
Advances in Skin-interfaced Wireless Physiological Monitoring Systems
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Wireless physiological monitoring has emerged as a transformative solution for continuous and non-invasive assessment of vital signs in both clinical and non-clinical environments. Conventional wired monitoring systems often suffer from the bulkiness, usability, and restrictive applications only in hospital. Recent advances in flexible electronics, low-power wireless communication, and integrated multimodal sensing have significantly improved the functionality, wearability, and usability of wireless physiological monitoring devices. This review outlines recent technical advances in the wireless monitoring into five groups based on sensing mechanisms: a) thermoelectric sensors for core body temperature, b) biopotential sensors for electrophysiology, c) optical sensors for photoplethysmography, d) electrochemical sensors for sweat, blood, and interstitial fluid monitoring, and e) mechanical and acoustic sensors for unconventional physiological monitoring.
Keywords:
Wireless physiological monitoring, Wearable sensors1. INTRODUCTION
The continuous monitoring of physiological signals has become integral to modern healthcare, enabling early detection of acute conditions and supporting prompt clinical intervention [1]. Extending beyond acute care, long-term monitoring provides valuable insights into disease progression and supports personalized therapeutic strategies by capturing individual-specific trends over time [2]. Furthermore, continuous monitoring, equipped with remote patient monitoring, is essential in preventive medicine. Detecting early physiological alterations associated with chronic conditions, such as cardiovascular disease, diabetes, and respiratory disorders, could facilitate timely intervention for lifestyle modifications and clinical treatment, with the potential to reduce long-term healthcare costs [3-5]. In the context of public health and outbreak management, continuous and wearable physiological monitoring has demonstrated significant value in improving healthcare delivery, even in environments with limited resources [6].
Traditional wired physiological monitoring systems have long been widely used in hospitals for their accuracy and reliability; however, several limitations affect not only clinical workflow but also patient safety and usability. For instance, rigid monitoring stations connected by multiple wires and cables often restrict patient mobility and complicate routine clinical tasks, such as repositioning or diagnostic imaging. During long-term monitoring, these systems often cause skin irritation or detachment, particularly in sensitive populations such as neonates [7]. Furthermore, conventional wired systems in hospital settings are not easily translated to remote patient monitoring. Wireless physiological monitoring systems have emerged as transformative solutions to address these significant challenges. Recent advances in stretchable biocompatible materials have enabled the development of wireless, non-invasive wearable sensors that can be worn comfortably for extended durations, significantly enhancing patient comfort and usability and reducing the risk of skin injury [8-10]. Moreover, the integration of low-power wireless communication protocols, such as Bluetooth low-energy (BLE) and near-field communication (NFC) supports real-time remote monitoring, which is central to data integration with real-time transmission [8,9,11]. The new class of physiology monitors equipped with wireless operations and other advanced capabilities also enables seamless integration into hospital information systems and cloud-based platforms, creating the foundation for proactive, personalized healthcare across diverse environments, including acute care settings, out-of-hospital care, and ambulatory use [9,11].
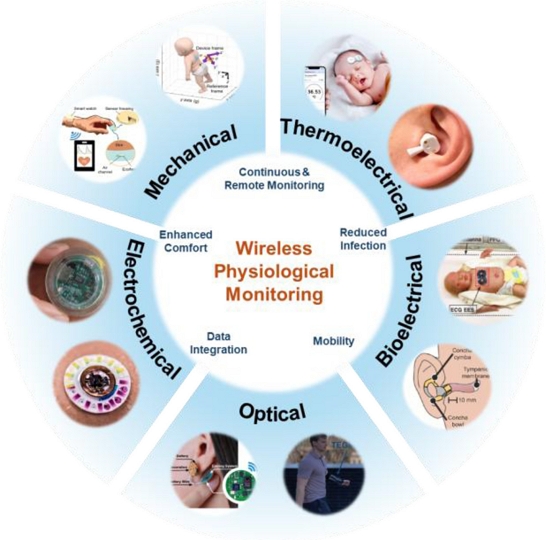
Overview of wireless physiological monitoring by sensing mechanism and core advantages. Adapted from Ref. [8,9,12-17], and THERMOSAFER XST400 by Dailytem.
In this review, skin-interfaced wireless physiological monitoring technologies are systematically categorized into five groups, based on their sensing mechanisms and the physical characteristics of the measured signals, to facilitate a clear comparison and application-oriented analysis. These categories are (1) thermoelectrical sensors (core body temperature (CBT)), (2) bioelectrical sensors (electrocardiography (ECG) and electroencephalography (EEG)), (3) optical sensors (photoplethysmography (PPG)), (4) biochemical sensors (sweat and interstitial fluid (ISF) sensors), and (5) mechanical acoustic sensors (inertial measurement unit (IMU) and pulse sensors). This classification reflects the fact that physiological signals are generated through diverse biological processes, both internally and externally, and thus, require fundamentally different sensing strategies for accurate detection and interpretation [18]. Because these signals vary in their physical properties, spatial origin, temporal dynamics, and measurement complexity, organizing them by sensing principles enables a structured evaluation of the advantages, limitations, and clinical applicability of each technology. In addition, this review identifies current challenges and outlines future directions for improvement in each sensor category.
