte concentration within clinically relevant ranges. Integration with a mobile device facilitates remote data transmission and real-time monitoring, highlighting the potential for point-of-care diagnostics and environmental sensing.
To extend the sensing capabilities across different physical states, a microstructured optical-fiber-based SPR sensor was designed for multienvironment detection [67,75]. Fig. 3 (e) shows a D-shaped hollow-core photonic crystal fiber (HC-PCF)- based SPR sensor designed for multienvironment detection [67]. The sensor comprises a hollow core surrounded by periodic air holes and features a side-polished region coated with a thin gold film to enable SPR excitation. Light propagating through the core evanescently interacts with the metal–dielectric interface, allowing the detection of refractive index changes induced by target analytes. Simulation results indicated that such structures can achieve wavelength sensitivities exceeding 12,000 nm/RUI and high amplitude sensitivity, with resolution limits down to the order of 10-6 RIU. Future research will focus on optimizing the relationship between the optical properties, sensing layer function, and device integration to extend the detection limits and ensure robustness across diverse environments [76]. The continued refinement of sensor designs, improvements in selectivity, and enhanced compatibility with compact and flexible substrates will facilitate the transition from experimental prototypes to commercial systems.
4. OPTIMIZING LSPR SENSOR PERFORMANCE WITH NANOSTRUCTURES
LSPR sensors operate through the collective oscillations of electrons confined within noble metal nanoparticles or nanostructures. Compared with conventional SPR sensors, LSPR sensors exhibit significantly shorter plasmon decay lengths, offering enhanced sensitivity to analytes bound directly to the sensor surface, such as biomolecules. However, achieving high sensitivity in gaseous environments remains a technical challenge because of the similarity in the refractive indices of gases and air [47,77]. Therefore, recent research has focused on tuning the nanoparticle size, shape, composition, and interparticle coupling to sharpen the plasmon resonance peak and increase the sensitivity [78]. For example, Proença et al. fabricated quasi-hexagonally ordered arrays of gold nanoparticles with particle diameters controlled between 72 and 88 nm and interparticle gaps between 18 and 29 nm to optimize plasmon interactions. The hexagonal arrangement, known for its efficient packing and uniform near-field interactions, supports strong plasmon coupling effects that improve the sensor performance. This high-resolution LSPR spectroscopy setup facilitated the real-time monitoring of transmittance spectra shifts under both liquid and gas-phase conditions (Fig. 4 (a)) [47]. In addition, the conceptual distinction between the adsorbed gas layer and bulk gas phase provided a foundation for more accurate sensitivity evaluation by addressing the limitations of conventional refractive index sensitivity metrics in gas sensing (Fig. 4 (b)). The scanning electron microscopy (SEM) images in Fig. 4 (c) show the morphology of the single-, double-, and triple-layer nanoparticle configurations, which were systematically tested to compare their sensitivities. The double-layer structure yielded the highest refractive index sensitivity, emphasizing the critical role of nanoparticle morphology and precise spatial control in maximizing LSPR sensor responsiveness.
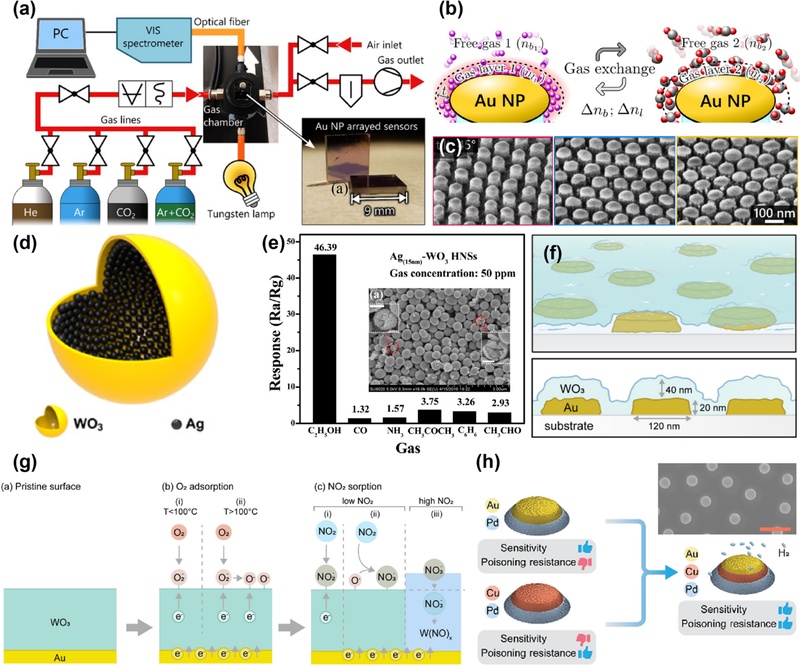
Strategies for performance enhancement of LSPR sensors through nanostructure arrangement and sensing layer integration. (a) High-resolution LSPR spectroscopy setup for real-time monitoring of sensor transmittance spectra. (b) Gas exchange process on the top of the ellipsoidal gold nanoparticles, considering surface-adsorbed gas layers. (c) SEM images showing three types of gold nanoparticle (Au NP) arrangements—single, double, and triple layers—on SiO2 nanopillars. The images were obtained at a 45˚ tilt and are displayed from right to left in the order of single, double, and triple layers. Reprinted with permission from Ref. [47] under a CC BY 4.0 license, Copyright (2024) American Chemical Society. (d) Schematic of Ag–WO3 hollow nanospheres (HNSs). (e) Response of Ag–WO3 HNSs to 50 ppm of various target gases at 230°C. Insets show low- and high-magnification FE-SEM images of Ag-WO3 HNSs. Reprinted with permission from Ref. [78], Copyright (2016) American Chemical Society. (f) Quasirandom array of Au nanodisks functionalized-with polycrystalline WO3 film for high-specificity NO2 detectio. (g) Proposed NO2 detection mechanism of Au–WO3 nanoplasmonic sensors. Reprinted with permission from Ref. [48] under a CC BY 4.0 license, Copyright (2022) American Chemical Society. (h) Tradeoff between sensitivity enhancement and CO-poisoning resistance provided by Au and Cu alloyants, respectively, in PdAuCu ternary alloy nanoparticles for plasmonic hydrogen sensing. This design achieves an optimized system with maximized sensitivity and CO-poisoning resistance. Insets show an SEM micrograph of the nanodisk array; the scale bar represents 500 nm. Reprinted with permission from Ref. [80] under a CC BY 4.0 license, Copyright (2021) American Chemical Society.
In addition to the morphology, the integration of heterogeneous nanomaterials has emerged as a promising route for improving both the sensitivity and selectivity. Yao Yao et al. synthesized Ag-WO3 hollow nanospheres via sonochemical methods, achieving a low operating temperature (230°C), ultralow detection limit (0.09 ppb), and fast response (7 s) for alcohol vapors [79]. The enhanced performance of the sensor was attributed to selective ethanol adsorption facilitated by the hydroxyl groups and subsequent surface oxidation, which enabled sensor reusability (Fig. 4 (d–e)). Similarly, ultrathin WO3 films deposited onto arrays of gold nanodisks offer spatially resolved and real-time detection of nitrogen dioxide (NO2) with high specificity (Fig. 4 (f–g)) [48]. Operated under laboratory, simulated urban, and field conditions, these nanoplasmonic sensors exploit the temperature-dependent oxygen adsorption chemistry on WO3 surfaces to bind NO2 selectively. The charge-transfer processes between NO2, WO3, and gold nanoparticles modulated the localized electron density, resulting in measurable redshifts in the LSPR spectrum. The combination of optical and chemical selectivity illustrates the potential of metal oxide–plasmonic nanoparticle composites for practical environmental sensing applications.
Material composition optimization through alloying offers another avenue for improving the sensor performance by tailoring the electronic and catalytic properties. The Pd65Au25Cu10 ternary alloy system exemplifies this approach, demonstrating enhanced hydrogen sensing with high sensitivity, a hysteresis-free response, and resistance to carbon monoxide (CO) poisoning (Fig. 4 (h)) [80]. Systematic compositional tuning and adherence to rigorous testing protocols confirmed the stability and suitability of the alloy for prolonged operation in challenging gas environments. This strategy underscores the value of multicomponent nanoparticle designs in addressing complex sensing challenges beyond morphological or hybrid material combinations. Collectively, these developments reveal a clear trajectory in LSPR sensor innovation, progressing from controlling the nanoparticle shape and spatial arrangement to the incorporation of functional hybrid materials and engineered alloy compositions. This multifaceted approach addresses fundamental sensing limitations, enabling highly sensitive and selective detection across diverse analyte environments.
5. BOOSTING SERS PERFORMANCE WITH NANOPARTICLES AND HYBRID STRUCTURES
SERS is a powerful analytical technique that amplifies molecular vibrational signals via plasmonic enhancement mechanisms. This enhancement primarily originates from two processes: the electromagnetic mechanism (EM), which intensifies the local electric field via collective oscillations of surface electrons on noble metals, and the chemical mechanism (CM), which is based on charge transfer between the analyte and substrate [81,82]. While the EM enhancement can reach a factor of 1010 and depends heavily on nanoparticle morphology and interparticle spacing, the CM generally contributes to a lower enhancement (~104) and is influenced by the electronic interaction between the material and target molecule [83,84]. Recent strategies have focused on engineered nanostructures that simultaneously boost EM and CM effects, with particular attention paid to ordered arrays and functional architectures (Fig. 5 (a)) [85].
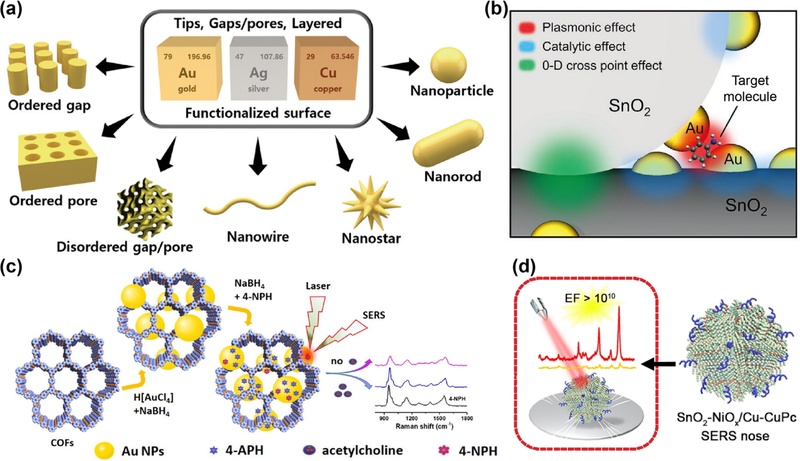
