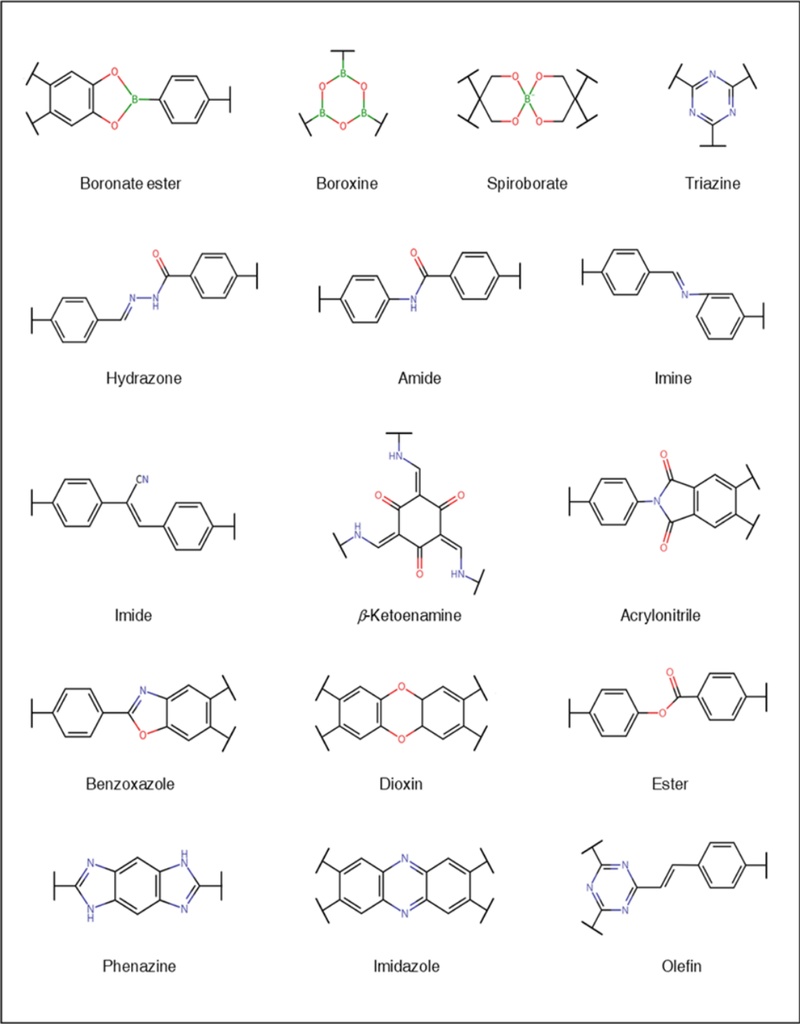
Synthetic Strategies and Linkage-Dependent Performance in Covalent Organic Frameworks-based Chemoresistive Gas Sensors
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Covalent organic frameworks (COFs) have garnered significant attention across diverse research fields due to their high specific surface area, intrinsic porosity, and tunable structural and chemical properties. In particular, COFs have emerged as promising materials for chemoresistive gas sensing, as their well-defined porous structures enable efficient surface interactions with analytes, facilitating sensitive and selective gas detection. This review summarizes recent advancements in COF-based chemoresistive gas sensors, outlining synthetic strategies for both powders and thin films, as well as representative sensing performances categorized by linkage chemistry. Additionally, proposed sensing mechanisms, including gas-framework interactions and charge transfer modulation, are discussed. Despite notable progress, several critical challenges remain, such as inherently low electrical conductivity, a limited range of target gases, and difficulties in scalable fabrication. Finally, we provide a brief outlook on potential strategies and future directions for developing practical, high-performance COF-based chemoresistive gas sensors.
Keywords:
COF, Chemoresistive gas sensor, 2D material, Porous material, COF fabrication, Sensitivity1. INTRODUCTION
The development of smart gas sensors has enabled the precise detection of airborne gases, providing alerts to ensure public safety and improve the quality of life. Initially, gas sensors were primarily used to monitor industrial pollutants and toxic gases [1,2]. However, their applications have since expanded to include everyday uses, such as food quality assessment, indoor air quality monitoring, and non-invasive disease diagnosis [3-5]. Chemoresistive gas sensors have been widely adopted for real-time gas monitoring because of their small size, rapid kinetics, and high sensitivity [6-8]. Metaloxide semiconductors (MOSs) are the most widely used materials for chemoresistive gas sensors because of their high thermal stability and catalytic activity for gas oxidation [9-11]. However, they typically require high operating temperatures and often exhibit poor selectivities. In recent years, two-dimensional (2D) materials such as transition metal dichalcogenides (TMDs), carbon-based materials, and metal–organic frameworks (MOFs) have emerged as attractive alternatives because of their large surface areas, mechanical flexibility, and abundant active sites [12-16]. Furthermore, as the demand for highly selective, low-temperature operation, and selective gas sensors has increased, researchers have focused on covalent organic frameworks (COFs) as a new class of crystalline porous materials with tunable chemistry and architecture.
COFs represent an emerging class of crystalline, porous organic polymers made from organic building blocks linked by strong covalent bonds. Since their discovery by Yaghi and colleagues in 2005, a wide range of COFs with tailored structural features and diverse linkage chemistries such as imines, triazines, and boronate esters, have been developed (Fig. 1) [17]. The geometry and symmetry of these building blocks determine the dimensionality and topology of the resulting frameworks, allowing precise control over their structural architecture. COFs offer a unique combination of properties, including high specific surface area, permanent porosity, thermal stability, low density, tunable pore size, and chemical functionality [18-20]. These characteristics make COFs promising materials for various applications, including catalysis [21,22], gas storage [23], batteries [24,25], and gas sensors [26,27]. In particular, 2D π-conjugated COFs have attracted increasing interest as platforms for chemoresistive gas sensing. Their layered structures facilitate both in-plane and interlayer π-electron delocalization, enhancing charge transport across the framework [28,29]. Combined with their porous nature, which allows rapid gas diffusion, conductive COFs hold significant potential as next-generation sensing materials, offering high sensitivity, selectivity, and operational versatility.
In this review, we comprehensively highlight recent advances in the development of COF-based chemoresistive gas sensors. First, the synthetic strategies for preparing COFs are discussed, covering both powder- and thin-film approaches, which are crucial for tuning their structural and electronic properties. Next, we review recent advances in COF-based chemoresistive gas sensors, highlighting representative studies that showcase exceptional sensitivity, selectivity, and reversible sensing behavior toward various target gases. The content follows a classification based on the types of bonding within the COF building blocks, illustrating their characteristic behaviors that may influence their gas-sensing properties. In these studies, the proposed gas-sensing mechanisms are also discussed. Finally, we conclude with a perspective on COF-based chemoresistive gas sensors, aiming to inspire further research on their practical applications.
2. FABRICATION METHODS OF COFs
2.1 Synthesis of COF powders
The solvothermal method is one of the most common approaches for the synthesis of well-ordered crystalline COF powders. Similar to the synthesis of inorganic zeolites and MOFs, the fabrication of COFs under solvothermal conditions typically requires sealed and pressurized vessels, extended reaction times (2–7 days), and elevated temperatures (80–130°C). Dalapati et al. prepared a Py-azine COF with high crystallinity, porosity, and chemical stability under solvothermal conditions [30]. The yellowish Py-Azine COF was obtained in 75% yield at 120°C for 7 days. Yang et al. first reported the fabrication of a naphthalimide-based COF using a solvothermal method [31]. Owing to their ordered nanoporous framework and robust stability, the resulting TFP-DANT COF and TFB-DANT COF show promising electrochemical performance as cathode materials for Li-ion batteries.
In ionothermal synthesis, ionic liquids simultaneously serve as both solvents and potential templates or structure-directing agents. Compared to the conventional solvothermal method, this approach eliminates the use of toxic solvents and catalysts, while significantly reducing the reaction time. In 2008, Thomas and colleagues synthesized a triazine-based COF using molten ZnCl2 as both a reactive high-temperature solvent and catalyst [32]. The nitrile monomers displayed good solubility in the ZnCl2 melt due to strong Lewis acid–base interactions, and ZnCl2 also facilitated the trimerization reaction. Similarly, Maschita et al. fabricated two crystalline and porous imine-based COFs (TAPB-PTCDA COF and TAPB-PMDA COF) using an ionothermal method [33]. Using pure ZnCl2 as the reaction medium, the fabrication of COFs was completed in less than 10 h, compared to 3–7 d required for the conventional solvothermal method. Moreover, they synthesized a perylene-based COF using a eutectic salt mixture as the reaction medium. They demonstrated that using a three-salt eutectic mixture with a melting point below 280°C facilitated the synthesis of COFs with less stable linkers, such as TAPA and TAPT.
Microwave heating has been explored for the synthesis of organic polymers with high yields, improved purities, and short fabrication time [34]. Cooper and co-workers prepared 2D COF-5 under microwave heating conditions [35]. COF-5 was obtained within 20 min, which is 200 times faster than conventional solvothermal methods, while exhibiting comparable physical properties. Moreover, Wei et al. reported a significant increase in the yield of 2D TFP-PDA COF with a reduced reaction time using the microwave heating method [36]. The resulting TFP-PDA COF powder was obtained with yields of up to 83% based on the starting materials, compared to only 8% yield achieved using the solvothermal method. The as-synthesized COF also demonstrated high crystallinity, good stability, a large surface area, and excellent CO2 uptake.
2.2 Synthesis of COF thin films
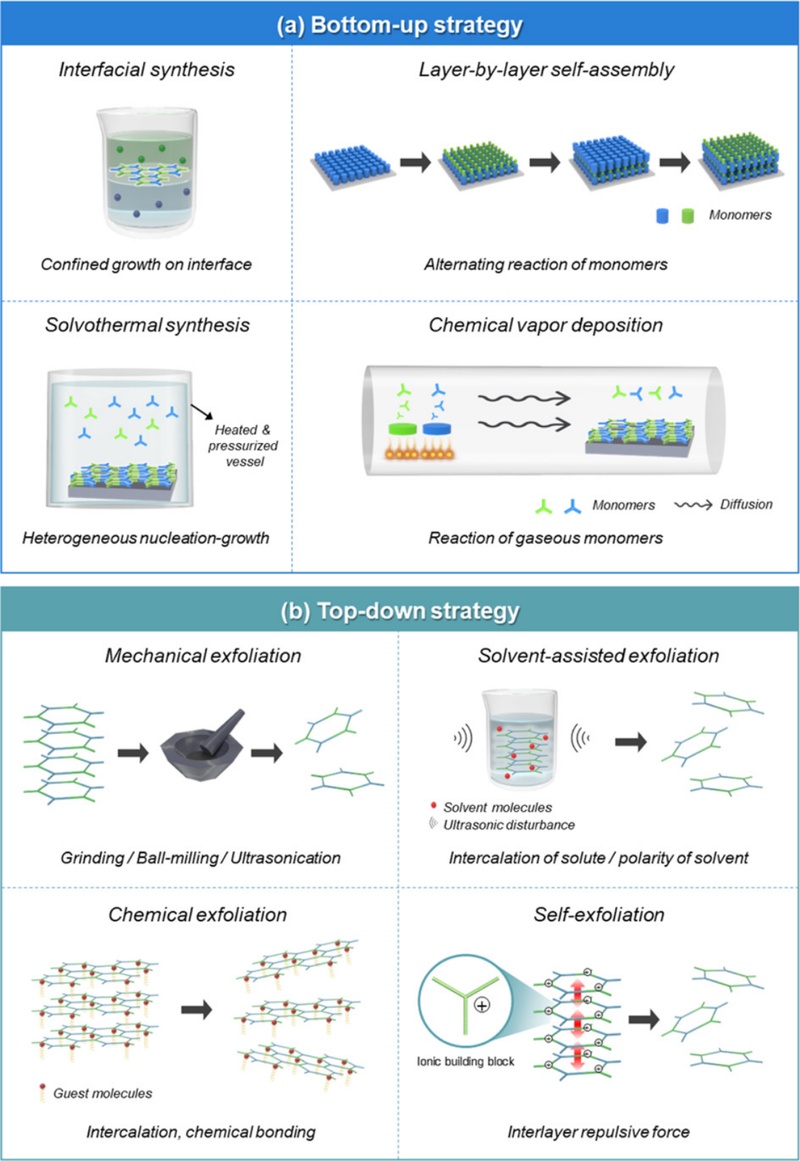
Through bottom-up synthesis, COFs can be directly synthesized at an interface or deposited onto a specific substrate with a controllable thickness and surface properties. During the fabrication process, the growth direction of the COF is influenced by the confined physical space in which the synthesis takes place. There are four common methods for preparing COF films: (1) interfacial polymerization, (2) synthesis on a substrate under solvothermal conditions, (3) layer-by-layer (LbL) self-assembly on a substrate, and (4) chemical vapor deposition (Fig. 2 (a)).
2.2.1.a. Interfacial synthesis