2. ADVANCES IN WIRELESS PHYSIOLOGICAL MONITORING
2.1 Thermoelectric Sensors for Core Body Temperature
The CBT is a critical physiological parameter that reflects the body’s internal thermal state and serves as a key indicator of metabolic activity, immune responses, thermoregulation, and acute illness [19]. Conventional measurement methods, such as rectal and esophageal temperature monitoring, offer high accuracy but are invasive and unsuitable for continuous monitoring, limiting their applicability in daily life [19]. To overcome these limitations, researchers have extensively investigated accurate wireless CBT sensors. These sensors can be classified into heat-flux-based and non-heat-flux-based types. Heat-flux-based sensors estimate the CBT by measuring the temperature gradient between the skin surface and an insulating layer to calculate the heat flux, whereas non-heat-flux-based sensors measure it indirectly through methods such as infrared sensing or physiological signal analysis [19,20]. The heat-flux approach can be classified into three types: single-heat flux (SHF), dual-heat flux (DHF), and zero-heat flux (ZHF). Lauronen et al. reported that the ZHF method closely matched the golden standard, with mean differences within approximately 0.1°C, indicating strong agreement with the reference method [21]. However, sensors using ZHF often require a heater to maintain a zero-temperature gradient, thereby increasing the power requirement and making wireless implementation challenging.
For the DHF method, Panunzio et al. developed a wireless multi-sensor patch consisting of two vertically stacked thermistors embedded in layers with different thermal resistances [22]. This structure enables real-time CBT estimation by solving inverse heat transfer problems, and the wireless configuration supports continuous monitoring under free-living conditions. Another example of the DHF approach is a smart mask integrated with a heat-flux sensor, developed by Lazaro et al., which can measure the CBT as the user passes through a screening gate (Fig. 2 (a)) [23]. This noncontact feature enables rapid measurements and is advantageous for short-term CBT assessments during activities such as public health screening. Commercially, the Moni-Patch by Murata Manufacturing applies DHF principles in a compact adhesive patch, demonstrating the clinical feasibility of remote patient monitoring (Fig. 2 (b)). Another example of a commercialized device is THERMOSAFER by Dailytem, which features a forehead-mounted design integrated with a low-power wireless communication module (Fig. 2 (c)).
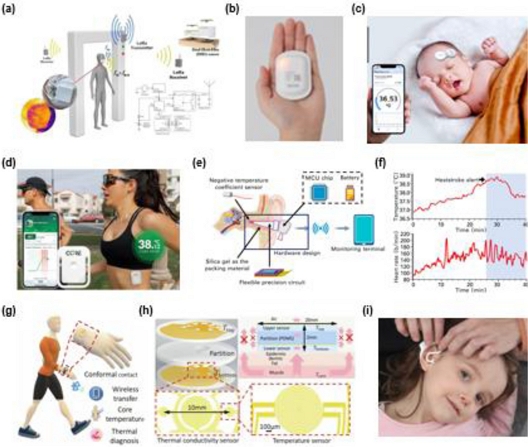
Representative wireless systems for core body temperature (CBT) monitoring. (a) Smart face mask with an integrated heat flux sensor. Adapted from Ref. [23]. (b) Moni-Patch® (Murata Manufacturing) by Murata Manufacturing. (c) THERMOSAFER XST400 by Dailytem. (d) CORE, a commercial wearable CBT used in sports and occupational settings by Greenteg. (e, f) Continuous CBT monitoring for heatstroke alert via a wearable in-ear thermometer. Adapted from Ref. [12]. (g, h) Flexible multimodal device for measuring body temperature, core temperature, thermal conductivity, and water content. Adapted from Ref. [26]. (i) Cosinuss degree° sensor by Cosinuss.
The SHF approach simplifies the system by using only one heat flow path. Hashimoto et al. introduced a reference-free SHF sensor that integrates thermal modeling to eliminate external calibration, making it more suitable for uncontrolled, mobile use [24]. The Greenteg CORE device is a prominent commercial SHF-based product that provides real-time CBT data via Bluetooth in a lightweight and wearable form (Fig. 2 (d)). The device demonstrates stability in sports and clinical settings, making it a potential solution for applications requiring comfort, accuracy, and power efficiency.
Non-heat-flux methods are compact and easy to integrate into wearables; however, their accuracy varies with physiology, environment, and device placement. In-ear thermometers have promising characteristics owing to their proximity to central thermal regions. Zhang et al. developed a compact wireless in-ear device using Bluetooth to enable continuous monitoring and real-time heatstroke alerts [12]. The device could track changes in core temperature and heart rate during heat exposure, offering more reliable measurements and greater comfort over extended periods than surface-mounted sensors (Fig. 2 (e) and (f)). Kato et al. developed an ear-canal infrared sensor that captured net heat flow from the tympanic membrane for continuous CBT monitoring in hot and humid work environments [25]. Positioned at a right angle to the membrane and shielded with medical film (TegadermTM, 3M) to reduce airflow effects, the device showed limits of agreement of the mean ± 0.3 – 0.6°C with rectal temperature, showing potential usages in sports or labor settings. Du et al. employed a multimodal approach instead of conventional single-parameter-based CBT estimation [26]. By integrating multiple physiological signals, such as water content and thermal conductivity, they improved the stability and accuracy of CBT estimation (Fig. 2 (g) and (h)). Oh et al. collected data from multiple sites to generate a heat transfer model and then synchronized the measurements to continuously observe whole-body temperature changes over time [27]. This approach exhibits a lower variability than single-point measurements, making it advantageous even in highly active environments. This evolution is further exemplified by the Cosinuss degree°, a commercially available in-ear device designed for CBT monitoring in infants (Fig. 2 (i)). Based on infrared sensing near the tympanic membrane, it estimates core temperature indirectly and transmits data wirelessly in real time. This demonstration in home-care settings highlights how compact, research-based sensor technologies can be adapted for safe and accessible pediatric use.
2.2 Biopotential Sensors for Electrocardiography
ECG is a non-invasive technique that records the electrical activity of the heart and serves as a fundamental diagnostic tool for detecting and managing various cardiovascular conditions [28]. Unlike conventional systems that use wired and multi-lead configurations, the emerging trend explores portable and wearable forms, enabling continuous monitoring in both hospital and home environments [29].