Strategies for enhancing SERS performance through novel nanoarchitecture development. (a) Categories of nanostructures with round tips and gaps or pores as SERS substrates, illustrating current research strategies. Reprinted with permission from Ref. [85] under a CC BY 4.0 license, Copyright (2022) Wiley-VCH GmbH. (b) The 3D-CMA structure depicting a cross-point junction of Au NP-decorated SnO2 nanowires. Reprinted with permission from Ref. [49], Copyright (2021) Wiley. (c) Schematic of the AuNPs@COF nanocomposite as a nanozyme for SERS-based ach detection. Reprinted with permission from Ref. [86], Copyright (2021) American Chemical Society. (d) Sponge-like Cu-doped SnO2-NiO p-n semiconductor heterostructure (SnO2-NiOx/Cu) functionalized with copper phthalocyanine (CuPc) molecules, forming a highly sensitive and selective SERS nose for VOC detection (SnO2-NiOx/Cu-CuPc). Reprinted with permission from Ref. [87], Copyright (2021) Wiley.
A representative implementation of structural engineering is the three-dimensional cross-point multifunctional architecture (3D-CMA) proposed by Han et al. [49], which integrates orthogonally stacked SnO2 nanowire frameworks with surface-decorated gold nanoparticles to enable electrical and SERS-based optical sensing simultaneously (Fig. 5 (b)). The intersecting nanowires establish discrete contact points for high-resolution electrical detection, whereas discontinuous Au nanoparticle coverage introduces plasmonic hotspots that amplify the Raman signals. Importantly, the design permits selective and label-free analysis of mixed aromatic gases, such as nitrobenzene and toluene, at concentrations below 100 ppm, demonstrating that hybridizing semiconducting frameworks with noble metals enhances multimodal sensitivity and target discrimination.
Enhancing the SERS functionality for gas-phase sensing requires not only the refinement of the nanostructure geometry, but also the incorporation of materials that are capable of analyte preconcentration and chemical discrimination. One promising strategy involves the integration of plasmonic nanostructures with porous organic frameworks to achieve simultaneous analyte capture and signal amplification. In this context, hybrid architectures consisting of gold nanoparticles grown in situ on covalent organic frameworks (AuNPs@COF) have been developed as bifunctional SERS platforms that combine catalytic activity and plasmonic enhancement [86]. The COF matrix, constructed using a TpPa-1 backbone, provides a high surface area and tunable porosity for efficient molecular adsorption, whereas the densely packed gold nanoparticles generate abundant Raman hotspots within the nanoscale interparticle gaps. Fu et al. demonstrated that such nanocomposites exhibit reductase-like catalytic activity toward 4-nitrophenol, enabling the sensitive and selective detection of inhibitory analytes that modulate the reduction pathway. With a detection limit as low as 0.3 pM for acetylcholine, the AuNPs@COF-based sensor showed excellent stability, reproducibility, and selectivity in both aqueous and serum matrices, underscoring its potential for real-time biochemical sensing and its applicability to the trace-level detection of biologically relevant molecules under ambient conditions.
In parallel with EM-dominant designs, efforts to maximize the chemical sensitivity have led to the exploration of chargetransfer-enhanced substrates. Zhou et al. reported a sponge-like Cu-doped SnO2-NiO p-n heterojunction structure that achieved an enhancement factor of 1.46 × 1010 primarily through the CM (Fig. 5 (d)) [87]. The p-n junction and Cu doping synergistically facilitated interfacial electron transfer, whereas the porous morphology increased the analyte concentration at the surface. When combined with copper phthalocyanine molecules to create a selective recognition layer, this platform enabled the detection of toxic VOCs in exhaled breath at the ppb level. Furthermore, the use of Raman intensity barcoding allowed for the intuitive visual differentiation of gas profiles, offering a promising avenue for early-stage disease diagnostics. Progress in SERS sensor technologies reflects a growing emphasis on multiscale precision—spanning nanostructure design, interface modulation, and molecular selectivity. Innovations in the structural geometry, material composition, and interfacial charge dynamics offer complementary pathways for overcoming gas-phase detection barriers. Despite advances in nanostructural engineering and hybrid material design, achieving consistent signal enhancement across large areas and ensuring chemical robustness in real-world environments remain ongoing challenges. Materials that combine high surface activity with intrinsic chemical stability are increasingly being considered vital for improving the reliability and reproducibility of SERS, particularly in complex sensing scenarios.
6. ADVANTAGES OF 2D MATERIALS IN OPTICAL CHEMICAL SENSORS
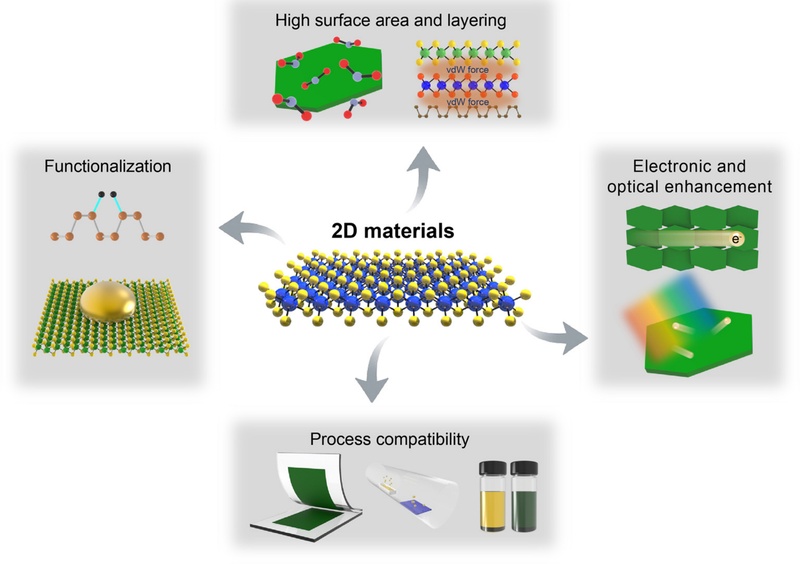
2D materials possess distinctive characteristics that offer promising solutions to long-standing challenges in optical chemical sensing. Atomic-scale thickness combined with a high surface-to-volume ratio significantly increases the number of available sites for analyte interactions, thereby enhancing the detection sensitivity of plasmonic platforms such as SPR and SERS [88,89]. Van der Waals (vdW) heterostructures, which are formed by stacking different 2D layers without lattice matching constraints, create hybrid systems with highly tunable optical and electronic properties (Fig. 6, top) [12,90]. Enhanced charge carrier mobility, interlayer tunneling, and Coulomb drag effects arise from such stacking, collectively improving electronic transport and light–matter coupling at the interfaces [91,92]. Engineered heterostructures expand the functional scope of sensors, enabling applications that require enhanced sensitivity, improved selectivity, and innovative sensing mechanisms.
The electronic and optical properties of 2D materials such as TMDs and black phosphorus exhibit strong tunability via layer thickness control, applied strain, and doping strategies [93-97]. The absence of dangling bonds in the basal plane, along with strong in-plane atomic bonding, supports the exceptional lateral charge-carrier mobility by minimizing electron scattering and facilitating efficient electrical transport [98]. High optical responsiveness arises from enhanced absorption and emission across a wide spectral range, specifically spanning from the visible (~400-700 nm, e.g., the PL peak of MoS2 near 660 nm) to the near-infrared (~700-900 nm, e.g., MoSe2 enhanced emission near 750 nm) region, depending on the material system and type of defect engineering applied [99]. Thickness-dependent bandgap modulation enables the precise tuning of electronic transitions and excitonic effects, directly impacting local electromagnetic field confinement and charge transfer processes [100,101]. The interaction between the electronic structure and optical response plays a critical role in enhancing plasmonic signals and enabling selective molecular detection (Fig. 6, right). Surface chemical functionalization involves diverse techniques ranging from covalent doping and ligand attachment to templated metal nanostructure growth [96,102-104]. The various modifications allow the tailored enhancement of electronic characteristics, introduction of molecular recognition sites, and improved integration with sensor surfaces. Notably, metal nanoparticles can be readily synthesized on the surface of 2D materials, which are highly suitable for plasmonic sensor applications. Atomic vacancies on the basal planes act as nucleation centers for metal nanoparticle formation, allowing a controlled spatial arrangement [103]. The intrinsically high electrical conductivity and chemical reactivity of 2D material templates promote synergistic charge transfer and catalytic interactions with deposited metals [104,105]. Functionalization strategies can offer fine control over the interfacial chemistry and morphology, ultimately advancing the sensor selectivity, stability, and overall performance (Fig. 6, left) [104].
A key practical advantage arises from the exceptional processability of 2D materials (Fig. 6, bottom). Traditional chemical vapor deposition (CVD) methods yield high-quality films, but encounter challenges in terms of scalability and substrate compatibility. Recent advancements in solution-based exfoliation and chemical synthesis have enabled the production of stable nanosheet dispersions with controlled thickness and phase purity [106]. Electrochemical intercalation using bulky organic ions, such as tetraheptylammonium bromide, expands the interlayer spacing of bulk MoS2, allowing gentle sonication-assisted exfoliation to produce uniform nanosheets. Solution-processed 2D nanosheets can be deposited onto various substrates using scalable coating methods, enabling their integration into diverse device architectures [107-109]. Clean vdW interfaces form between the stacked layers, preserving excellent charge transport properties comparable to those of mechanically exfoliated or CVD-grown films [110]. This processability broadens the application potential of optical sensing across the SPR, LSPR, and SERS platforms, advancing the development of highly sensitive, selective, and adaptable chemical sensors.