Interfacial synthesis is a widely used method for preparing COF thin films, in which monomers react at confined interfaces to promote controlled film growth. Various types of interfaces, such as liquid-air, liquid-liquid, and solid-vapor interfaces, can be employed, depending on the desired synthesis conditions [37-39]. For example, in liquid-air interfacial synthesis, Shinde et al. synthesized a crystalline TFP-DHF membrane at the water-air interface [40]. The TFP and DHF solutions were dissolved in toluene and gently spread onto the water surface. After the evaporation of toluene, the resulting layer was compressed, and polymerization was initiated by adding catalytic trifluoroacetic acid. The as-synthesized TFP-DHF thin film was formed at the interface and transferred using the Langmuir-Blodgett method. The volatilization of low-boiling-point organic solvents can create liquid-air interfaces. In liquid-liquid interfacial synthesis, each monomer is dissolved in immiscible solvents such as water-dichloromethane and water-ethyl acetate. Dey et al. reported the synthesis of a TFP-BPy thin film on a water-dichloromethane interface using a three-layered system: TFP dissolved in dichloromethane as the bottom layer, pure water as the middle layer, and BPy-PTSA salt in water as the topmost layer [41]. The intermediate water prevented the premature mixing of the precursors and promoted the slow diffusion of monomers, yielding a highly crystalline COF. In solid-vapor interfacial synthesis, one monomer is immobilized on a solid substrate, while the other is introduced in the vapor phase. This strategy promotes controlled COF growth solely at the confined interface and allows heating without disturbing the interface. For instance, Khan et al. fabricated highly crystalline and ultrathin TFP-PDA membranes by spin coating TFP onto a Si/SiO2 substrate followed by exposure to PDA vapor at 150°C [42]. The elevated temperature substantially reduced the reaction time and ensured sufficient PDA vapor pressure, thereby enhancing membrane quality.
2.2.1.b. Solvothermal synthesis
The in-situ solvothermal approach facilitated the fabrication of highly crystalline, uniform, and dense COF structures with fewer defects. Colson et al. synthesized 2D COF films on single-layer graphene under simple solvothermal conditions [43]. The as-synthesized COF films were stacked perpendicular to the single-layer graphene surface with a well-oriented structure and exhibited enhanced crystallinity compared to COF powders. Medina et al. fabricated BDT COF on different metal oxide substrates including indium tin oxide (ITO) glass and NiO-modified ITO glass [44]. The as-synthesized BDT COF films exhibited a crack-free and uniform structure with a thickness of approximately 150 nm and good adherence to the ITO surface. Sun et al. synthesized a TFPPy-PDA COF film on single-layer graphene under optimized solvothermal conditions [45]. The resulting TFPPy-PDA COF exhibited a well-defined structure and high crystallinity, making it a suitable semiconducting layer for vertical field-effect transistor devices.
2.2.1.c. Layer-by-layer self-assembly
LbL self-assembly is a convenient method of membrane fabrication that involves alternating reactions between two or more precursors on a substrate surface. Compared to conventional coating methods, LbL self-assembly offers distinct advantages, such as simplicity of the fabrication process and precise control over the film thickness [46]. Selection of an appropriate substrate surface is crucial for initiating and sustaining sequential polymerization reactions. In 2018, Shi et al. reported the fabrication of a crystalline TFP-BD COF on a porous hydrolyzed polyacrylonitrile (HPAN) membrane using the LbL process [47]. The abundant surface carboxyl groups on HPAN facilitate the anchoring of amine monomers, enabling subsequent COF growth by alternating exposure to ethanolic solutions of amine and aldehyde monomers. The thickness of the TFP-BD layer was controlled by simply adjusting the number of LbL cycles.
2.2.1.d. Chemical vapor deposition
Chemical vapor deposition (CVD) offers distinctive advantages, including simplicity of fabrication and a fast reaction rate. Because it does not require organic solvents to dissolve the monomer reactants, it is environmentally friendly. Synthetic conditions such as temperature, reaction time, pore size of the basement membrane, and pressure determine the crystallinity, thickness, and quality of the COF films. In 2021, Hao et al. reported the fabrication of a TFP-PDA-1 COF membrane via chemical vapor deposition [48]. The authors systematically elucidated the effects of the evaporation temperature, evaporation time, amount of Fe antioxidant, and pore size of the basement substrate on the quality of TFP-PDA-1.
Thin-film fabrication via a top-down approach relies on the exfoliation of bulk COFs into 2D nanosheets (NSs), which can subsequently be assembled into continuous films. These bulk COFs comprise periodically stacked layers assembled through weak intermolecular forces, such as van der Waals interactions, π–π stacking, and hydrogen bonding. Upon the application of an external force, these layered structures can be delaminated into single- or few-layered NSs (Fig. 2 (b)). This top-down strategy offers an efficient, scalable, and cost-effective route for producing highly crystalline COF films.
2.2.2.a. Mechanical exfoliation
External mechanical forces can be applied by ball milling, grinding, ultrasonication, or tape-assisted exfoliation. In 2013, Banerjee et al. demonstrated mechanical delamination of bulk COFs to produce a series of covalent organic nanosheets (CONs) [49]. They synthesized eight types of COFs (TFP-PDA-1, TFP-PDA-2, TFP-BD, TFP-PDA-NO2, TFP-PDA-F4, TFP-BD-(NO2)2, TFP-BD-Me2, and TFP-BD-(OMe)2) and successfully exfoliated them via a simple and safe mechanical grinding method. Since individual layers are stacked by relatively weak π-π interactions and possess inherent chemical stability, mild mechanical forces are sufficient to exfoliate them. Wang et al. demonstrated the exfoliation of a 2D COF into few-layered NSs with abundant redox acid sites for Li-ion battery applications [50]. To shorten the Li-ion transport pathway, a simple ball milling process was used to reduce the COF thickness. The micron-sized DAAQ-TFP COF was ball-milled at room temperature without the use of additional exfoliating agents, resulting in thinner, sheet-like delaminated CONs.
2.2.2.b. Solvent assisted exfoliation
Solvent molecules can intercalate between COF layers, weakening π-π interactions and facilitating exfoliation. This process is typically enhanced by sonication, which further aids in the separation of individual layers. Berlanga et al. reported the delamination of COF-8 into 10–20 layers by sonication in dichloromethane, which was chosen for its volatility, low water content, and high purity [51]. Similarly, Bunck, et al. emonstrated the solvent-induced exfoliation of COF-43 by immersion in tetrahydrofuran, trichloromethane, toluene, and methanol [52]. Although X-ray diffraction (XRD) analysis revealed a loss of crystallinity, Fourier-transform infrared (FTIR) and in-situ IR spectra confirmed that no new chemical species were formed during soaking, indicating that the observed structural changes originated from physical delamination rather than chemical decomposition. These findings demonstrate that solvent-assisted exfoliation can effectively yield few-layered CONs, while preserving the intrinsic chemical structure of the framework.
2.2.2.c. Chemical exfoliation
Chemical exfoliation utilizes specific chemical reactions, such as oxidation, reduction, or intercalation, to weaken the interlayer interactions and promote delamination. Banerjee et al. demonstrated the chemical exfoliation of an anthracene-based COF (DA-TFP) to yield CONs with tunable thicknesses and high dispersibility [53]. A bulk DA-TFP COF was synthesized via a solvothermal reaction, and exfoliation was then induced by incorporating N-hexylmaleimide molecules into the anthracene moieties of the COF through a Diels–Alder cycloaddition. This incorporation disrupted the π–π interactions between layers, facilitating exfoliation. The chemically exfoliated NSs were subsequently assembled onto silicon substrates via air-water interfacial LbL deposition, enabling the fabrication of uniform, thickness-tunable films. Similarly, Chen et al. chemically exfoliated a 2D TFPB-PDA COF through oxidative intercalation using HClO4 and KMnO4 [54]. The in-situ formation of MnO2 nanoparticles effectively inhibited the restacking of the exfoliated NSs, thereby improving access to redox-active sites and enhancing the Liion storage performance.
2.2.2.d. Self-exfoliation