Zhuang et al. demonstrated a design with an intensive care unit (ICU)-grade breathable cardiac electronic skin system (BreaCARES) for real-time, wireless, and comfortable cardiac care that utilizes multilayer-patterned, permeable, and stretchable liquid metal (LM) microcircuits to prevent skin erythema or inflammation over a week-long attachment (Fig. 3 (a)) [30]. Similarly, in pursuit of skin-friendly and long-term wearables in clinical settings, Rauf et al. developed fully screen-printed, skin-friendly wet ECG electrodes integrated with a miniaturized printed circuit board (PCB) and packaged within a 3D-printed antenna-on-package (AoP) structure (Fig. 3 (b)) [31]. Wireless ECG also offers advantages in vulnerable populations such as premature babies and pregnant women, where these applications often demand high accuracy owing to the low amplitude of fetal and neonatal ECG signals, susceptibility to interference, and the need for precise measurements to guide timely interventions [8,32]. Chung et al. demonstrated a fully wireless bi-nodal system that continuously captures the full suite of vital signs in neonatal intensive care units (NICUs) (Fig. 3 (c)) [8]. Ryu et al. developed an integrated platform for monitoring key pregnancy biomarkers such as fetal heart rate (FHR), uterine contraction (UC), and maternal heart rate (MHR) using wireless and wearable skin patches, as shown in Fig. 3 (d) [11]. Nair et al. also demonstrated a novel ni-fECG device for monitoring FHR, UCs, and MHR during labor [33]. Compared with conventional ultrasound-based CTG, the device offers improved mobility and signal stability.
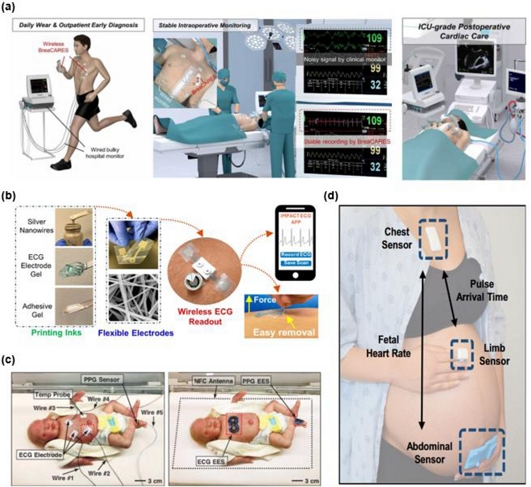
Representative advances of wireless ECG monitoring (a) Schematic illustrations demonstrating the versatility of the wireless BreaCARES system. Adapted from Ref. [30]. (b) Illustration of the components and workflow of the fully screen-printed gentle- to-skin wet ECG electrode system. Adapted from Ref. [31]. (c) Fully wireless vital signs platform in NICUs. Adapted from Ref. [8]. (d) Wireless and wearable pregnancy monitoring platform. Adapted from Ref. [11].
2.3 Biopotential Sensors for Electroencephalography
EEG is a non-invasive technique that records the electrophysiological activity of the cerebral cortex via scalp electrodes, capturing brain states such as arousal, sleep, cognition, emotion, and seizures [34]. EEG signals, generated primarily by postsynaptic currents in pyramidal neurons [35], are widely used to diagnose and manage neurological disorders, including epilepsy, stroke, delirium, dementia, and sleep-related conditions [34]. Recent technological advances in electrode technology, such as hydrogel, stretchable, and microneedle-based designs, have improved wearability, mechanical durability, and skin conformity, thereby enabling long-term monitoring in sensitive populations such as neonates [36-38]. Enhancements in electrode–substrate adhesion have improved the stability under repeated bending and stretching [39]. Furthermore, traditional single-signal EEG devices are evolving into multimodal platforms that integrate ECG, PPG, SpO₂, temperature, and motion sensing to enhance artifact suppression, diagnostic accuracy, and applicability beyond the clinical environment [40,41].
In clinical settings, wireless EEG systems address the needs of neonates, pediatric patients, and hospitalized adults, who require continuous monitoring, minimal skin irritation, and compatibility with hospital equipment [42-44]. Zhou et al. developed a stretchable epidermal soft patch for NICU use that offers BLE/NFC-based real-time streaming with high skin compliance [42]. Their system overcomes the limitations of rigid patches by leveraging stretchable circuit materials and demonstrates high skin compliance, specifically tailored for NICU settings. Wong et al. introduced a compact multimodal wireless platform for pediatric monitoring that integrates EEG, ECG, and respiratory signals into a single wearable unit [43]. Designed with flexible dry electrodes and a compact form, the system facilitates hospital workflow integration while minimizing skin irritation and setup time in sensitive pediatric populations. Wickramasinghe et al. proposed an active dry-contact EEG monitoring system optimized for neonatal care [44]. By eliminating conductive gels, which can cause skin irritation in neonates, and incorporating onboard seizure detection, the system enables long-term, low-irritation monitoring with real-time diagnostic capabilities. Jiang et al. developed an IoT-enabled multimodal monitoring system for neurocritical care of hospitalized patients using a foam-type dry electrode cap [45]. This platform supports the real-time acquisition of EEG, ECG, respiration, and motion signals with integrated artifact suppression and strong compatibility with the hospital infrastructure. Additionally, van Bohemen et al. validated a wearable EEG–ECG system in stroke patients, enabling continuous monitoring for over 9 h [46]. Unlike CT perfusion imaging, which provides only a time-point snapshot, this system offers real-time tracking of the brain and cardiac activity. Its compatibility with CT environments highlights its potential to complement conventional imaging techniques during stroke recovery.