7. ENHANCING OPTICAL CHEMICAL SENSORS WITH 2D MATERIALS
Owing to the promising attributes of 2D materials, an increasing number of studies have integrated them into the SPR, LSPR, and SERS platforms in recent years. Numerous studies have supported the potential of 2D materials for addressing the specific limitations of each sensing modality [111,112]. Coating with heterostructures such as PtSe2 and BlueP–MoS2 significantly enhances the sensitivity and stability of SPR sensors, addressing challenges related to low analyte concentrations and environmental robustness (Fig. 7 (a)) [113]. Chowdhury et al. employed FDTD simulations to design a Kretschmann-configuration SPR sensor. The architecture features a low-refractive-index CaF2 prism, a PtSe2 layer that elevates the effective refractive index to boost sensitivity, an Au metal film for SPR excitation, and a BlueP–MoS2 heterostructure as the sensing layer that directly interacts with analytes. The heterostructure exploits the distinct electrical and optical properties of BlueP and TMDs, enabling a tunable photoresponse, heightened sensitivity, and greater ambient stability [114,115]. Plasmon–exciton coupling is intensified, amplifying the electromagnetic fields at the sensor interface and lowering the detection limits [116]. Angular interrogation combined with FDTD simulations optimizes the analyte thickness and field distribution, resulting in a 3.2-fold sensitivity improvement compared with conventional gold-based SPR sensors.
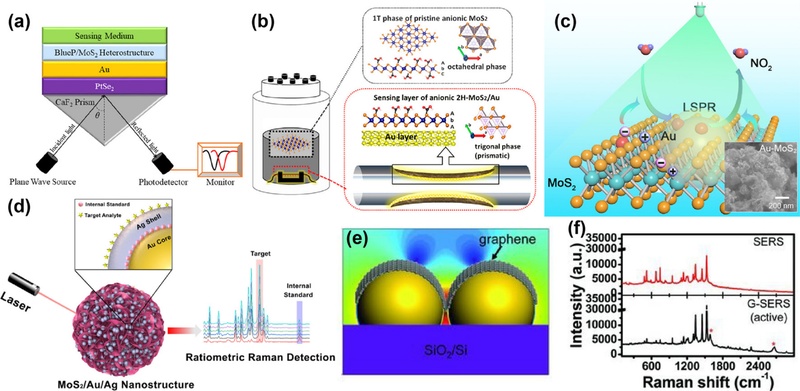
Recent advances in external enhancement of 2Dmaterial-based nanoplasmonic sensors. (a) SPR sensor based on PtSe2 and BlueP–MoS2 heterostructure, with a PtSe2 layer that increases the effective refractive index to boost sensitivity and a BlueP–MoS2 heterostructure serving as the sensing layer directly interacting with analytes. Reprinted with permission from Ref. [113], Copyright (2024) American Chemical Society. (b) Overall process for surface functionalization of an optical fiber by coating an anionic 2H-phase MoS2/Au sensing layer via a one-step recovery hydrothermal method. Reprinted with permission from Ref. [117], Copyright (2020) American Chemical Society. (c) 530 nm light-assisted Au–MoS2 gas sensor utilizing the LSPR effect of Au nanoparticles (NPs). The inset shows an FE-SEM image of Au–MoS2. Reprinted with permission from Ref. [50], Copyright (2021) American Chemical Society. (d) SERS sensor based on 3D MoS2 nanoflowers combined with Au/Ag nanostructures for ratiometric analysis of pesticide residues. Reprinted with permission from Ref. [51], Copyright (2023) American Chemical Society. (e) Graphene-veiled SERS substrates. (f) SERS performance of normal SERS (top) and G-SERS regions (bottom) after annealing. Reprinted with permission from Ref. [52], Copyright (2013) Wiley.
Fiber-optic SPR sensors also benefit from 2D material functionalization, which improves the VOC detection performance. Ashkavand et al. synthesized carboxyl (COOH)- functionalized MoS2 nanosheets using a low-temperature hydrothermal method and introduced them as sensing layers on gold-unclad polymeric optical fibers (Fig. 7 (b)) [117]. The presence of the gold substrate during synthesis induces a 1T-to-2H phase transition in MoS2, yielding semiconducting properties [118]. Strong chemical bonding between the COOH groups and gold ensures a uniform and robust nanosheet attachment to the curved fiber surfaces. Enhanced interactions with hydroxyl-containing VOCs increases the sensor sensitivity and selectivity. The sensor responses were evaluated for ethanol, propanol, and methanol concentrations ranging from 600 to 5000 ppm and showed excellent linearity (R2 = 0.999) and stable performance at 40% relative humidity.
The advantages of LSPR sensors include the integration of 2D gold nanostructures with light illumination to boost their gas-sensing capabilities. A 530 nm light-assisted Au–MoS2 sensor demonstrated NO2 detection limits as low as 10 ppb at room temperature without external heating while maintaining strong resistance to humidity (Fig. 7 (c)) [50]. Optical simulations and sensor characterization confirmed that LSPR excitation by 30 nm gold nanoparticles under 530 nm illumination significantly enhanced visible light absorption and increased the surface carrier concentration in MoS2, markedly improving the sensor sensitivity.
In SERS systems, the combination of 2D materials with nanostructures creates synergistic effects that amplify the signal enhancement mechanisms. Semiconductor-based core–molecule–shell nanoparticles show great promise as quantitative SERS substrates, particularly when used alongside ratiometric analysis (Fig. 7 (d)) [51]. Internal standard molecules such as 4-mercaptobenzonitrile embedded within the silver shell protect against environmental perturbations and enhance the detection accuracy. Gold nanoparticles serve as cores to amplify the Raman signals, whereas MoS2 nanoflowers contribute to strong chemical enhancement and provide a large surface area for supporting nanomaterials and analytes [119]. The 3D architecture generates electromagnetic hotspots, facilitating the sensitive detection of pesticide residues [120].
Recent developments have introduced graphene as a key component of shell-isolated SERS substrates. Mechanically exfoliated graphene contacts an 8 nm-thick island-like gold film following thermal annealing, forming curved gold nanoislands coated with graphene (Fig. 7 (e)) [52]. As opposed to conventional substrates that rely on direct molecular adsorption to metal surfaces, graphene coatings offer a passivated surface that is favorable for Raman analysis by enhancing molecular interactions, reducing signal loss, and improving reproducibility [121,122]. Comparisons of the Raman signals from the graphene-covered versus gold-only regions revealed significantly enhanced and more consistent signals in the graphene areas (Fig. 7 (f)). The shell-isolation approach provides insights into the influence of surface morphology on SERS performance and clarifies the mechanisms underlying Raman signal enhancement.
8. CONCLUSIONS AND OUTLOOK
2D materials offer distinct advantages that align well with the demands of optical chemical sensing technologies. Their atomic-scale thickness, large surface-to-volume ratio, and tunable electronic and optical properties create strong synergy with plasmonic sensors. In recent years, efforts to integrate 2D materials into SPR, LSPR, and SERS platforms have led to notable improvements in their sensitivity, selectivity, and stability. Initial strategies focused on heterostructure assembly, surface functionalization, and hybrid nanostructure formation to enhance light–matter interactions and charge transport while improving sensor responsiveness under realistic conditions. The use of 2D materials as active sensing layers or functional coatings has helped to overcome challenges related to detecting low-concentration analytes and operating in complex environments.
However, challenges remain in achieving uniform large-area synthesis and seamless integration with diverse device architectures. The sensor stability and reproducibility under practical operating conditions require further investigation. Moreover, scalable fabrication techniques that are compatible with various substrates are essential for transitioning laboratory prototypes into commercial products. Future research is expected to focus on optimized material design, interface engineering, and the development of multifunctional sensing platforms. Addressing integration challenges via scalable and substrate-compatible fabrication methods is critical for fully leveraging the benefits of 2D materials. In this regard, advances in solution-based fabrication offer versatility and compatibility, providing a promising path towards real-world applications of high-performance optical chemical sensors. Scalable solution-based processes facilitate the production of high-quality 2D nanosheets and their straightforward integration into flexible and rigid substrates [109]. This progress can significantly lower the barriers to material integration and accelerate the commercialization of advanced optical chemical sensing technologies. By combining material innovation with scalable manufacturing, the realization (e.g., high performance, reliability, and adaptability) of optical chemical sensors is becoming increasingly attainable.
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean Government (MSIT) (RS-2023-00208538).
References
-
B. Pejcic, P. Eadington, A. Ross, Environmental Monitoring of Hydrocarbons: A Chemical Sensor Perspective, Environ. Sci. Technol. 41 (2007) 6333–6342.
[https://doi.org/10.1021/es0704535]