Self-exfoliation refers to the spontaneous delamination of layered materials driven by internal forces or molecular interactions without external energy or chemical treatment. In particular, ionic COFs have shown potential for the direct synthesis of CONs without external treatment. For example, Mitra et al. reported the self-exfoliation of guanidinium halide ionic CONs [55]. The inherent positive charge of guanidinium induced electrostatic repulsion, overcoming the π–π interactions between layers. Similarly, Zhang et al. synthesized an ionic COF via a one-pot method, which subsequently self-exfoliated upon dispersion in polar solvents (dioxane and mesitylene) [56]. In this system, BF3·OEt2, a Lewis acid catalyst, not only facilitated the formation of boron-containing COFs but also coordinated with nitrogen atoms to form organic ion pairs. The incorporation of ionic moieties played a crucial role in enabling self-exfoliation, and the resulting nanosheets were successfully assembled into uniform thin films on SiO2 substrates.
3. COF-BASED CHEMORESISTORS
3.1 Imine-linked COF chemoresistors
Imine-linked COFs are the most extensively explored class of COFs owing to their high crystallinity, well-defined porosity, and superior structural regularity. Compared with boron-containing COFs, imine-linked COFs exhibit superior chemical stability in a wide range of organic solvents and water. In 2009, Yaghi and co-workers first reported a 3D crystalline porous imine-linked COF (COF-300) synthesized via the condensation between tetrahedral tetra-(4-aniyl)-methane and linear terephthaldehyde [57]. Since then, numerous imine-linked COFs have been developed via the condensation of aldehydes with amines or hydrazides, with growing interest in their electrochemical applications, including chemoresistive sensors. For instance, Niu et al. prepared an imine-linked COF by condensing TAPB and PBDA for NH3 detection (Fig. 3 (a)) [58]. The resulting TAPB-PBDA COF showed a notable response to NH3 (SNH3 = 23% to 100 ppm) at room temperature, along with rapid response/recovery times (10/106 s) and ultra-high selectivity (Figs. 3 (b) and 3 (c)). These distinct NH3 sensing properties were attributed to the strong hydrogen bonding between the imine N and NH3. The authors also found that structurally analogous imine-linked COFs, such as TFB-DAB COF, TAPB-PDA COF, and TAPB-TPDA COF, exhibited similar NH3 sensing behavior. Various post-treatments have been investigated to improve the gas-sensing properties. Liu et al. reported the metallization of a TAPP-PCBA COF with Co ions on the porphyrin rings using a heat-assisted reflux method (Fig. 3 (d)) [59]. The introduction of Co substantially enhanced the response to NO2, as Co-TAPP-PCBA COF exhibited approximately twice the response (SNO2 = 2,713 to 100 ppm), about 1.5 times shorter response time (5.3 min), and nearly 180 times lower detection limit (6.8 ppb) compared to the pristine TAPP-PCBA COF (Fig. 3 (e)). These improvements were ascribed to the strong interaction between coporphyrin and NO2 molecules, confirmed by in-situ diffuse reflectance infrared Fourier transform (DRIFT) spectroscopy and density functional theory (DFT) calculations (Fig. 3 (f)). Moreover, Xu and co-workers reported a facile one-pot post-treatment to generate dangling bonds (–NH2) on a Zn-TAPP-DFP COF for sensitive NO2 detection [60]. Zn metallization induced the elongation of the imine linkage, which was easily broken by the subsequent hydrolysis reaction, forming dangling bonds (Fig. 3 (g)). The resulting Zn-TAPP-DFP COF exhibited a high NO2 response (SNO2 = 543 to 100 ppm) under visible-light irradiation, with an ultra-low detection limit of 7.9 ppb and high selectivity against interfering gases such as NH3 and SO2 (Fig. 3 (h)). From the DFT calculations and in-situ FTIR, the authors revealed that dangling –NH2 groups were the functional sites for NO2 adsorption, resulting in distinct NO2 sensing performance (Fig. 3 (i)).
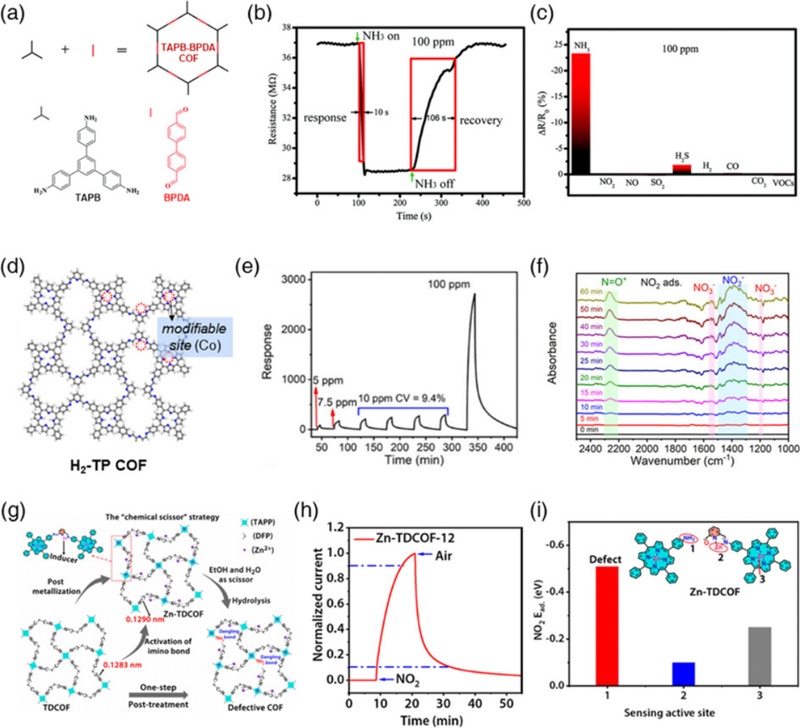
Gas sensing performance of imine-linked COFs. (a) Structure of TAPB-BPDA COF. (b) Dynamic sensing transient of TAPB-BPDA COF to 100 ppm NH3. (c) Selectivity of TAPB-BPDA COF over various interfering gases. Reprinted with permission from Ref. [58], Copyright (2021) Royal Society of Chemistry. (d) Structure of H2-TP COF. (e) Dynamic response transient of Co-TP COF toward 5–100 ppm NO2. (f) In-situ DRIFT spectra of Co-TP COF during NO2 adsorption. Reprinted with permission from Ref. [59], Copyright (2022) Wiley-VCH. (g) Schematic illustration of defective COF synthesis. (h) Dynamic response transient of Zn-TD COF to 100 ppm NO2. (i) NO2 adsorption energy of Zn-TD COF on different sites. Reprinted with permission from Ref. [60], Copyright (2023) Royal Society of Chemistry.
3.2 β-ketoenamine-linked COF chemoresistors
The β-ketoenamine-linked COFs are formed via an irreversible enol-to-keto tautomerization from imine-linked COFs. This irreversible tautomerism imparts exceptional chemical and thermal stability to the framework and enhances its electronic conductivity through extended π-electron delocalization [61,62]. These structural and electronic features make β-ketoenamine COFs promising candidates for chemoresistive gas-sensing applications. To investigate the influence of linkage type on sensing performance, Yan et al. prepared two isostructural COFs, β-keto-An COF and imine-An COF, that share identical backbones but differ in their linkages (Fig. 4 (a)) [63]. The I-V curve revealed that β-keto-An COF exhibited higher conductivity than imine-An COF, attributed to the extended intralayer π–π conjugation and reduced interlayer stacking distance due to the carbonyl group (–C=O) within the β-ketoenamine linkage (Fig. 4 (b)). The β-keto-An COF also demonstrated better thermal and chemical robustness, owing to the irreversible nature of its tautomerism. Notably, the β-keto-An COF showed a significantly higher NH3 response (SNH3 = 189.4% to 10 ppm), which was approximately 32 times higher than that of imine-An COF, along with high selectivity (response ratio ≥7.33) (Fig. 4 (c)). The superior sensing performance was attributed to strong hydrogen bonding interactions between NH3 and the carbonyl group within the β-ketoenamine linkage, as supported by DRIFT and DFT calculations. These results demonstrate that β-ketoenamine-linked COFs are favorable sensing materials for NH3 and NO2.
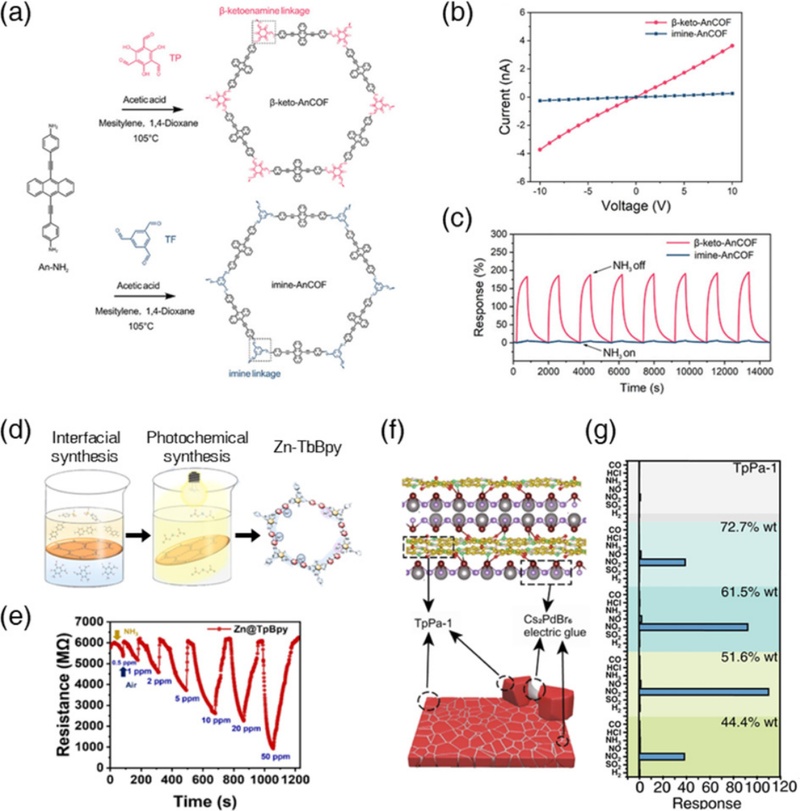
Gas sensing performance of β-ketoenamine-linked COFs. (a) Synthetic routs for β-keto-An COF and imine-An COF. (b) I-V curves of β-keto-An COF and imine-An COF. (c) Repetitive response curve of β-keto-An COF and imine-An COF to 10 ppm NH3. Reprinted with permission from Ref. [63], Copyright (2021) Royal Society of Chemistry. (d) Structure of H2-TP COF. (e) Dynamic response transients of Co-TP COF toward 5–100 ppm NO2. Reprinted with permission from Ref. [64], Copyright (2022) Wiley-VCH. (f) Theoretical models of the combination of Pd exposed by surface detects of perovskites and N and O in TpPa-1. (g) Gas response of TaPa-1 and TaPa-1/Cs2PdBr6. Reprinted with permission from Ref. [27], Copyright (2023) Springer Nature.
Despite their relatively high intrinsic conductivity compared to other COF linkages β-ketoenamine-linked COFs still suffer from low absolute conductivity compared to traditional semiconducting chemiresistors such as metal oxides or carbon-based materials. This often challenges resistance measurements using conventional and cost-effective electrical circuits. To address this, post-synthetic metalation strategies have been explored, utilizing the functional groups of β-ketoenamine-linkages as coordination sites to simultaneously enhance charge transport and gas adsorption. For example, Mei et al. fabricated a Zn-coordinated TFP-BPy COF via interfacial and photochemical methods, where Zn2+ was loaded onto both β-ketoenamine and BPy sites (Fig. 4 (d)) [64]. The resulting Zn-TFP-BPy COF exhibited a substantial decrease in resistance (~6 GΩ), a significantly enhanced NH3 response (SNH3 = 1,043% to 50 ppm), and fast response/recovery times (55/111 s), while the pristine TFP-BPy COF showed negligible gas response and extremely high resistance (>100 GΩ) (Fig. 4 (e)). The authors revealed that Zn2+ coordination at the BPy site contributes to an electron-donating structure, significantly enhancing electron density in the n-type semiconducting TFP-BPy COFs. Meanwhile, the Zn2+ coordination at β-ketoenamine linkage sites leads to stronger and more reversible NH3 adsorption. Ye et al. reported the integration of a TFP-PDA-1 COF with a halide perovskite (Cs2PdBr6) to improve its electrical conductivity and activate NO2 sensing [27]. The Cs2PdBr6 acted as electronic glue, forming Pd4+ coordination bonds with both carbonyl O and imine N in the β-ketoenamine linkage (Fig. 4 (f)). This coordination significantly reduced the boundary resistance between the COF crystallites, lowering the overall sensor resistance by two orders of magnitude. The TFP-PDA-1/Cs2PdBr6 COF exhibited a significantly high NO2 response (SNO2 = 110.3 to 2 ppm) and outstanding selectivity over interfering gases such as NO and SO2 (response ratio >70), whereas pristine TFP-PDA-1 showed negligible gas responses (Fig. 4 (g)).
3.3 Triazine-linked COF chemoresistors