In personal healthcare, priorities such as comfort, miniaturization, adaptability, motion tolerance, and long-term wearability are critical [13,47]. Kaveh et al. developed a compact in-ear EEG system for detecting driver drowsiness in naturalistic environments (Fig. 4 (a)) [13]. Incorporating BLE connectivity, motion-tolerant dry electrodes, and automatic artifact rejection, the system uses a universal ear tip design to maintain comfort and stability, reliably detecting early drowsiness through alpha-band EEG features (Fig. 4 (b)) [58]. Tang et al. proposed a non-contact electric potential sensor (EPS) capable of detecting EEG signals several centimeters away (Fig. 4 (c)) [47]. By combining high-sensitivity electric field detection with adaptive filtering, their system captures low-frequency brain activity under ambient conditions, offering an unobtrusive option for mental health monitoring. Zhou et al. introduced a wireless microneedle-based EEG platform employing shape-memory polymers that soften at body temperature (Fig. 4 (d)) [37]. This modulus-adaptive approach enhances skin conformity, lowers the contact impedance, and ensures long-term signal fidelity, making it more suitable than conventional dry electrodes for extended personal use. Beyond passive sensing, Huang et al. presented a closed-loop brain–computer interface (BCI) that enables users to actively regulate emotional states [48]. This home-validated neurofeedback system provides real-time feedback and adaptive training protocols, demonstrating its potential for non-pharmacological self-directed mental health support, including stress relief and emotional regulation.
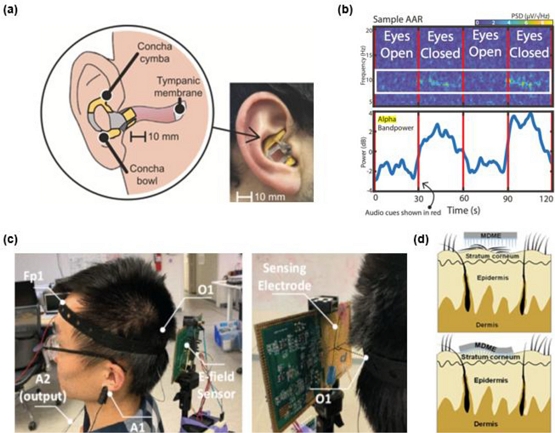
Wireless EEG systems for personal health monitoring. (a, b) In-ear EEG platform for detecting driver drowsiness with representative alpha-band power variations during eyes-open/eyes-closed task. Adapted from Ref. [13]. (c) Non-contact electric potential sensing (EPS) system. Adapted from Ref. [47]. (d) Modulus-adaptive microneedle EEG electrodes fabricated from PEDOT:PSS and elastic polymers. Adapted from Ref. [37].
2.4 Optical Sensors for Photoplethysmography
PPG is a non-invasive optical method that detects blood volume changes, thereby providing essential insights into cardiovascular function and hemodynamics [49]. In this review, PPG systems are categorized according to the choice of optical wavelength, as the wavelength directly determines the tissue penetration depth, signal-to-noise ratio, and specificity of measurements for different physiological parameters.
Red/IR PPG. The use of red and infrared (IR) light enables greater tissue penetration, establishing red/IR PPG as the conventional approach in pulse oximeters for non-invasive SpO₂ assessment. For instance, Koseeyaporn et al. introduced a pulse oximeter design that applies quadrature multiplexing of amplitude-modulated (AM) red and IR PPG signals, thereby improving SpO₂ accuracy in environments with significant noise [50]. Lee et al. developed standalone skin-like healthcare patches (SHPs) capable of delivering real-time heart rate data [51]. As shown in Fig. 5 (a), their forearm-mounted SHP acquires wrist-based PPG signals and presents them directly through an integrated OLED matrix. Expanding on sensor design, Kumar et al. developed a flexible organic photoplethysmography sensor (FOPS) incorporating organic photodetectors that function in both reflection and transmission modes, enabling blood pressure (BP) monitoring through both cuff-based and cuffless artificial neural network (ANN) techniques (Fig. 5 (b)) [52]. To mitigate the limitation of restricted operation time, Zadan et al. integrated high-performance thermoelectric generators (TEGs) using a stretchable LM–epoxy composite that offers superior thermal conductivity compared to existing thermal interface materials for wearable applications [15]. Similarly, Li et al. reported the development of ultrathin, self-powered photodetectors (PDs) fabricated from heavy-metal-free Cu–In–Se quantum dots (QDs) [53], which exploit the built-in photovoltaic potential to operate without external power, and their mechanical deformability supports their integration into skin-attachable PPG sensors for continuous heart rate monitoring in mobile bioelectronic systems.
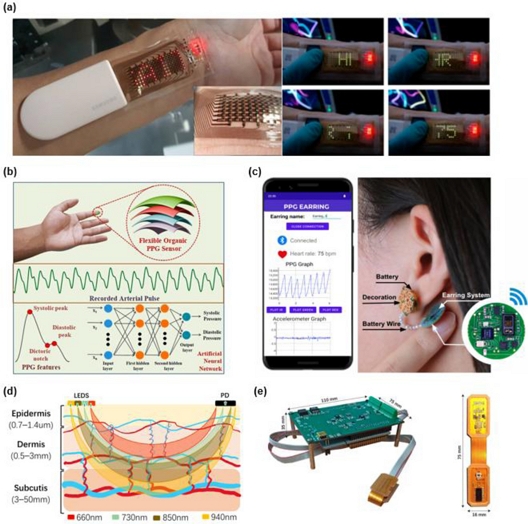
Wireless PPG systems for multimodal sensing and applications. (a) Standalone SHP device attached on the forearm with the red OLED array in dynamic operation. Adapted from Ref. [51]. (b) Flexible organic PPG sensor capturing arterial pulse signals and extracting PPG features. Adapted from Ref. [52]. (c) Demonstration of the PPG Earring system. Adapted from Ref. [14]. (d) Schematic of reflective MWPPG collection used for fusion-based BP prediction algorithm. Adapted from Ref. [56]. (e) Complete three-wavelength SPAD-based pulse oximeter system. Adapted from Ref. [57].