-
M.A.A. Mamun, M.R. Yuce, Recent Progress in Nanomaterial Enabled Chemical Sensors for Wearable Environmental Monitoring Applications, Adv. Funct. Mater. 30 (2020) 2005703.
[https://doi.org/10.1002/adfm.202005703]

-
T. Arakawa, D.V. Dao, K. Mitsubayashi, Biosensors and Chemical Sensors for Healthcare Monitoring: A Review, IEEJ Trans. Electr. Electron. Eng. 17 (2022) 626–636.
[https://doi.org/10.1002/tee.23580]

-
R. Zhang, J. Jiang, W. Wu, Wearable chemical sensors based on 2D materials for healthcare applications, Nanoscale 15 (2023) 3079–3105.
[https://doi.org/10.1039/D2NR05447G]

-
R. Bogue, Optical chemical sensors for industrial applications, Sens. Rev. 27 (2007) 86–90.
[https://doi.org/10.1108/02602280710731632]

-
P.K. Sekhar, E.L. Brosha, R. Mukundan, F. Garzon, Chemical Sensors for Environmental Monitoring and Homeland Security, Electrochem. Soc. Interface 19 (2010) 35.
[https://doi.org/10.1149/2.F04104if]

-
A. Hulanicki, S. Glab, F. Ingman, Chemical sensors: definitions and classification, Pure Appl. Chem. 63 (1991) 1247–1250.
[https://doi.org/10.1351/pac199163091247]

-
K. Lang, T. Liu, D.J. Padilla, M. Nelson, C.W. Landorf, R.J. Patel, et al., Nanofibers enabled advanced gas sensors: A review, Adv. Sens. Energy Mater. 3 (2024) 100093.
[https://doi.org/10.1016/j.asems.2024.100093]

-
I.J. Choi, B.J. Kim, S.H. Lee, B.J. Jeong, T. Nasir, Y.S. Cho, et al., Fabrication of a room-temperature NO2 gas sensor using morphology controlled CVD-grown tellurium nanostructures, Sens. Actuators B Chem. 333 (2021) 128891.
[https://doi.org/10.1016/j.snb.2020.128891]

-
N. Joshi, G. Pransu, C.A. Conte-Junior, Critical review and recent advances of 2D materials-Based gas sensors for food spoilage detection, Crit. Rev. Food Sci. Nutr. 63 (2023) 10536–10559.
[https://doi.org/10.1080/10408398.2022.2078950]

-
X. Zhang, J. Zhang, C. Li, X. Zhang, J. Yun, D. Cao, A review on nanofiber-based composites for toxic and flammable gas sensing, Adv. Compos. Hybrid Mater. 7 (2024) 108.
[https://doi.org/10.1007/s42114-024-00922-6]

-
C. Anichini, W. Czepa, D. Pakulski, A. Aliprandi, A. Ciesielski, P. Samorì, Chemical sensing with 2D materials, Chem. Soc. Rev. 47 (2018) 4860–4908.
[https://doi.org/10.1039/C8CS00417J]

-
H. Chen, D. Huo, J. Zhang, Gas Recognition in E-Nose System: A Review, IEEE Trans. Biomed. Circuits Syst. 16 (2022) 169–184.
[https://doi.org/10.1109/TBCAS.2022.3166530]

-
S. Dhanekar, Smart and Intelligent E-nose for Sensitive and Selective Chemical Sensing Applications, In: H. Hallil, H. Heidari (Eds.), Smart Sensors for Environmental and Medical Applications, John Wiley & Sons, Inc., Hoboken, 2020, pp. 149–171.
[https://doi.org/10.1002/9781119587422.ch8]

-
H. Hu, X. Yang, X. Guo, K. Khaliji, S.R. Biswas, F.J. García de Abajo, et al., Gas identification with graphene plasmons, Nat. Commun. 10 (2019) 1131.
[https://doi.org/10.1038/s41467-019-09008-0]

-
K. Kumar, S.N. Chaudhri, N.S. Rajput, A.V. Shvetsov, R. Sahal, S.H. Alsamhi, An IoT-Enabled E-Nose for Remote Detection and Monitoring of Airborne Pollution Hazards Using LoRa Network Protocol, Sensors 23 (2023) 4885.
[https://doi.org/10.3390/s23104885]

-
K.K. Pulluri, V.N. Kumar, Development of an Integrated Soft E-Nose for Food Quality Assessment, IEEE Sens. J. 22 (2022) 15111–15122.
[https://doi.org/10.1109/JSEN.2022.3182480]

-
J. Fitzgerald, H. Fenniri, Cutting Edge Methods for Non-Invasive Disease Diagnosis Using E-Tongue and E-Nose Devices, Biosensors 7 (2017) 59.
[https://doi.org/10.3390/bios7040059]