Covalent triazine-based frameworks (CTFs), first reported by Kuhn et al. in 2008, feature a well-defined architecture composed of aromatic C=N linkages (triazine units) and offer excellent chemical stability and high nitrogen content [32]. Tao et al. demonstrated the room-temperature NH3 sensing capability of CTFs (CTF-A-1) synthesized via the ionothermal trimerization of 1,4-dicyanobenzene (Fig. 5 (a)) [65]. The developed sensor exhibited high sensitivity (SNH3 = ~4.17% to 1 ppm) and reversible behavior, with response/recovery times of ~100/~420 s (Figs. 5 (b) and 5 (c)). The sensing ability was attributed to strong n-π interactions between the lone pair electrons of NH3 and the π-conjugated, electron-deficient triazine rings, which promote partial charge transfer to the framework. Similarly, Niu et al. prepared a series of P2O5-catalyzed CTFs (pCTFs) and evaluated their NH3 sensing performance at room temperature [66]. As the condensation temperature of 1,4-dicyanobenzene increased, the nitrogen content and triazine ring density decreased, resulting in a diminished NH3 response, thereby highlighting the crucial role of triazine units in NH3 detection (Fig. 5 (d)). This trend was consistently observed when the monomers 1,4-dicyanobenzene (for CTF and pCTF) and 1,3,5-benzene tricarboxamide (for pCTF-3) were varied to modulate the triazine ring density (Figs. 5 (e) and 5 (f)). DFT calculations further supported this mechanism by confirming that NH3 molecules preferentially adsorb onto triazine rings, donating electrons and narrowing the bandgap, which explains the observed decrease in resistance. Triazine-based frameworks have also shown selective detection of NO2. Yang et al. exfoliated a CTF synthesized via the triflic-acid-catalyzed cyclotrimerization of 1,4-dicyanobenzene into CONs (Fig. 5 (g)) [67]. The sensor exhibited the highest response toward NO2 (SNO2 = 505% to 1 ppm), while the responses to other gases were quite low (SNH3 = 32% and SH2S= 9.8% to 1 ppm) or negligible (Figs. 5 (h) and 5 (i)). The high sensitivity and selectivity toward NO2 are attributed to the intrinsic periodic pore structure of the 2D NSs, which facilitates gas diffusion and interaction with the active sites. Similarly, Niu et al. reported a viologen-based CTF that exhibited excellent NO2 sensing performance, with a 43% response to 50 ppm NO2 [68]. This superior sensing capability was attributed to the presence of abundant active sites within the viologen units, which promoted the effective adsorption of NO2 molecules.
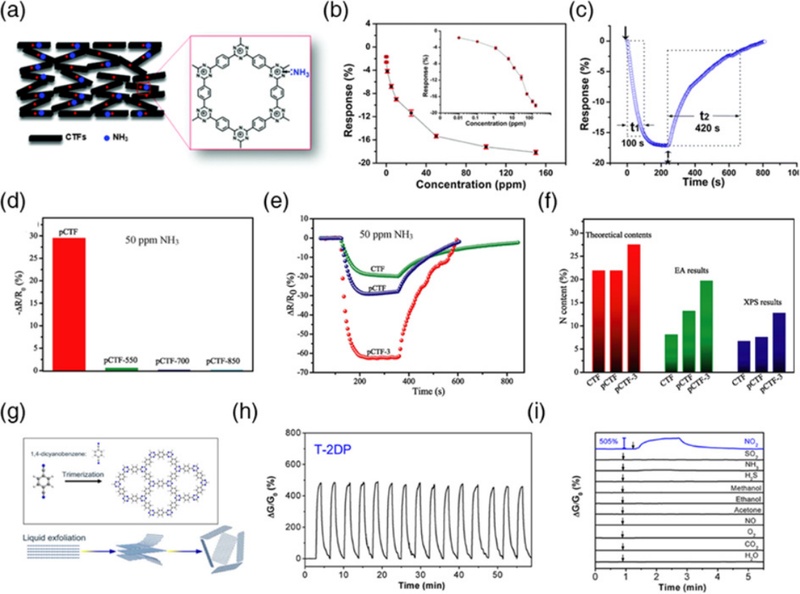
Gas sensing performance of triazine-linked COFs. (a) Structure of CTF-1-A COF. (b) Response of CTF-1-A COF toward varying concentrations of NH3. (c) Dynamic sensing response of CTF-1-A COF toward 100 ppm NH3. Reprinted with permission from Ref. [65], Copyright (2014) Royal Society of Chemistry. (d) Responses of various pCTFs sensors synthesized at different condensation temperatures to 50 ppm NH3. (e) Dynamic sensing responses toward 50 ppm NH3 and (f) calculated N contents of CTF, pCTF, and pCTF-3. Represented with permission from Ref. [66], Copyright (2020), Elsevier. (g) Structure of T-2DP COF. (h) Repetitive sensing response of T-2DP toward 1 ppm NO2. (i) Response of T-2DP toward 1 ppm of various analyte gases. Represented with permission from Ref. [67], Copyright (2019), American Chemical Society.
3.4 Others
Imide bond (O=C–N–C=O)-based COF, as demonstrated by Yan and co-workers, exhibited excellent thermal stability along with a high surface area and ordered porosity, making it a promising candidate for chemoresistive gas sensing [69]. Thokala et al. synthesized a redox-active COF via solvothermal imidization of TAPA and NTCDA, followed by exfoliation into CONs (Fig. 6 (a)) [70]. While the bulk NTCDA-TAPA COF showed a negligible response to H2, the NTCDA-TAPA CON exhibited a significant response of 30.7% (10,000 ppm H2), with response/recovery times of 4.5 and 3.9 s, respectively (Fig. 6 (b)). DFT calculations indicated that the carbonyl oxygens of the NDI moiety serve as favorable adsorption sites for H2 (binding energy = –2.2 kcal mol–1), while exfoliation improves gas accessibility and facilitates interaction with the active sites. Both factors synergistically contribute to the enhanced sensing performance (Fig. 6 (c)). Similarly, Gao et al. fabricated a freestanding TPTCDA-THSTZ membrane for the selective detection of trimethylamine (N(CH3)3, TMA). The sensor exhibited “a response of 3.47 to” 40 ppm TMA at room temperature, with response and recovery times of 96 and 63 s, respectively [71]. The sensor exhibited marked selectivity toward TMA, with a response 5.5–22 times higher than that of the other interfering gases. Insitu FTIR spectroscopy revealed that the sensing reaction relied on adsorbed oxygen species-mediated charge transfer facilitated by hydrogen bonding between the carbonyl oxygen of TPTCDA-THSTZ and the amino hydrogen of TMA.
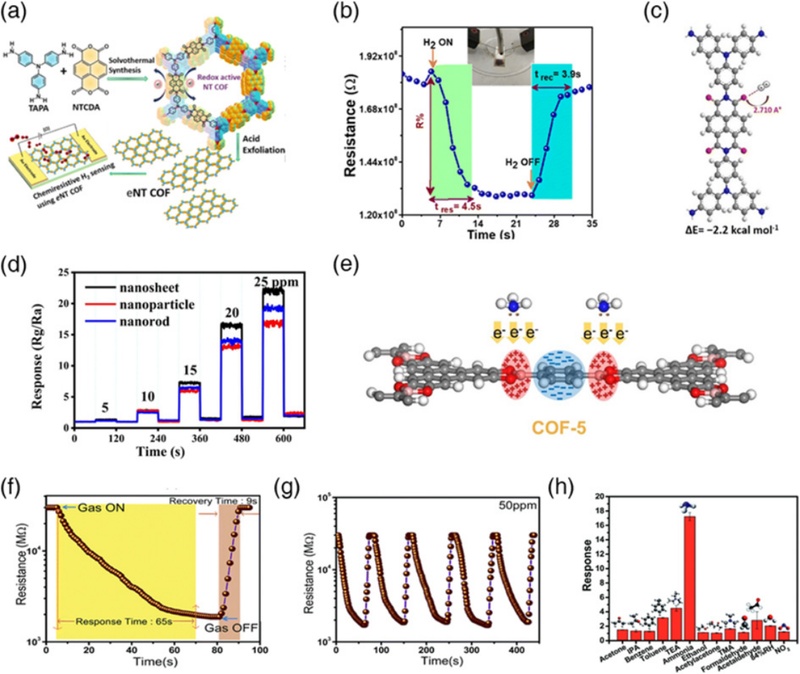
(a) Schematic illustration of the structure and fabrication process of NT-COF and NT-CON. (b) Dynamic H2 sensing transient of the NT-CON sensor. (c) Molecular electrostatic potential plot of the optimized binding site in NT-COF. Represented with permission from Ref. [71], Copyright (2023), Royal Society of Chemistry. (d) Response transients of COF-5 sensors with different morphologies toward various concentrations of NH3. (e) Proposed NH3 sensing mechanism of COF-5 NSs. Represented with permission from Ref. [73], Copyright (2023), Elsevier. (f) Response and recovery times of the HMP-TAPB-1 sensor exposed to 50 ppm NH3 at 47% RH. (g) Cyclic sensing transient of HMP-TAPB-1 sensor. (h) Response of HMP-TAPB-1 sensor to various analytes. Represented with permission from Ref. [74], Copyright (2018), Royal Society of Chemistry.
Boron-based linkages such as boronate esters and boroxines were among the first to be employed for constructing crystalline COFs because of their dynamic reversibility, enabling the formation of well-ordered structures despite their limited hydrolytic stability [17,72]. For instance, Yang et al. fabricated a series of COF-5-based NH3 sensors by modulating the nanostructure, nanoparticles, nanorods, and NSs via controlling the HHTP concentration, which governed the relative rates of lateral and longitudinal growth [73]. The nanosheets exhibited the highest sensitivity (SNH3 = 3.13 to 10 ppm) at 60°C, which attributed to their enlarged surface area and efficient gas accessibility (Fig. 6 (d)). The strong Lewis basicity of NH3 enables selective interactions with the boronate ester linkages, which serve as adsorption sites (Fig. 6 (e)).
Nitrogen-rich heterocyclic linkages other than triazine, such as heptazine (C6H3N7) and phenazine (C12H8N2), have also been incorporated into COFs, offering distinct electron-deficient frameworks favorable for chemoresistive gas sensing. Sharma et al. developed a heptazine-based COF (TCH-TAPB-1) for room-temperature NH3 sensing, achieving a high response of 16.64 to 50 ppm NH3 with fast response/recovery times (65/9 s) and selectivity exceeding 5.36 (Figs. 6 (f) and 6 (g)) [74]. The strong affinity toward NH3 was attributed to donor–acceptor interactions between the electron-rich NH3 and the electron-deficient heptazine moiety, along with the small molecular size of NH3, which enabled efficient diffusion into the porous structure of the COF. To enhance sensing performance, metal centers, such as Ni have been incorporated into COFs, enabling both redox activity and selective analyte coordination. Mirica and co-workers synthesized phenazine-linked COF-DC-8 from NiOAPc and TOPyr building blocks, which exhibited an outstanding chemoresistive performance toward various gases [26]. The sensor showed responses of SNH3 = 39%, SH2S = 62%, SNO = 3,939%, and SNO2 = −6,338% to 40 ppm, with corresponding limits of detection as low as 70 ppb (NH3), 204 ppb (H2S), 5 ppb (NO), and 16 ppb (NO2) (Fig. 6 (h)) Electron paramagnetic resonance (EPR) and X-ray photoelectron spectroscopy (XPS) revealed that the sensing behavior originated from charge transfer between the analytes and Ni-phthalocyanine units within COF-DC-8, involving weak and reversible interactions for NH3 and redox-driven processes in the case of H2S, NO, and NO2.
4. SUMMARY AND OUTLOOK
Recent advances in the development of robust, stable, and highly porous COFs, coupled with progress in chemical modification and a fundamental understanding of their physical and chemical properties, have paved the way for their application in chemoresistive gas sensing. The distinct characteristics of COFs, including their highly ordered porous structures and large surface areas, facilitate efficient surface reactions with the analyte molecules, making them promising candidates.
Candidates as gas-sensing materials. In this review, we provide an overview of synthetic strategies for COF powders and thin films and summarize the recent progress in COF-based chemoresistors with various types of linkages. The representative gas-sensing performances of COF-based sensors are listed in Table 1. Although significant achievements have been made in this research field, several key challenges remain to be addressed to realize the practical and reliable use of COF-based gas sensors.