Green PPG. Green-light PPG, typically within the 525–565 nm range, provides shallower penetration but superior signal-to-noise ratios for detecting pulsatile blood flow, making it particularly well suited for wearable devices such as smartwatches and fitness trackers. Corresponding with this trend, Ahmmed et al. developed a wearable wristband incorporating a compressed sensing (CS)-based ultralow-power PPG ASIC, representing the first on-chip CS-enabled PPG system for energy-efficient cardiovascular monitoring [54]. To address the challenge of motion artifacts prevalent in wrist-based green PPG devices, Xue et al. designed a lightweight and compact sensor optimized for continuous heart rate measurement (Fig. 5 (c)) [14]. By leveraging the anatomical advantages of the earlobe, the system demonstrated reliable heart rate detection, even during exercise.
Multiwavelength PPG. The increasing interest in multiwavelength photoplethysmography (MWPPG) has extended the scope of PPG beyond conventional SpO₂ and heart rate monitoring, thereby enabling more comprehensive cardiovascular assessments. For instance, Li et al. proposed an MWPPG system integrated with temperature and humidity sensors combined with an attention-based neural network to achieve non-invasive blood glucose monitoring [55]. Similarly, Cui et al. introduced a four-wavelength PPG fusion approach employing an attention-based convolutional neural network and bidirectional long short-term memory architecture for cuffless BP measurements (Fig. 5 (d)) [56]. Cusini et al. further advanced the field by developing the first contact-mode single-photon avalanche diode (SPAD)-based pulse oximeter, representing the initial demonstration of contact-mode SPAD-enabled MWPPG for simultaneous HR and SpO₂ monitoring (Fig. 5 (e)) [57]. Additionally, Franklin et al. reported a wireless skin-interfaced platform that integrates a chest-worn ECG with peripheral MWPPG sensors, thereby facilitating continuous hemodynamic monitoring and precise classification of cardiovascular states [58].
Remote PPG (rPPG). Outside wearable sensor innovation, demand for non-contact health monitoring is increasing. rPPG has emerged as a promising approach for extracting heart rate information by analyzing subtle skin color fluctuations [59-61]. Within this domain, Liu et al. proposed a self-supervised rPPG framework based on a masked autoencoder, which enables the extraction of physiological signals without requiring labeled datasets [62]. Zhang et al. developed a deep-learning-based method that demonstrated robustness for contactless heart rate monitoring under real-world conditions [63]. Further advancing this area, Li et al. introduced an end-to-end 3D spatio-temporal convolutional network augmented with a multi-hierarchical feature fusion (MHFF) attention module, which significantly improved the accuracy and robustness of rPPG signal extraction from facial videos [64].
2.5 Electrochemical Sensors
Innovation in wireless electrochemical systems for monitoring sweat, blood, and ISF, provides new opportunities for real-time physiological tracking. Sweat is an attractive biofluid because it can be collected non-invasively and continuously, offering access to electrolytes, metabolites, and other biomarkers. Over the past decade, sweat sensors have evolved from rigid, bench-top systems to soft, skin-conformal, and wireless devices. Unlike traditional laboratory sensors, wireless sweat sensors support on-body monitoring during daily activities, exercise, and clinical observations. Their minimal invasiveness makes them particularly promising for personalized health tracking and early disease detection.
To enable continuous measurements, recent sweat sensors integrate soft microfluidics with wireless modules, eliminating the need for bulky equipment. A notable early example by Gao et al. introduced a flexible sweat patch that routes sweat through microfluidic channels and measures analytes using colorimetric sensors [65]. Building on this foundation, He et al. developed a textile-based sensor that is both stretchable and breathable, with sweat traveling through embedded microchannels to reach electrochemical sensors for glucose detection [66]. A compact BLE module transmits data in real time to a smartphone application, thereby enhancing usability. Fig. 6 (a) highlights the glucose-sensing performance with representative demonstration of selectivity against interfering substances (Na⁺, K⁺, lactate, uric acid, ascorbic acid). Because an efficient power supply mechanism is of interest in designing wireless platforms, several groups have explored self-sustaining solutions. Zhao et al. proposed a freestanding electrochemical patch without a rigid backing, where a microfluidic layer directs sweat to the electrodes, whereas a compact wireless module transmits data with minimal power consumption [67]. Cao et al., demonstrated a powering scheme using a piezoelectric ceramic harvester that converts mechanical energy from body motion into electricity [68], keeping the device lightweight and energy-efficient.