-
G. Neri, First Fifty Years of Chemoresistive Gas Sensors, Chemosensors 3 (2015) 1–20.
[https://doi.org/10.3390/chemosensors3010001]

-
M. Kang, I. Cho, J. Park, J. Jeong, K. Lee, B. Lee, et al., High Accuracy Real-Time Multi-Gas Identification by a Batch-Uniform Gas Sensor Array and Deep Learning Algorithm, ACS Sens. 7 (2022) 430–440.
[https://doi.org/10.1021/acssensors.1c01204]

-
M. Strauch, A. Lüdke, D. Münch, T. Laudes, C.G. Galizia, E. Martinelli, et al., More than apples and oranges - Detecting cancer with a fruit fly's antenna, Sci. Rep. 4 (2014) 3576.
[https://doi.org/10.1038/srep03576]

-
M. Lee, H. Yang, D. Kim, M. Yang, T.H. Park, S. Hong, Human-like smelling of a rose scent using an olfactory receptor nanodisc-based bioelectronic nose, Sci. Rep. 8 (2018) 13945.
[https://doi.org/10.1038/s41598-018-32155-1]

-
K. Mitsubayashi, K. Yokoyama, T. Takeuchi, I. Karube, Gas-Phase Biosensor for Ethanol, Anal. Chem. 66 (1994) 3297–3302.
[https://doi.org/10.1021/ac00092a004]

-
K. Mitsubayashi, K. Toma, K. Iitani, T. Arakawa, Gas-phase biosensors: A review, Sens. Actuators B Chem. 367 (2022) 132053.
[https://doi.org/10.1016/j.snb.2022.132053]

-
Dhanjai, X. Lu, L. Wu, J. Chen, Y. Lu, Robust Single-Molecule Enzyme Nanocapsules for Biosensing with Significantly Improved Biosensor Stability, Anal. Chem. 92 (2020) 5830–5837.
[https://doi.org/10.1021/acs.analchem.9b05466]

-
G. Goumas, E.N. Vlachothanasi, E.C. Fradelos, D.S. Mouliou, Biosensors, Artificial Intelligence Biosensors, False Results and Novel Future Perspectives, Diagnostics 15 (2025) 1037.
[https://doi.org/10.3390/diagnostics15081037]

-
K.H. Kim, S. Jo, S.E. Seo, J. Kim, D.-S. Lee, S. Joo, et al., Ultrasensitive Gas Detection Based on Electrically Enhanced Nanoplasmonic Sensor with Graphene-Encased Gold Nanorod, ACS Sens. 8 (2023) 2169–2178.
[https://doi.org/10.1021/acssensors.2c02414]

-
L.G. Bousiakou, H. Gebavi, L. Mikac, S. Karapetis, M. Ivanda, Surface enhanced Raman spectroscopy for molecular identification-A review on surface plasmon resonance (SPR) and localised surface plasmon resonance (LSPR) in optical nanobiosensing, Croat. Chem. Acta 92 (2019) 479–494.
[https://doi.org/10.5562/cca3558]

-
J. Homola, Surface Plasmon Resonance Sensors for Detection of Chemical and Biological Species, Chem. Rev. 108 (2008) 462–493.
[https://doi.org/10.1021/cr068107d]

-
J. Homola, Present and future of surface plasmon resonance biosensors, Anal. Bioanal. Chem. 377 (2003) 528–539.
[https://doi.org/10.1007/s00216-003-2101-0]

-
J.-F. Masson, Portable and field-deployed surface plasmon resonance and plasmonic sensors, Analyst 145 (2020) 3776–3800.
[https://doi.org/10.1039/D0AN00316F]

-
C. Caucheteur, T. Guo, J. Albert, Review of plasmonic fiber optic biochemical sensors: improving the limit of detection, Anal. Bioanal. Chem. 407 (2015) 3883–3897.
[https://doi.org/10.1007/s00216-014-8411-6]

-
J.A. Jackman, A. Rahim Ferhan, N.-J. Cho, Nanoplasmonic sensors for biointerfacial science, Chem. Soc. Rev. 46 (2017) 3615–3660.
[https://doi.org/10.1039/C6CS00494F]

-
R.K. Gangwar, A.K. Pathak, F. Chiavaioli, M.A. Bakar, Y. Kamil, M. Mahdi, et al., Optical fiber SERS sensors: Unveiling advances, challenges, and applications in a miniaturized technology, Coord. Chem. Rev. 510 (2024) 215861.
[https://doi.org/10.1016/j.ccr.2024.215861]

-
F. Mazzotta, T.W. Johnson, A.B. Dahlin, J. Shaver, S.-H. Oh, F. Höök, Influence of the Evanescent Field Decay Length on the Sensitivity of Plasmonic Nanodisks and Nanoholes, ACS Photonics 2 (2015) 256–262.
[https://doi.org/10.1021/ph500360d]

-
K.M. Mayer, J.H. Hafner, Localized Surface Plasmon Resonance Sensors, Chem. Rev. 111 (2011) 3828–3857.
[https://doi.org/10.1021/cr100313v]

-
S. Farooq, F. Wali, D.M. Zezell, R.E. de Araujo, D. Rativa, Optimizing and Quantifying Gold Nanospheres Based on LSPR Label-Free Biosensor for Dengue Diagnosis, Polymers 14 (2022) 1592.
[https://doi.org/10.3390/polym14081592]

-
D.I. Meira, A.I. Barbosa, J. Borges, R.L. Reis, V.M. Correlo, F. Vaz, Label-free localized surface plasmon resonance (LSPR) biosensor, based on Au-Ag NPs embedded in TiO2 matrix, for detection of Ochratoxin-A (OTA) in wine, Talanta 284 (2025) 127238.
[https://doi.org/10.1016/j.talanta.2024.127238]

-
W. Liu, K. Chung, S. Yu, L.P. Lee, Nanoplasmonic biosensors for environmental sustainability and human health, Chem. Soc. Rev. 53 (2024) 10491–10522.
[https://doi.org/10.1039/D3CS00941F]

-
S. Jayawardhana, L. Rosa, S. Juodkazis, P.R. Stoddart, Additional Enhancement of Electric Field in Surface-Enhanced Raman Scattering due to Fresnel Mechanism, Sci. Rep. 3 (2013) 2335.
[https://doi.org/10.1038/srep02335]

-
R. Pilot, R. Signorini, C. Durante, L. Orian, M. Bhamidipati, L. Fabris, A Review on Surface-Enhanced Raman Scattering, Biosensors 9 (2019) 57.
[https://doi.org/10.3390/bios9020057]

-
B.P. Nanda, P. Rani, P. Paul, Aman, S.S. Ganti, R. Bhatia, Recent trends and impact of localized surface plasmon resonance (LSPR) and surface-enhanced Raman spectroscopy (SERS) in modern analysis, J. Pharm. Anal. 14 (2024) 100959.
[https://doi.org/10.1016/j.jpha.2024.02.013]

-
H. Kim, B.T. Trinh, K.H. Kim, J. Moon, H. Kang, K. Jo, et al., Au@ZIF-8 SERS paper for food spoilage detection, Biosens. Bioelectron. 179 (2021) 113063.
[https://doi.org/10.1016/j.bios.2021.113063]

-
K.A. Willets, Super-resolution imaging of SERS hot spots, Chem. Soc. Rev. 43 (2014) 3854–3864.
[https://doi.org/10.1039/C3CS60334B]

-
G.A. Lopez, M.C. Estevez, M. Soler, L.M. Lechuga, Recent advances in nanoplasmonic biosensors: applications and lab-on-a-chip integration, Nanophotonics 6 (2017) 123–136.
[https://doi.org/10.1515/nanoph-2016-0101]

-
K.L. Kelly, E. Coronado, L.L. Zhao, G.C. Schatz, The Optical Properties of Metal Nanoparticles: The Influence of Size, Shape, and Dielectric Environment, J. Phys. Chem. B. 107 (2003) 668–677.
[https://doi.org/10.1021/jp026731y]

-
M. Proença, T. Lednický, D.I. Meira, M.S. Rodrigues, F. Vaz, J. Borges, et al., New Parameter for Benchmarking Plasmonic Gas Sensors Demonstrated with Densely Packed Au Nanoparticle Layers, ACS Appl. Mater. Interfaces 16 (2024) 57832–57842.
[https://doi.org/10.1021/acsami.4c11102]