The summary of the sensing properties of COF-based chemoresistive gas sensors (*: calculated detection limit, **: experimental detection limit).
First, the inherently insulating nature of covalent bonds within 2D COFs restricts charge transport primarily to π-π stacking between adjacent layers, limiting the effectiveness of signal measurements (e.g., resistance, current, and conductivity) through conventional and cost-effective electronic circuits. Second, the overall gas-sensing performance of COFs, particularly in terms of gas response magnitude, sensitivity, stability, and signal reproducibility, requires further improvement compared to conventional sensing materials. Moreover, the selectivity of current COF-based sensors is restricted to a limited range of analytes, primarily NO2 and NH3. Therefore, expanding the selectivity to a broader range of gases is critical. Third, challenges remain in the scalable synthesis of COFs and their integration into practical device platforms, as most reported COFs have been synthesized under solvothermal conditions, which are time-consuming and unsuitable for large-scale fabrication.
To overcome these challenges, several promising strategies have been proposed: (1) Developing synthetic methods that reduce defects and improve framework crystallinity; (2) Incorporating electron-linking units to enhance intrinsic electrical conductivity; (3) Controlling COF morphology and film thickness to improve analyte accessibility and diffusion; (4) Introducing post-synthetic modifications or combining COFs with catalysts to enhance analyte adsorption and promote selective surface interactions; (5) Exploring scalable fabrication techniques, such as CVD. COFs are expected to evolve into promising platforms for next-generation gas sensors due to their outstanding structural tunability, chemical diversity, and potential for functionalization. We hope this review provides valuable guidance and insights for advancing the practical development of high-performance COF-based chemoresistive sensors.
Abbreviation
| BD : | Benzidine |
| BD-(NO2)2 : | 3,3’-Dinitrobenzidine |
| BD-(OMe)2 : | o-Dianisidine |
| BD-Me2 : | o-Tolidine |
| BDT : | Benzodithiophene |
| BPy : | 2,2’-Bipyridine-5,5’-diamine |
| DAAQ : | 2,6-Diamino-anthraquinone |
| DAB : | 1,4-Diaminobenzene |
| DANT : | 2,7-Diaminobenzo[lmn][3,8]phenanthroline-1,3,6,8(2H,7H)-tetraone |
| DFP : | 2,6-Diformylpyridine |
| DHF : | 9,9-Dihexylfluorene-2,7-diamine |
| NiOAPc : | Nickelphthalocyanine |
| NTCDA : | Naphthalene-1,4,5,8-tetracarboxylic dianhydride |
| Pa-SO3H : | 2,5-Diaminobenzenesulfonic Acid |
| PCBA : | 2,6-Pyridinedicarboxaldehyde |
| PDA : | p-Henylenediamine |
| PDA-F4 : | 2,3,5,6-Tetrafluoro-1,4-phenylenediamine |
| PMDA : | Pyromellitic dianhydride |
| PTCDA : | Perylene-3,4,9,10-tetracarboxylic dianhydride |
| PTSA : | Amine-p-toluene sulfonic acid |
| Py : | Pyrene |
| TAPA : | Tris(4-aminophenyl)amine |
| TAPB : | 2,4,6-Tris(4-aminophenyl)-benzene |
| TAPP : | 5,10,15,20-Tetrakis(4-aminophenyl)porphyrin |
| TAPT : | 2,4,6-Tris(4-aminophenyl)-triazine |
| TCH : | 2,5,8-Trichloroheptazine |
| TFB : | 1,3,5-Triformylbenzene |
| TFP : | 1,3,5-Triformylphloroglucinol |
| TFPB : | 1,2,4,5-Tetrakis-(4-formylphenyl) benzene |
| TFPPy : | 1,3,6,8-Tetrakis(4-formylphenyl) pyrene |
| THSTZ : | 2,4,6-Trihydrazinyl-1,3,5-triazine |
| TOPyr : | Pyrenetetraone |
| TPDA : | [1,1’:4’,1”-Terphenyl]-4,4”-dicarboxaldehyde |
| TPTCDA : | 1,6,7,12-Tetrachloroperylene tetracarboxylic acid dianhydride |
Acknowledgments
This study was financially supported by the Cooperative Research Program for Agriculture Science and Technology Development (Project No. PJ01706703) of the Rural Development Administration, Republic of Korea. This work was also financially supported by the Nano & Material Technology Development Program (RS-2024-00405016) through the National Research Foundation of Korea (NRF), funded by the Ministry of Science and ICT, Republic of Korea. The Inter-University Semiconductor Research Center and Institute of Engineering Research at Seoul National University provided research facilities for this work.
References
-
S.-Y. Jeong, Y.K. Moon, J. Wang, J.-H. Lee, Exclusive detection of volatile aromatic hydrocarbons using bilayer oxide chemiresistors with catalytic overlayers, Nat. Commun. 14 (2023) 233.
[https://doi.org/10.1038/s41467-023-35916-3]