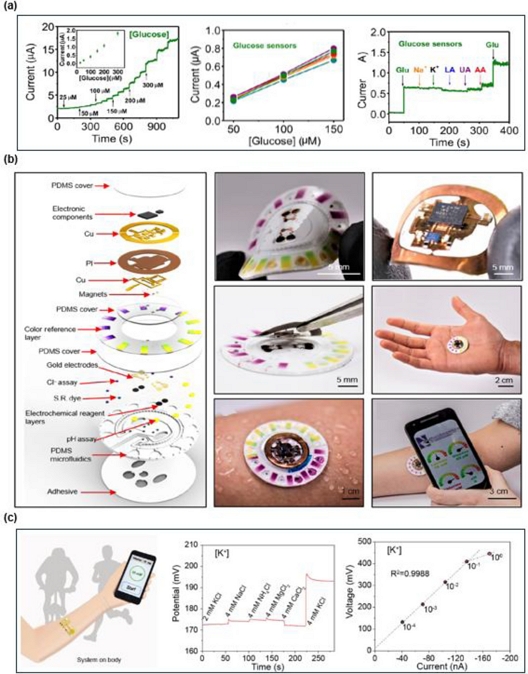
Design and performance highlights of wireless sweat sensors. (a) Electrochemical performance and selectivity of textile-based sensor for glucose detection in sweat. Adapted from Ref. [66]. (b) Battery-free sweat analysis patch integrating microfluidics, multimodal sensing, and wireless NFC transmission. Adapted from Ref. [69]. (c) Calibration performance of the KRSM sensor across potassium ion concentrations, showing high linearity. Adapted from Ref. [70].
Beyond physical properties, such as sweat rate and conductivity, emphasis has shifted toward detecting biochemical markers in real time. Bandodkar et al. developed a battery-free microfluidic/electronic platform that integrates biofuel-cell-based electrochemical sensors with colorimetric assays (Fig. 6 (b)) [69]. A disposable soft microfluidic layer guides sweat into separate compartments for glucose, lactate, chloride, pH, and sweat loss, thereby minimizing cross-contamination. A detachable NFC module couples magnetically to the patch, enabling wireless power harvesting and smartphone-based readouts.
Wu et al. further advanced sweat sensing inspired by insect olfaction [70]. Their Kevlar aramid nanofiber membrane (KANM) mimics the selective pores of insect sensilla, capturing potassium ions (K⁺) with high specificity. As shown in Fig. 6 (c), the Kevlar-reinforced separation-sensing membrane (KRSM) sensor provides a near-linear voltage response to K⁺, even in the presence of competing ions. Similarly, Tu et al. introduced a BLE-enabled wireless patch that detects C-reactive protein (CRP) via an electrochemical immunoassay, supporting real-time inflammation tracking for chronic disease management [71]. Other designs integrate fluidic and biochemical information to improve the reliability of analysis. Kwon et al. developed a platform that combines sweat flow rate and temperature sensing with biochemical detection, accounting for dilution and temporal variability [72]. Their system continuously tracks cumulative sweat loss and transmits data wirelessly, thereby improving the accuracy of sweat-based biomarker interpretation.
Blood and ISF are excellent sources of physiological information. Compared with sweat, these fluids exhibit tighter homeostatic regulation, making them more suitable for clinical-grade sensing. ISF is particularly attractive because of its biochemical similarity to blood plasma and accessibility through minimally invasive approaches. Recent epidermal devices employ microneedle arrays or porous membranes to painlessly extract ISF, enabling the real-time electrochemical detection of glucose and electrolytes. Electrochemical sensing, which has dominated the global glucose monitoring market for over four decades because of its sensitivity, low cost, and ease of use, remains the backbone of these systems [73]. In addition, antenna-based biosensors are emerging that leverage microwave or radiofrequency (RF) interactions with glucose-dependent dielectric changes in subcutaneous tissues for non-invasive monitoring [74,75].
Early approaches focused on ISF extraction using microneedles and thin-film electronics. Chen et al. designed a skin-like biosensor with embedded electrochemical channels for enzymatic glucose monitoring [76]. Lipani et al. advanced this concept using a graphene-based patch that enhances selectivity and skin compatibility [77]. More recently, Pu et al. reported a thermally activated microfluidic platform that integrates microheaters with iontophoresis electrodes, significantly improving the ISF sampling efficiency, as shown in Fig. 7 (a) [78].
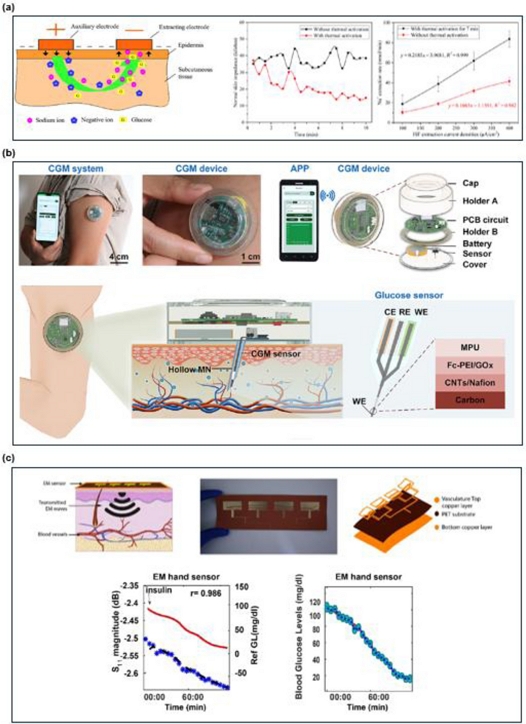
Recent advances in wireless blood/ISF monitoring platforms. (a) Thermal activation to improve the efficiency of transdermal ISF extraction. Adapted from Ref. [78]. (b) Smartphone-integrated microneedle-based continuous glucose monitoring (CGM) system and glucose sensor architecture. Adapted from Ref. [16]. (c) In-vivo validation of antenna-based wireless glucose monitoring using electromagnetic sensors. Adapted from Ref. [81].
Multifunctional wireless ISF platforms are gaining popularity. Yang et al. demonstrated a smartphone-controlled microneedle patch that integrates ISF extraction, ferrocene-mediated enzymatic detection, and a wireless data display (Fig. 7 (b)) [16]. Similarly, Yang et al. proposed a masticatory-system-inspired microneedle theragnostic platform (MNTP) that combines biosensing with motion-triggered insulin delivery [79]. The system employs hybrid carbon-nanomaterial-based electrochemical sensing integrated with flexible electronics to achieve closed-loop glucose regulation.