-
I. Tanyeli, I. Darmadi, M. Sech, C. Tiburski, J. Fritzsche, O. Andersson, et al., Nanoplasmonic NO2 Sensor with a Sub-10 Parts per Billion Limit of Detection in Urban Air, ACS Sens. 7 (2022) 1008–1018.
[https://doi.org/10.1021/acssensors.1c02463]

-
H.J. Han, S.H. Cho, S. Han, J.S. Jang, G.R. Lee, E.N. Cho, et al., Synergistic Integration of Chemo-Resistive and SERS Sensing for Label-Free Multiplex Gas Detection, Adv. Mater. 33 (2021) 2105199.
[https://doi.org/10.1002/adma.202105199]

-
P. Chen, J. Hu, M. Yin, W. Bai, X. Chen, Y. Zhang, MoS2 Nanoflowers Decorated with Au Nanoparticles for Visible-Light-Enhanced Gas Sensing, ACS Appl. Nano Mater. 4 (2021) 5981–5991.
[https://doi.org/10.1021/acsanm.1c00847]

-
X. Yu, Y. Sun, J. Hu, J. Wang, X. Zhuang, S. Zhang, et al., MoS2/Au/Ag Nanostructures for Ratiometric Surface-Enhanced Raman Scattering Determination of Pesticide Residues, ACS Appl. Nano Mater. 6 (2023) 685–694.
[https://doi.org/10.1021/acsanm.2c04843]

-
W. Xu, J. Xiao, Y. Chen, Y. Chen, X. Ling, J. Zhang, Graphene-Veiled Gold Substrate for Surface-Enhanced Raman Spectroscopy, Adv. Mater. 25 (2013) 928–933.
[https://doi.org/10.1002/adma.201204355]

-
A. Mirzaei, J.-Y. Kim, H.W. Kim, S.S. Kim, Resistive Gas Sensors Based on 2D TMDs and MXenes, Acc. Chem. Res. 57 (2024) 2395–2413.
[https://doi.org/10.1021/acs.accounts.4c00323]

-
S.-Y. Cho, H.-J. Koh, H.-W. Yoo, H.-T. Jung, Tunable Chemical Sensing Performance of Black Phosphorus by Controlled Functionalization with Noble Metals, Chem. Mater. 29 (2017) 7197–7205.
[https://doi.org/10.1021/acs.chemmater.7b01353]

-
A.F. Palmstrom, P.K. Santra, S.F. Bent, Atomic layer deposition in nanostructured photovoltaics: tuning optical, electronic and surface properties, Nanoscale 7 (2015) 12266–12283.
[https://doi.org/10.1039/C5NR02080H]

-
B.I. Karawdeniya, A.M. Damry, K. Murugappan, S. Manjunath, Y.N.D. Bandara, C.J. Jackson, et al., Surface Functionalization and Texturing of Optical Metasurfaces for Sensing Applications, Chem. Rev. 122 (2022) 14990–15030.
[https://doi.org/10.1021/acs.chemrev.1c00990]

-
L. Zhang, K. Khan, J. Zou, H. Zhang, Y. Li, Recent Advances in Emerging 2D Material-Based Gas Sensors: Potential in Disease Diagnosis, Adv. Mater. Interfaces 6 (2019) 1901329.
[https://doi.org/10.1002/admi.201901329]

-
X. Liu, T. Galfsky, Z. Sun, F. Xia, E.-C. Lin, Y.-H. Lee, et al., Strong light–matter coupling in two-dimensional atomic crystals, Nat. Photonics 9 (2015) 30–34.
[https://doi.org/10.1038/nphoton.2014.304]

-
Y. Yanase, T. Hiragun, K. Ishii, T. Kawaguchi, T. Yanase, M. Kawai, et al., Surface Plasmon Resonance for Cell-Based Clinical Diagnosis, Sensors 14 (2014) 4948–4959.
[https://doi.org/10.3390/s140304948]

-
M. Piliarik, P. Kvasnička, N. Galler, J.R. Krenn, J. Homola, Local refractive index sensitivity of plasmonic nanoparticles, Opt. Express 19 (2011) 9213–9220.
[https://doi.org/10.1364/OE.19.009213]

-
B.A. Prabowo, A. Purwidyantri, K.-C. Liu, Surface Plasmon Resonance Optical Sensor: A Review on Light Source Technology, Biosensors 8 (2018) 80.
[https://doi.org/10.3390/bios8030080]

-
R.C. Maher, SERS Hot Spots, In: C.S.S.R. Kumar (Ed.), Raman Spectroscopy for Nanomaterials Characterization, Springer, Berlin, 2012, pp. 215–260.
[https://doi.org/10.1007/978-3-642-20620-7_10]

-
L. Chen, H. Guo, F. Sassa, B. Chen, K. Hayashi, SERS Gas Sensors Based on Multiple Polymer Films with High Design Flexibility for Gas Recognition, Sensors 21 (2021) 5546.
[https://doi.org/10.3390/s21165546]

-
S. Ekgasit, C. Thammacharoen, W. Knoll, Surface Plasmon Resonance Spectroscopy Based on Evanescent Field Treatment, Anal. Chem. 76 (2004) 561–568.
[https://doi.org/10.1021/ac035042v]

-
P.S. Pandey, A. Shadab, M. Akkur, Y. Yadav, M. Kumar, M.S. Naruka, et al., Advancements in Fiber and Prism-Based Surface Plasmon Resonance Sensors: Comparative Analysis and Applications in Disease Detection and Biosensing, Plasmonics (2025) 1–27.
[https://doi.org/10.1007/s11468-024-02745-z]

-
S. Zhang, B. Han, Y.-N. Zhang, Y. Liu, W. Zheng, Y. Zhao, Multichannel Fiber Optic SPR Sensors: Realization Methods, Application Status, and Future Prospects, Laser Photonics Rev. 16 (2022) 2200009.
[https://doi.org/10.1002/lpor.202200009]

-
S. Kaziz, F. Echouchene, M.H. Gazzah, Optimizing PCF-SPR sensor design through Taguchi approach, machine learning, and genetic algorithms, Sci. Rep. 14 (2024) 7837.
[https://doi.org/10.1038/s41598-024-55817-9]

-
A. Cherifi, B. Bouhafs, Potential of SPR sensors based on multilayer interfaces with gold and LHM for biosensing applications, Photonic Sens. 7 (2017) 199–205.
[https://doi.org/10.1007/s13320-017-0425-1]

-
F. Xia, H. Song, Y. Zhao, W.-M. Zhao, Q. Wang, X.-Z. Wang, et al., Ultra-high sensitivity SPR fiber sensor based on multilayer nanoparticle and Au film coupling enhancement, Measurement 164 (2020) 108083.
[https://doi.org/10.1016/j.measurement.2020.108083]

-
K. Aliqab, V. Sorathiya, M. Alsharari, K. Dave, A. Armghan, Numerical analysis of hafnium oxide and phase change material-based multi-layered infrared and visible frequency sensor for biomolecules sensing application, Sci. Rep. 13 (2023) 7698.
[https://doi.org/10.1038/s41598-023-34817-1]

-
A. Estelmann, R. Prien, W. Marz, M. Elbing, P. Harz, G. Rehder, An SPR-Based In Situ Methane Sensor for the Aqueous and Gas Phase, Anal. Chem. 96 (2024) 16203–16214.
[https://doi.org/10.1021/acs.analchem.4c02875]

-
A.S. Lambert, S.N. Valiulis, A.S. Malinick, I. Tanabe, Q. Cheng, Plasmonic Biosensing with Aluminum Thin Films under the Kretschmann Configuration, Anal. Chem. 92 (2020) 8654–8659.
[https://doi.org/10.1021/acs.analchem.0c01631]

-
X. Zhang, Z. Li, W. Yan, A. Li, F. Zhang, X. Li, et al., Customizable miniaturized SPR instrument, Talanta 269 (2024) 125440.
[https://doi.org/10.1016/j.talanta.2023.125440]

-
Y. Liu, S. Chen, Q. Liu, J.-F. Masson, W. Peng, Compact multi-channel surface plasmon resonance sensor for real-time multi-analyte biosensing, Opt. Express 23 (2015) 20540–20548.
[https://doi.org/10.1364/OE.23.020540]