-
Y.-M. Jo, K. Lim, J.W. Yoon, Y.K. Jo, Y.K. Moon, H.W. Jang, et al., Visible-light-activated Type II heterojunction in Cu3(hexahydroxytriphenylene)2/Fe2O3 hybrids for reversible NO2 sensing: Critical role of π–π* transition, ACS Cent. Sci. 7 (2021) 1176–1182.
[https://doi.org/10.1021/acscentsci.1c00289]

-
Y.K. Jo, S.-Y. Jeong, Y.K. Moon, Y.-M. Jo, J.-W. Yoon, J.-H. Lee, Exclusive and ultrasensitive detection of formaldehyde at room temperature using a flexible and monolithic chemiresistive sensor, Nat. Commun. 12 (2021) 1–9.
[https://doi.org/10.1038/s41467-021-25290-3]

-
S.J. Park, Y.K. Moon, S.-W. Park, S.M. Lee, T.-H. Kim, S.Y. Kim, et al., Highly sensitive and selective real-time breath isoprene detection using the gas reforming reaction of MOF-derived nanoreactors, ACS Appl. Mater. Interfaces 15 (2023) 7102–7111.
[https://doi.org/10.1021/acsami.2c20416]

-
S.J. Park, S.M. Lee, M.-H. Oh, Y.S. Huh, H.W. Jang, Food quality assessment using chemiresistive gas sensors: achievements and future perspectives, Sustainable Food Technol. 2 (2024) 266–280.
[https://doi.org/10.1039/D3FB00196B]

-
S.Y. Park, Y. Kim, T. Kim, T.H. Eom, S.Y. Kim, H.W. Jang, Chemiresistive materials for electronic nose: Progress, perspectives, and challenges, InfoMat 1 (2019) 289–316.
[https://doi.org/10.1002/inf2.12029]

-
S.Y. Jeong, J.S. Kim, J.H. Lee, Rational design of semiconductor-based chemiresistors and their libraries for next-generation artificial olfaction, Adv. Mater. 32 (2020) 2002075.
[https://doi.org/10.1002/adma.202002075]

-
M. Kim, S. Park, J. Ahn, J. W. Baek, D.-H. Kim, H. Shin, et al., Vitalizing Perovskite Oxide-Based Acetone Sensors with Metal−Organic Framework Derived Heterogeneous Oxide Catalysts, ACS Sens. 9 (2024) 6492–6501.
[https://doi.org/10.1021/acssensors.4c01852]

-
S.M. Lee, Y.K. Moon, K. Lim, S.-W. Park, S.J. Park, T.-H. Kim, et al., One-dimensional In2O3 nanofibers patterned onto functionalized catalytic electrodes: A novel approach for selective xylene detection, Sens. Actuators B Chem. 382 (2023) 133494.
[https://doi.org/10.1016/j.snb.2023.133494]

-
G.B. Nam, T.H. Eom, S.H. Cho, Y.J. Kim, S. Choi, W.S. Cheon, et al., Maximized nanojunctions in Pd/SnO2 nanoparticles for ultrasensitive and rapid H2 detection, Chem. Eng. J. 494 (2024) 153116.
[https://doi.org/10.1016/j.cej.2024.153116]

-
S.J. Park, S.M. Lee, J. Lee, S. Choi, G.B. Nam, Y.K. Jo, et al., Pd-W18O49 Nanowire MEMS Gas Sensor for Ultraselective Dual Detection of Hydrogen and Ammonia, Small 21 (2025) 2405809.
[https://doi.org/10.1002/smll.202405809]

-
S.M. Lee, Y.J. Kim, S.J. Park, W.S. Cheon, J. Kim, G.B. Nam, et al., In-Situ Growth of 2D MOFs as a Molecular Sieving Layer on SnS2 Nanoflakes for Realizing Ultraselective H2S Detection, Adv. Funct. Mater. 35 (2025) 2417019.
[https://doi.org/10.1002/adfm.202417019]

-
Y.H. Kim, J.S. Park, Y.-R. Choi, S.Y. Park, S.Y. Lee, W. Sohn, et al., Chemically fluorinated graphene oxide for room temperature ammonia detection at ppb levels, J. Mater. Chem. A 5 (2017) 19116–19125.
[https://doi.org/10.1039/C7TA05766K]

-
Y.M. Jo, Y.K. Jo, J.H. Lee, H.W. Jang, I.S. Hwang, D.J. Yoo, MOF-Based Chemiresistive Gas Sensors: Toward New Functionalities, Adv. Mater. 35 (2023) 2206842.
[https://doi.org/10.1002/adma.202206842]

-
T. Kim, T.H. Lee, S.Y. Park, T.H. Eom, I. Cho, Y. Kim, et al., Drastic Gas Sensing Selectivity in 2-Dimensional MoS2 Nanoflakes by Noble Metal Decoration, ACS Nano 17 (2023) 4404–4413.
[https://doi.org/10.1021/acsnano.2c09733]

-
Y.-M. Jo, D.-H. Kim, J. Wang, J.J. Oppenheim, M. Dincă, Humidity-mediated dual ionic–electronic conductivity enables high sensitivity in MOF chemiresistors, J. Am. Chem. Soc. 146 (2024) 20213–20220.
[https://doi.org/10.1021/jacs.4c05343]

-
A.P. Cote, A.I. Benin, N.W. Ockwig, M. O'Keeffe, A.J. Matzger, O.M. Yaghi, Porous, crystalline, covalent organic frameworks, Science 310 (2005) 1166–1170.
[https://doi.org/10.1126/science.1120411]

-
K. Geng, T. He, R. Liu, S. Dalapati, K.T. Tan, Z. Li, et al., Covalent organic frameworks: design, synthesis, and functions, Chem. Rev. 120 (2020) 8814–8933.
[https://doi.org/10.1021/acs.chemrev.9b00550]

-
S.-Y. Ding, W. Wang, Covalent organic frameworks (COFs): from design to applications, Chem. Soc. Rev. 42 (2013) 548–568.
[https://doi.org/10.1039/C2CS35072F]

-
Z. Xiang, D. Cao, Porous covalent–organic materials: synthesis, clean energy application and design, J. Mater. Chem. A 1 (2013) 2691–2718.
[https://doi.org/10.1039/C2TA00063F]