Antenna-based biosensors offer another promising approach for non-invasive glucose sensing. Xiao et al. employed ultra-wideband microwave spectrometry to analyze S-parameters across broad frequencies to estimate glucose concentrations [80]. Hamouda et al. further miniaturized flexible antennas using magnetodielectric nanocomposites and achieved heightened sensitivity to physiological dielectric changes [75]. Lv et al. enhanced the selectivity by integrating complementary split-ring resonators (CSRRs) into an antenna, enabling resonance-based glucose detection [74]. To estimate glucose, Hanna et al. demonstrated a quasi-antenna array system with highly accurate clinical trial results by incorporating environmental and physiological compensation and applying Gaussian process regression (Fig. 7 (c)) [81].
2.6 Mechanical and Acoustic Sensors
An IMU is a motion-sensing device that measures linear acceleration, angular velocity, and orientation by combining accelerometers, gyroscopes, and sometimes magnetometers [82,83]. IMUs are increasingly used in wearable healthcare for the continuous tracking of movement and posture in infants, athletes, and the elderly. For infants, they support the monitoring of motor development and interventions, such as kangaroo care [9,84]. For athletes and general users, IMUs aid rehabilitation, posture correction, and gait analysis for performance optimization or injury recovery [85,86]. In older adults, IMUs are applied to fall prevention, gait assessment, and monitoring movement patterns associated with dementia and Parkinson’s disease [87,88]. This review summarizes the current research trends in IMU technologies across these groups.
Wireless IMUs have proven effective for the unobtrusive monitoring of posture and movement in daily and clinical contexts. Airaksinen et al. developed a motor ability assessment of infants using the JUmpsuit (MAIJU), a wearable jumpsuit with multiple inertial sensors, enabling continuous monitoring of spontaneous motor behaviors in infants aged 5–19 months (Figs. 8 (a)–(c)) [84]. Chung et al. designed a soft skin-interfaced IMU platform for neonatal intensive care units, enabling continuous posture monitoring during kangaroo care (Fig. 8 (d) and (e)) [9]. Collectively, these systems demonstrate how wireless IMUs can provide automated and objective assessments of infant motor development both at home and in hospitals.
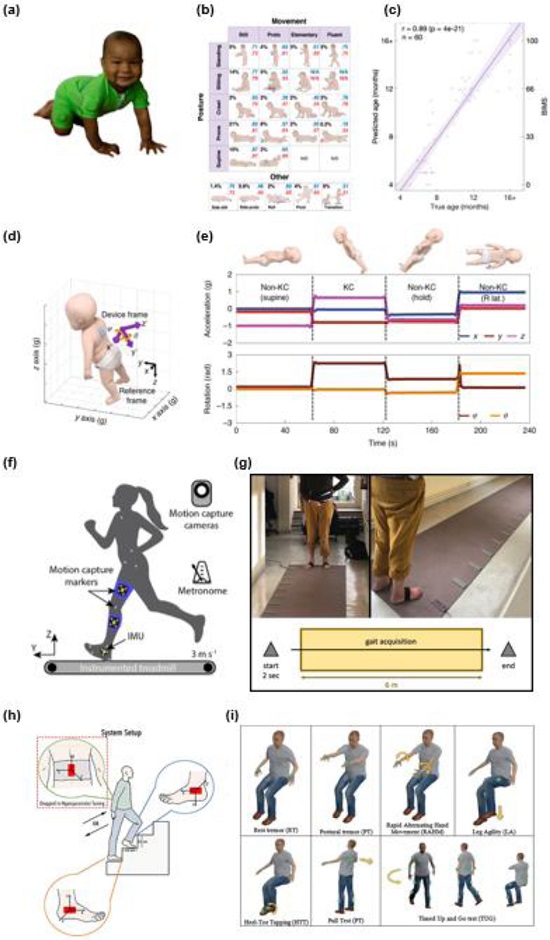
Representative wireless IMU systems for movement monitoring across different age groups. (a-c) MAIJU wearable jumpsuit for infants. Adapted from Ref. [84]. (d, e) Soft, skin-interfaced IMU platform for neonatal monitoring during kangaroo care. Adapted from Ref. [9]. (f) IMU-based system for predicting overstriding during running by measuring shank motion. Adapted from Ref. [86]. (g) Foot-mounted IMU system. Adapted from Ref. [85]. (h) Wireless IMUs on lower limbs and pelvis for stair-climbing assessment. Adapted from Ref. [87]. (i) Seven IMU-based motor tasks for classifying early-stage Parkinson’s disease. Adapted from Ref. [91].
For healthy individuals and athletes, IMUs enable real-time motion tracking outside a laboratory environment. Baker et al. developed a shank-mounted IMU system that predicts overstriding during running, detecting gait abnormalities linked to injury risk under natural conditions (Fig. 8 (f)) [86]. Kim and Park mounted IMUs directly on paddles to analyze stroke orientation and timing; the parameters were not easily captured using body-mounted sensors [89]. For patients with gait impairments, Voisard et al. introduced a foot-mounted IMU method using adaptive thresholding and temporal pattern analysis to identify gait events without external references, proving its accuracy even for severe deficits (Fig. 8 (g)) [85]. Unger et al. further showed that IMUs detect clinically meaningful motor differences after a stroke, supporting their use in portable and cost-effective rehabilitation [90]. Collectively, these studies highlighted the flexibility of IMUs in sports, movement correction, and remote physiotherapy.