-
W. Liu, C. Liu, J. Wang, J. Lv, Y. Lv, L. Yang, et al., Surface plasmon resonance sensor composed of microstructured optical fibers for monitoring of external and internal environments in biological and environmental sensing, Results Phys. 47 (2023) 106365.
[https://doi.org/10.1016/j.rinp.2023.106365]

-
J. Jing, K. Liu, J. Jiang, T. Xu, S. Wang, J. Ma, et al., Performance improvement approaches for optical fiber SPR sensors and their sensing applications, Photonics Res. 10 (2022) 126–147.
[https://doi.org/10.1364/PRJ.439861]

-
S.J. Zalyubovskiy, M. Bogdanova, A. Deinega, Y. Lozovik, A.D. Pris, K.H. An, et al., Theoretical limit of localized surface plasmon resonance sensitivity to local refractive index change and its comparison to conventional surface plasmon resonance sensor, J. Opt. Soc. Am. A 29 (2012) 994–1002.
[https://doi.org/10.1364/JOSAA.29.000994]

-
H. Yockell-Lelièvre, F. Lussier, J.F. Masson, Influence of the Particle Shape and Density of Self-Assembled Gold Nanoparticle Sensors on LSPR and SERS, J. Phys. Chem. C 119 (2015) 28577–28585.
[https://doi.org/10.1021/acs.jpcc.5b09570]

-
Y. Yao, F. Ji, M. Yin, X. Ren, Q. Ma, J. Yan, et al., Ag Nanoparticle-Sensitized WO3 Hollow Nanosphere for Localized Surface Plasmon Enhanced Gas Sensors, ACS Appl. Mater. Interfaces 8 (2016) 18165–18172.
[https://doi.org/10.1021/acsami.6b04692]

-
I. Darmadi, S.Z. Khairunnisa, D. Tomeček, C. Langhammer, Optimization of the Composition of PdAuCu Ternary Alloy Nanoparticles for Plasmonic Hydrogen Sensing, ACS Appl. Nano Mater. 4 (2021) 8716–8722.
[https://doi.org/10.1021/acsanm.1c01242]

-
A. Campion, P. Kambhampati, Surface-enhanced Raman scattering, Chem. Soc. Rev. 27 (1998) 241–250.
[https://doi.org/10.1039/a827241z]

-
J. Kneipp, H. Kneipp, K. Kneipp, SERS—a single-molecule and nanoscale tool for bioanalytics, Chem. Soc. Rev. 37 (2008) 1052–1060.
[https://doi.org/10.1039/b708459p]

-
J.R. Lombardi, R.L. Birke, A Unified View of Surface-Enhanced Raman Scattering, Acc. Chem. Res. 42 (2009) 734–742.
[https://doi.org/10.1021/ar800249y]

-
R.D. Rodriguez, C.J. Villagomez, A. Khodadadi, S. Kupfer, A. Averkiev, L. Dedelaite, et al., Chemical Enhancement vs Molecule–Substrate Geometry in Plasmon-Enhanced Spectroscopy, ACS Photonics 8 (2021) 2243–2255.
[https://doi.org/10.1021/acsphotonics.1c00001]

-
O. Guselnikova, H. Lim, H.J. Kim, S.H. Kim, A. Gorbunova, M. Eguchi, et al., New Trends in Nanoarchitectured SERS Substrates: Nanospaces, 2D Materials, and Organic Heterostructures, Small 18 (2022) 2107182.
[https://doi.org/10.1002/smll.202107182]

-
C. Fu, Y. Li, X. Lei, J. Su, Y. Chen, Y. Wu, et al., SERS Sensor for Acetylcholine Detection Based on Covalent Organic Framework Hybridized Gold Nanoparticles As Nanozymes, Anal. Chem. 96 (2024) 18585–18589.
[https://doi.org/10.1021/acs.analchem.4c02471]

-
Y. Zhou, Q. Gu, T. Qiu, X. He, J. Chen, R. Qi, et al., Ultrasensitive Sensing of Volatile Organic Compounds Using a Cu-Doped SnO2-NiO p-n Heterostructure That Shows Significant Raman Enhancement, Angew. Chem. 133 (2021) 26464–26471.
[https://doi.org/10.1002/ange.202112367]

-
S. Singh, P.K. Singh, A. Umar, P. Lohia, H. Albargi, L. Castañeda, et al., 2D Nanomaterial-Based Surface Plasmon Resonance Sensors for Biosensing Applications, Micro-machines 11 (2020) 779.
[https://doi.org/10.3390/mi11080779]

-
C. Chen, K. Wang, L. Luo, AuNPs and 2D functional nanomaterial-assisted SPR development for the cancer detection: a critical review, Cancer Nanotechnol. 13 (2022) 29.
[https://doi.org/10.1186/s12645-022-00138-7]

-
H.-L. Hou, C. Anichini, P. Samorì, A. Criado, M. Prato, 2D Van der Waals Heterostructures for Chemical Sensing, Adv. Funct. Mater. 32 (2022) 2207065.
[https://doi.org/10.1002/adfm.202207065]

-
M.O. Li, D. Esseni, J.J. Nahas, D. Jena, H.G. Xing, Two-Dimensional Heterojunction Interlayer Tunneling Field Effect Transistors (Thin-TFETs), IEEE J. Electron Devices Soc. 3 (2015) 200–207.
[https://doi.org/10.1109/JEDS.2015.2390643]

-
D.Y.H. Ho, I. Yudhistira, B.Y.-K. Hu, S. Adam, Theory of Coulomb drag in spatially inhomogeneous 2D materials, Commun. Phys. 1 (2018) 41.
[https://doi.org/10.1038/s42005-018-0039-y]

-
Y. S. Cho, D. Rhee, J. Lee, S. Y. Jung, J. Eom, V. Mazanek, et al., Electronic and electrocatalytic applications based on solution-processed two-dimensional platinum diselenide with thickness-dependent electronic properties, EcoMat 5 (2023) e12358.
[https://doi.org/10.1002/eom2.12358]

-
J. Kang, J.D. Wood, S.A. Wells, J.-H. Lee, X. Liu, K.-S. Chen, et al., Solvent Exfoliation of Electronic-Grade, Two-Dimensional Black Phosphorus, ACS Nano 9 (2015) 3596–3604.
[https://doi.org/10.1021/acsnano.5b01143]

-
Z. Dai, L. Liu, Z. Zhang, Strain Engineering of 2D Materials: Issues and Opportunities at the Interface, Adv. Mater. 31 (2019) 1805417.
[https://doi.org/10.1002/adma.201805417]

-
D. Wang, X.-B. Li, H.-B. Sun, Modulation Doping: A Strategy for 2D Materials Electronics, Nano Lett. 21 (2021) 6298–6303.
[https://doi.org/10.1021/acs.nanolett.1c02192]

-
J. Kim, M. Jung, D.U. Lim, D. Rhee, S.H. Jung, H.K. Cho, et al., Area-Selective Chemical Doping on Solution-Processed MoS2 Thin-Film for Multi-Valued Logic Gates, Nano Lett. 22 (2022) 570–577.
[https://doi.org/10.1021/acs.nanolett.1c02947]

-
S.-L. Li, K. Tsukagoshi, E. Orgiu, P. Samorì, Charge transport and mobility engineering in two-dimensional transition metal chalcogenide semiconductors, Chem. Soc. Rev. 45 (2016) 118–151.
[https://doi.org/10.1039/C5CS00517E]

-
J. Jiang, T. Xu, J. Lu, L. Sun, Z. Ni, Defect Engineering in 2D Materials: Precise Manipulation and Improved Functionalities, Research 2019 (2019) 4641739.
[https://doi.org/10.34133/2019/4641739]

-
D. Lembke, A. Allain, A. Kis, Thickness-dependent mobility in two-dimensional MoS2 transistors, Nanoscale 7 (2015) 6255–6260.
[https://doi.org/10.1039/C4NR06331G]

-
C. Hsu, R. Frisenda, R. Schmidt, A. Arora, S.M. De Vasconcellos, R. Bratschitsch, et al., Thickness-Dependent Refractive Index of 1L, 2L, and 3L MoS2, MoSe2, WS2, and WSe2, Adv. Opt. Mater. 7 (2019) 1900239.
[https://doi.org/10.1002/adom.201900239]

-
P. Behera, S. Karunakaran, J. Sahoo, P. Bhatt, S. Rana, M. De, Ligand Exchange on MoS2 Nanosheets: Applications in Array-Based Sensing and Drug Delivery, ACS Nano 17 (2023) 1000–1011.
[https://doi.org/10.1021/acsnano.2c06994]