-
M.K. Lee, M. Shokouhimehr, S.Y. Kim, H.W. Jang, Two-Dimensional Metal–Organic Frameworks and Covalent–Organic Frameworks for Electrocatalysis: Distinct Merits by the Reduced Dimension, Adv. Energy Mater. 12 (2022) 2003990.
[https://doi.org/10.1002/aenm.202003990]

-
S.-Y. Ding, J. Gao, Q. Wang, Y. Zhang, W.-G. Song, C.-Y. Su, et al., Construction of covalent organic framework for catalysis: Pd/COF-LZU1 in Suzuki–Miyaura coupling reaction, J. Am. Chem. Soc. 133 (2011) 19816–19822.
[https://doi.org/10.1021/ja206846p]

-
H. Furukawa, O.M. Yaghi, Storage of hydrogen, methane, and carbon dioxide in highly porous covalent organic frameworks for clean energy applications, J. Am. Chem. Soc. 131 (2009) 8875–8883.
[https://doi.org/10.1021/ja9015765]

-
D. Zhu, G. Xu, M. Barnes, Y. Li, C.P. Tseng, Z. Zhang, et al., Covalent organic frameworks for batteries, Adv. Funct. Mater. 31 (2021) 2100505.
[https://doi.org/10.1002/adfm.202100505]

-
T. Sun, J. Xie, W. Guo, D.S. Li, Q. Zhang, Covalent–organic frameworks: advanced organic electrode materials for rechargeable batteries, Adv. Energy Mater. 10 (2020) 1904199.
[https://doi.org/10.1002/aenm.201904199]

-
Z. Meng, R.M. Stolz, K.A. Mirica, Two-dimensional chemiresistive covalent organic framework with high intrinsic conductivity, J. Am. Chem. Soc. 141 (2019) 11929–11937.
[https://doi.org/10.1021/jacs.9b03441]

-
W. Ye, L. Zhao, H.-Z. Lin, L. Ding, Q. Cao, Z.-K. Chen, et al., Halide Perovskite glues activate two-dimensional covalent organic framework crystallites for selective NO2 sensing, Nat. Commun. 14 (2023) 2133.
[https://doi.org/10.1038/s41467-023-37296-0]

-
H. Yang, S. Zhang, L. Han, Z. Zhang, Z. Xue, J. Gao, et al., High conductive two-dimensional covalent organic framework for lithium storage with large capacity, ACS Appl. Mater. interfaces 8 (2016) 5366–5375.
[https://doi.org/10.1021/acsami.5b12370]

-
S. Wan, F. Gándara, A. Asano, H. Furukawa, A. Saeki, S.K. Dey, et al., Covalent organic frameworks with high charge carrier mobility, Chem. Mater. 23 (2011) 4094–4097.
[https://doi.org/10.1021/cm201140r]

-
S. Dalapati, S. Jin, J. Gao, Y. Xu, A. Nagai, D. Jiang, An azine-linked covalent organic framework, J. Am. Chem. Soc. 135 (2013) 17310–17313.
[https://doi.org/10.1021/ja4103293]

-
D.-H. Yang, Z.-Q. Yao, D. Wu, Y.-H. Zhang, Z. Zhou, X.-H. Bu, Structure-modulated crystalline covalent organic frameworks as high-rate cathodes for Li-ion batteries, J. Mater. Chem. A 4 (2016) 18621–18627.
[https://doi.org/10.1039/C6TA07606H]

-
P. Kuhn, M. Antonietti, A. Thomas, Porous, covalent triazine-based frameworks prepared by ionothermal synthesis, Angew. Chem. Int. Ed. 47 (2008) 3450–3453.
[https://doi.org/10.1002/anie.200705710]

-
J. Maschita, T. Banerjee, G. Savasci, F. Haase, C. Ochsenfeld, B.V. Lotsch, Ionothermal synthesis of imide-linked covalent organic frameworks, Angew. Chem. Int. Ed. 59 (2020) 15750–15758.
[https://doi.org/10.1002/anie.202007372]

-
C.O. Kappe, Controlled microwave heating in modern organic synthesis, Angew. Chem. Int. Ed. 43 (2004) 6250–6284.
[https://doi.org/10.1002/anie.200400655]

-
N.L. Campbell, R. Clowes, L.K. Ritchie, A.I. Cooper, Rapid microwave synthesis and purification of porous covalent organic frameworks, Chem. Mater. 21 (2009) 204–206.
[https://doi.org/10.1021/cm802981m]

-
H. Wei, S. Chai, N. Hu, Z. Yang, L. Wei, L. Wang, The microwave-assisted solvothermal synthesis of a crystalline two-dimensional covalent organic framework with high CO2 capacity, Chem. Commun. 51 (2015) 12178–12181.
[https://doi.org/10.1039/C5CC04680G]

-
T. Kim, J. Oh, S.C. Kim, J.G. Ahn, S. Kim, Y.Y. Kim, H. Lim, Photochemical and Patternable Synthesis of 2D Covalent Organic Framework Thin Film Using Dynamic Liquid/Solid Interface, Small Methods 8 (2024) 2400063.
[https://doi.org/10.1002/smtd.202400063]

-
L.-Y. Chen, Y.-N. Gai, X.-T. Gai, J. Qin, Z.-G. Wang, L.-S. Cui, et al., Interfacial synthesized covalent organic framework nanofiltration membranes for precisely ultrafast sieving, Chem. Eng. J. 430 (2022) 133024.
[https://doi.org/10.1016/j.cej.2021.133024]

-
W. Dai, F. Shao, J. Szczerbiński, R. McCaffrey, R. Zenobi, Y. Jin, et al., Synthesis of a two-dimensional covalent organic monolayer through dynamic imine chemistry at the air/water Interface, Angew. Chem. 128 (2016) 221–225.
[https://doi.org/10.1002/ange.201508473]

-
D.B. Shinde, G. Sheng, X. Li, M. Ostwal, A.-H. Emwas, K.-W. Huang, et al., Crystalline 2D covalent organic framework membranes for high-flux organic solvent nanofiltration, J. Am. Chem. Soc. 140 (2018) 14342–14349.
[https://doi.org/10.1021/jacs.8b08788]

-
K. Dey, M. Pal, K.C. Rout, S. Kunjattu H, A. Das, R. Mukherjee, et al., Selective molecular separation by interfacially crystallized covalent organic framework thin films, J. Am. Chem. Soc. 139 (2017) 13083–13091.
[https://doi.org/10.1021/jacs.7b06640]

-
N.A. Khan, R. Zhang, H. Wu, J. Shen, J. Yuan, C. Fan, et al., Solid–vapor interface engineered covalent organic framework membranes for molecular separation, J. Am. Chem. Soc. 142 (2020) 13450–13458.
[https://doi.org/10.1021/jacs.0c04589]

-
J.W. Colson, A.R. Woll, A. Mukherjee, M.P. Levendorf, E.L. Spitler, V.B. Shields, et al., Oriented 2D covalent organic framework thin films on single-layer graphene, Science 332 (2011) 228–231.
[https://doi.org/10.1126/science.1202747]

-
D.D. Medina, V. Werner, F. Auras, R. Tautz, M. Dogru, J. Schuster, et al., Oriented thin films of a benzodithiophene covalent organic framework, ACS Nano 8 (2014) 4042–4052.
[https://doi.org/10.1021/nn5000223]

-
B. Sun, C.-H. Zhu, Y. Liu, C. Wang, L.-J. Wan, D. Wang, Oriented covalent organic framework film on graphene for robust ambipolar vertical organic field-effect transistor, Chem. Mater. 29 (2017) 4367–4374.
[https://doi.org/10.1021/acs.chemmater.7b00800]

-
M.S. Yao, X.J. Lv, Z.H. Fu, W.H. Li, W.H. Deng, G.D. Wu, et al., Layer-by-layer assembled conductive metal–organic framework nanofilms for room-temperature chemiresistive sensing, Angew. Chem. Int. Ed. 56 (2017) 16510–16514.
[https://doi.org/10.1002/anie.201709558]

-
X. Shi, R. Wang, A. Xiao, T. Jia, S.-P. Sun, Y. Wang, Layer-by-layer synthesis of covalent organic frameworks on porous substrates for fast molecular separations, ACS Appl. Nano Mater. 1 (2018) 6320–6326.
[https://doi.org/10.1021/acsanm.8b01537]

-
S. Hao, T. Zhang, S. Fan, Z. Jia, Y. Yang, Preparation of COF-TpPa1 membranes by chemical vapor deposition method for separation of dyes, Chem. Eng. J. 421 (2021) 129750.
[https://doi.org/10.1016/j.cej.2021.129750]

-
S. Chandra, S. Kandambeth, B.P. Biswal, B. Lukose, S.M. Kunjir, M. Chaudhary, et al., Chemically stable multilayered covalent organic nanosheets from covalent organic frameworks via mechanical delamination, J. Am. Chem. Soc. 135 (2013) 17853–17861.
[https://doi.org/10.1021/ja408121p]

-
S. Wang, Q. Wang, P. Shao, Y. Han, X. Gao, L. Ma, et al., Exfoliation of covalent organic frameworks into few-layer redox-active nanosheets as cathode materials for lithium-ion batteries, J. Am. Chem. Soc. 139 (2017) 4258–4261.
[https://doi.org/10.1021/jacs.7b02648]

-
I. Berlanga, M.L. Ruiz-González, J.M. González-Calbet, J.L.G. Fierro, R. Mas-Ballesté, F. Zamora, Delamination of layered covalent organic frameworks, Small 7 (2011) 1207–1211.
[https://doi.org/10.1002/smll.201002264]

-
D.N. Bunck, W.R. Dichtel, Bulk synthesis of exfoliated two-dimensional polymers using hydrazone-linked covalent organic frameworks, J. Am. Chem. Soc. 135 (2013) 14952–14955.
[https://doi.org/10.1021/ja408243n]

-
M.A. Khayum, S. Kandambeth, S. Mitra, S.B. Nair, A. Das, S.S. Nagane, R. Mukherjee, R. Banerjee, Chemically delaminated free-standing ultrathin covalent organic nanosheets, Angew. Chem. Int. Ed. 55 (2016) 15604–15608.
[https://doi.org/10.1002/anie.201607812]

-
X. Chen, Y. Li, L. Wang, Y. Xu, A. Nie, Q. Li, et al., High-lithium-affinity chemically exfoliated 2D covalent organic frameworks, Adv. Mater. 31 (2019) 1901640.
[https://doi.org/10.1002/adma.201901640]

-
S. Mitra, S. Kandambeth, B.P. Biswal, A. Khayum M, C.K. Choudhury, M. Mehta, et al., Self-exfoliated guanidinium-based ionic covalent organic nanosheets (iCONs), J. Am. Chem. Soc. 138 (2016) 2823–2828.
[https://doi.org/10.1021/jacs.5b13533]

-
N. Zhang, T. Wang, X. Wu, C. Jiang, F. Chen, W. Bai, et al., Self-exfoliation of 2D covalent organic frameworks: morphology transformation induced by solvent polarity, RSC Adv. 8 (2018) 3803–3808.
[https://doi.org/10.1039/C7RA09647J]

-
F.J. Uribe-Romo, J.R. Hunt, H. Furukawa, C. Klock, M. O’Keeffe, O.M. Yaghi, A crystalline imine-linked 3-D porous covalent organic framework, J. Am. Chem. Soc. 131 (2009) 4570–4571.
[https://doi.org/10.1021/ja8096256]

-
F. Niu, Z.-W. Shao, J.-L. Zhu, L.-M. Tao, Y. Ding, Structural evolution of imine-linked covalent organic frameworks and their NH3 sensing performance, J. Mater. Chem. C 9 (2021) 8562–8569.
[https://doi.org/10.1039/D1TC01662H]

-
M. Liu, Y.J. Chen, X. Huang, L.Z. Dong, M. Lu, C. Guo, et al., Porphyrin-based COF 2D materials: variable modification of sensing performances by post-metallization, Angew. Chem. Int. Ed. 61 (2022) e202115308.
[https://doi.org/10.1002/anie.202115308]

-
Y.-J. Chen, M. Liu, J. Chen, X. Huang, Q.-H. Li, X.-L. Ye, et al., Dangling bond formation on COF nanosheets for enhancing sensing performances, Chem. Sci. 14 (2023) 4824–4831.
[https://doi.org/10.1039/D3SC00562C]

-
S. Kandambeth, A. Mallick, B. Lukose, M.V. Mane, T. Heine, R. Banerjee, Construction of crystalline 2D covalent organic frameworks with remarkable chemical (acid/base) stability via a combined reversible and irreversible route, J. Am. Chem. Soc. 134 (2012) 19524–19527.
[https://doi.org/10.1021/ja308278w]

-
F. Zhang, K. Xiong, X. Lang, Two-Dimensional β-Ketoenamine-Linked Covalent Organic Frameworks for Visible Light Photocatalysis, ChemCatChem 15 (2023) e202301179.
[https://doi.org/10.1002/cctc.202301179]

-
Z. Yan, M. Fang, L. Wang, H. Gao, Y. Ying, J. Yang, J. Yang, et al., Linkage Engineering of Semiconductive Covalent-Organic Frameworks toward Room-Temperature Ppb-Level Selective Ammonia Sensing, Small 21 (2025) 2407436.
[https://doi.org/10.1002/smll.202407436]

-
A. Mei, M. Zhou, W. Zhang, Y. Bao, Y. Liu, W. Chen, Multi-functionalized sites in metalated COFs film for chemiresistive gas sensing at room temperature, Chem. Eng. J. 514 (2025) 163087.
[https://doi.org/10.1016/j.cej.2025.163087]

-
L.-M. Tao, F. Niu, D. Zhang, T.-M. Wang, Q.-H. Wang, Amorphous covalent triazine frameworks for high performance room temperature ammonia gas sensing, New J. Chem. 38 (2014) 2774–2777.
[https://doi.org/10.1039/c4nj00476k]

-
F. Niu, Z.-W. Shao, L.-M. Tao, Y. Ding, Covalent triazine-based frameworks for NH3 gas sensing at room temperature, Sens. Actuators B Chem. 321 (2020) 128513.
[https://doi.org/10.1016/j.snb.2020.128513]

-
K. Yang, W. Yuan, Z. Hua, Y. Tang, F. Yin, D. Xia, Triazine-based two-dimensional organic polymer for selective NO2 sensing with excellent performance, ACS Appl. Mater. Interfaces 12 (2019) 3919–3927.
[https://doi.org/10.1021/acsami.9b17450]

-
F. Niu, J.-L. Zhu, M.-K. Xu, Y. Ding, L.-M. Tao, J. Jin, Viologen-based covalent triazine frameworks for NO2 sensing at room temperature, Sens. Actuators B Chem. 352 (2022) 131045.
[https://doi.org/10.1016/j.snb.2021.131045]

-
Q. Fang, Z. Zhuang, S. Gu, R.B. Kaspar, J. Zheng, J. Wang, et al., Designed synthesis of large-pore crystalline polyimide covalent organic frameworks, Nat. Commun. 5 (2014) 4503.
[https://doi.org/10.1038/ncomms5503]

-
N. Thokala, K. Vankayala, A.D. Gaonkar, G. Periyasamy, K. Fazl-Ur-Rahman, K. Valle, et al., An exfoliated redox active imide covalent organic framework for metal free hydrogen gas sensing, Sens. Diagn. 2 (2023) 1176–1180.
[https://doi.org/10.1039/D3SD00097D]

-
W. Gao, Y. Bai, X. Wang, H. Fu, P. Zhao, P. Zhu, et al., Self-standing perylene diimide covalent organic framework membranes for trace TMA sensing at room temperature, J. Colloid Interface Sci. 663 (2024) 262–269.
[https://doi.org/10.1016/j.jcis.2024.02.145]

-
X. Guan, F. Chen, Q. Fang, S. Qiu, Design and applications of three dimensional covalent organic frameworks, Chem. Soc. Rev. 49 (2020) 1357–1384.
[https://doi.org/10.1039/C9CS00911F]

-
Z. Yang, A. Mei, W. Chen, Z. Wang, H. Guo, Y. Liu, Control of morphology of covalent organic framework-5 for chemiresistive low-temperature ammonia sensors, Sens. Actuators B Chem 392 (2023) 134051.
[https://doi.org/10.1016/j.snb.2023.134051]

-
N. Sharma, N. Sharma, P. Srinivasan, S. Kumar, J.B.B. Rayappan, K. Kailasam, Heptazine based organic framework as a chemiresistive sensor for ammonia detection at room temperature, J. Mater. Chem. A 6 (2018) 18389–18395.
[https://doi.org/10.1039/C8TA06937A]

-
W. Zhang, Q. Sun, Y. Zhu, J. Sun, Z. Wu, N. Tian, High-Performance Trimethylamine Sensor Based on an Imine Covalent Organic Framework, ACS Sens. 9 (2024) 3262–3271.
[https://doi.org/10.1021/acssensors.4c00613]

-
J. Wang, Q. Cao, X.-F. Cheng, W. Ye, J.-H. He, J.-M. Lu, Moisture-Insensitive and Highly Selective Detection of NO2 by Ion-in-Conjugation Covalent Organic Frameworks, ACS Sens. 7 (2022) 3782–3789.
[https://doi.org/10.1021/acssensors.2c01631]

-
V. Krishnaveni, M. Esclance DMello, P. Sahoo, N. Thokala, V.R. Bakuru, K. Vankayala, et al., Palladium-nanoparticle-decorated covalent organic framework nanosheets for effective hydrogen gas sensors, ACS Appl. Nano Mater. 6 (2023) 10960–10966.
[https://doi.org/10.1021/acsanm.3c01806]

-
Y. Yue, P. Cai, X. Xu, H. Li, H. Chen, H.C. Zhou, et al., Conductive metallophthalocyanine framework films with high carrier mobility as efficient chemiresistors, Angew. Chem. Int. Ed. 60 (2021) 10806–10813.
[https://doi.org/10.1002/anie.202100717]


Soo Min Lee is currently a PhD candidate under the supervision of Prof. Ho Won Jang of the Department of Materials Science and Engineering, Seoul National University. She received her B.S. and M.S. degrees from the Department of Materials Science and Engineering at Korea University in 2021 and 2023, respectively. Her current research is focused on the synthesis and design of chemoresistive gas sensors.

Seon Ju Park is currently a PhD candidate under the supervision of Professor Ho Won Jang of the Department of Materials Science and Engineering at Seoul National University. She received her B.S. and M.S. degrees from the Department of Materials Science and Engineering at Korea University in 2021 and 2023, respectively. Her current research is focused on the synthesis and design of chemoresistive gas sensors.

Ho Won Jang is a full professor in the Department of Materials Science and Engineering at Seoul National University. He completed his Ph.D. in Materials Science and Engineering at the Pohang University of Science and Technology in 2004. From 2006 to 2009, he served as a Research Associate at the University of Wisconsin, Madison. Before joining Seoul National University in 2012, he worked as a senior research scientist at the Korea Institute of Science and Technology. His research focuses on material synthesis and device fabrication for applications such as chemical sensing, solar fuel generation, nonvolatile data storage, neuromorphic computing, plasmonics, ferroelectronics, and metal-insulator transitions. He has authored more than 650 publications in international journals.