-
Y.S. Cho, D. Rhee, J. Eom, J. Kim, M. Jung, Y. Son, et al., Scalable Synthesis of Pt Nanoflowers on Solution-Processed MoS2 Thin Film for Efficient Hydrogen Evolution Reaction, Small Sci. 2 (2022) 2200043.
[https://doi.org/10.1002/smsc.202270018]

-
D. Sarkar, X. Xie, J. Kang, H. Zhang, W. Liu, J. Navarrete, et al., Functionalization of Transition Metal Dichalcogenides with Metallic Nanoparticles: Implications for Doping and Gas-Sensing, Nano Lett. 15 (2015) 2852–2862.
[https://doi.org/10.1021/nl504454u]

-
J. Eom, Y. S. Cho, J. Lee, J. W. Heo, I. Plutnarová, Z. Sofer, et al., Scalable 2D Semiconductor-Based van der Waals Heterostructure Interface with Built-in Electric Field for Enhanced Electrochemical Water Splitting, Small Struct. 5 (2024) 2400257.
[https://doi.org/10.1002/sstr.202400257]

-
Z. Lin, Y. Liu, U. Halim, M. Ding, Y. Liu, Y. Wang, et al., Solution-processable 2D semiconductors for high-performance large-area electronics, Nature 562 (2018) 254–258.
[https://doi.org/10.1038/s41586-018-0574-4]

-
O. Song, D. Rhee, J. Kim, M. Jung, S. Kim, I. S. Kim, et al., Solution-Processed 2D Transition Metal Dichalcogenide Networks for Scalable, Flexible Photosynaptic Device Arrays, IEEE J. Sel. Top. Quantum Electron. 30 (2024) 1–8.
[https://doi.org/10.1109/JSTQE.2023.3307515]

-
D. Rhee, B. Han, M. Jung, J. Kim, O. Song, J. Kang, Hierarchical Nanoscale Structuring of Solution-Processed 2D van der Waals Networks for Wafer-Scale, Stretchable Electronics, ACS Appl. Mater. Interfaces 14 (2022) 57153–57164.
[https://doi.org/10.1021/acsami.2c16738]

-
D. Rhee, D. Jariwala, J.H. Cho, J. Kang, Solution-processed 2D van der Waals networks: Fabrication strategies, properties, and scalable device applications, Appl. Phys. Rev. 11 (2024) 021310.
[https://doi.org/10.1063/5.0205192]

-
Y.A. Kwon, J. Kim, S.B. Jo, D.G. Roe, D. Rhee, Y. Song, et al., Wafer-scale transistor arrays fabricated using slot-die printing of molybdenum disulfide and sodium-embedded alumina, Nat. Electron. 6 (2023) 443–450.
[https://doi.org/10.1038/s41928-023-00971-7]

-
M. Sebek, N.T.K. Thanh, X. Su, J. Teng, A Genetic Algorithm for Universal Optimization of Ultrasensitive Surface Plasmon Resonance Sensors with 2D Materials, ACS Omega 8 (2023) 20792–20800.
[https://doi.org/10.1021/acsomega.3c01387]

-
J. Zhou, T. Yang, J. Chen, C. Wang, H. Zhang, Y. Shao, Two-dimensional nanomaterial-based plasmonic sensing applications: Advances and challenges, Coord. Chem. Rev. 410 (2020) 213218.
[https://doi.org/10.1016/j.ccr.2020.213218]

-
A.S. Chowdhury, M.A. Islam, M.S. Islam, B. Dey, J. Park, Design and Analysis of PtSe2 and Blue Phosphorus/MoS2 Heterostructure-Based SPR Biosensor, ACS Appl. Opt. Mater. 2 (2024) 1046–1059.
[https://doi.org/10.1021/acsaom.4c00050]

-
N. Liu, S. Wang, Q. Cheng, B. Pang, J. Lv, High Sensitivity in Ni-Based SPR Sensor of Blue Phosphorene/Transition Metal Dichalcogenides Hybrid Nanostructure, Plasmonics 16 (2021) 1567–1576.
[https://doi.org/10.1007/s11468-021-01421-w]

-
C. Yue, Y. Ding, L. Tao, S. Zhou, Y. Guo, Differential Evolution Particle Swarm Optimization for Phase-Sensitivity Enhancement of Surface Plasmon Resonance Gas Sensor Based on MXene and Blue Phosphorene/Transition Metal Dichalcogenide Hybrid Structure, Sensors 23 (2023) 8401.
[https://doi.org/10.3390/s23208401]

-
K.M. Ishtiak, S.-A. Imam, Q.D.M. Khosru, BaTiO3 - Blue Phosphorus/WS2 hybrid structure-based surface plasmon resonance biosensor with enhanced sensor performance for rapid bacterial detection, Results Eng. 16 (2022) 100698.
[https://doi.org/10.1016/j.rineng.2022.100698]

-
Z. Ashkavand, E. Sadeghi, R. Parvizi, M. Zare, Developed Low-Temperature Anionic 2H-MoS2/Au Sensing Layer Coated Optical Fiber Gas Sensor, ACS Appl. Mater. Interfaces 12 (2020) 34283–34296.
[https://doi.org/10.1021/acsami.0c05108]

-
P. Topolovsek, L. Cmok, C. Gadermaier, M. Borovsak, J. Kovac, A. Mrzel, Thiol click chemistry on gold-decorated MoS2: elastomer composites and structural phase transitions, Nanoscale 8 (2016) 10016–10020.
[https://doi.org/10.1039/C6NR01490A]

-
X. Ling, W. Fang, Y.-H. Lee, P.T. Araujo, X. Zhang, J.F. Rodriguez-Nieva, et al., Raman Enhancement Effect on Two-Dimensional Layered Materials: Graphene, h-BN and MoS2, Nano Lett. 14 (2014) 3033–3040.
[https://doi.org/10.1021/nl404610c]

-
Y. Chen, H. Liu, X. Li, S. Tang, C. Gu, G. Wei, et al., Development of RGO@MoS2@Ag ternary nanocomposites with tunable geometry structure for recyclable SERS detection, Sens. Actuators B Chem. 339 (2021) 129856.
[https://doi.org/10.1016/j.snb.2021.129856]

-
X. Ling, J. Wu, W. Xu, J. Zhang, Probing the Effect of Molecular Orientation on the Intensity of Chemical Enhancement Using Graphene-Enhanced Raman Spectroscopy, Small 8 (2012) 1365–1372.
[https://doi.org/10.1002/smll.201102223]

-
H. Xu, Y. Chen, W. Xu, H. Zhang, J. Kong, M.S. Dresselhaus, et al., Modulating the Charge-Transfer Enhancement in GERS using an Electrical Field under Vacuum and an n/p-Doping Atmosphere, Small 7 (2011) 2945–2952.
[https://doi.org/10.1002/smll.201100546]


Yun Seong Cho is a PhD student at the School of Advanced Materials Science & Engineering, Sungkyunkwan University (SKKU). His doctoral research has been supervised by Prof. Joohoon Kang, who is currently with the Department of Chemical and Biomolecular Engineering at Yonsei University. He received his BS degree in Advanced Materials Science & Engineering from SKKU in 2021. His research interests include the synthesis, processing, and electrochemical applications of nanomaterials.

Su Yeon Jung is a PhD student at the School of Advanced Materials Science & Engineering, Sungkyunkwan University (SKKU). Her doctoral research has been supervised by Prof. Joohoon Kang, who is currently with the Department of Chemical and Biomolecular Engineering at Yonsei University. She received her BS degree in the School of Chemical Engineering in 2022 from Ajou University. Her research interests include the synthesis, surface engineering, and photoelectrochemical applications of nanomaterials.

Prof. Joohoon Kang is an Associate Professor at the Department of Chemical and Biomolecular Engineering at Yonsei University. He received his BS and MS degrees in materials science and engineering from Yonsei University in Korea in 2009 and 2011, respectively, and his PhD degree in materials science and engineering from Northwestern University in 2018. He then moved to the University of California at Berkeley as a postdoctoral fellow in the College of Chemistry. He was an Assistant/Associate Professor in the School of Advanced Materials Science & Engineering at Sungkyunkwan University (SKKU) before he moved to Yonsei University in 2025. His research interests include the synthesis, processing, and (opto)electronic applications of nanomaterials. For more information, please visit http://mfmp.yonsei.ac.kr.