For older adults, IMUs are applied to mobility monitoring, fall detection, and early disease diagnosis. Tseng et al. developed a fall detection system integrating IMUs with GNSS and NB-IoT communication for reliable localization and rapid alerts, reducing the false alarms common in threshold-based methods [88]. Holloway et al. employed lower-limb- and pelvis-mounted IMUs to capture kinematic features during stair climbing in patients with dementia, achieving >87% accuracy in classifying dementia subtypes (Fig. 8 (h)) [87]. Lazzaro et al. used seven IMU-based motor tasks to classify early-stage Parkinson’s disease with up to 97% accuracy, with the heel-to-toe, timed up and go (TUG), and pull tests identified as the most sensitive indicators (Fig. 8 (i)) [91]. These advances underscore the potential of IMUs supporting independence, improving safety, and enabling early diagnosis in the elderly people.
The arterial pulse, generated by rhythmic cardiac contractions, provides insights into ventricular function, arterial compliance, and autonomic regulation [92-94]. Continuous monitoring enables early cardiovascular diagnosis and long-term management. Although PPG and cuff-based BP methods are common, they suffer from motion artifacts, posture dependence, and limited comfort [95,96]. To overcome these limitations, wireless mechano-acoustic sensing platforms have emerged, leveraging physical manifestations of cardiovascular activity—vibrations, body recoil, skin deformation, and internal acoustic waves—via four modalities: seismocardiography (SCG), ballistocardiography (BCG), pressure sensing, and acoustic sensing [17,92,97,98].
SCG measures the micro-vibrations of the chest wall induced by cardiac mechanical events, offering correlations distinct from ECG [92,99]. Lee et al. developed a soft wireless mechano-acoustic patch integrating a triaxial accelerometer and BLE module, enabling real-time streaming and the detection of features such as aortic valve opening during daily activities [97]. Guess et al. introduced a battery-less SCG patch powered by NFC, eliminating recharging and enabling long-term monitoring (Fig. 9 (a) and (b)) [98]. These systems demonstrate the suitability of SCG for high-resolution ambulatory cardiac assessment. BCG captures whole-body recoil forces from ventricular ejection, reflecting the stroke volume and cardiac output [94]. Zhou et al. designed a flexible wireless BCG patch capable of simultaneous respiration and cardiac monitoring using signal separation algorithms in daily life conditions (Figs. 9 (c–e)) [93]. Fu et al. embedded accelerometers into commercial earbuds to detect cranial recoil forces, enabling unobtrusive cardiovascular monitoring during routine activities, such as sleep [94].
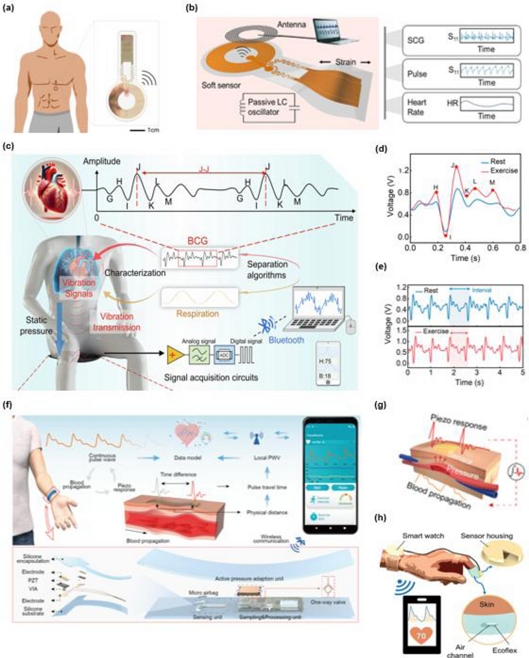
Wireless pulse monitoring systems based on mechanical and acoustic sensing principles. (a, b) Soft wireless SCG patch with BLE transmission and battery-less NFC-powered SCG platform. Adapted from Ref. [98]. (c–e) Wireless BCG systems. Adapted from Ref. [93]. (f, g) Ultra-thin PZT-based mechano-acoustic pressure sensor. Adapted from Ref. [101]. (h) Flexible mechano-acoustic sensor with soft air channel for low-frequency body sound monitoring. Adapted from Ref. [17].
Pressure sensors capture skin deformation from pulsatile blood flow, enabling cuffless estimation of BP and pulse wave velocity (PWV) [100,101]. Zhao et al. developed a multichannel pressure-sensing platform with flexible arrays and active airbag control by applying machine learning to digitize the multi-pressure pulse diagnosis [100]. Li et al. reported an ultrathin PZT-based pressure sensor with a micro-airbag interface for optimal radial artery contact, achieving high accuracy through PWV analysis (Fig. 9 (f) and (g)) [101]. Micro-electro-mechanical system (MEMS) microphones and ultrasound transducers can capture low-frequency cardiac sounds (S1/S2) and vessel wall reflections. Hoang et al. created a wearable mechano-acoustic sensor that transmits vibrations via a soft Ecoflex air channel to a piezoresistive element, enabling long-term motion-artifact-resistant monitoring for daily use (Fig. 9 (h)) [17]. Zhou et al. developed a soft ultrasound patch for continuous central BP monitoring by imaging carotid artery dynamics, bridging high-fidelity clinical imaging and portable wearables [102].
3. DISCUSSIONS
Wireless soft sensors represent a transformative advance in continuous physiological monitoring; however, several barriers remain before widespread clinical and consumer adoption can be achieved. A persistent challenge is maintaining a stable performance during long-term wear. Daily movements, perspiration, and skin regeneration can disrupt the sensor–skin interface, producing motion artifacts or reducing signal fidelity. Therefore, the development of advanced adhesives, breathable encapsulants, and mechanically adaptive materials remains a key priority for balancing durability and comfort.
Non-contact sensing approaches offer advantages in terms of comfort and hygiene, but their reliability is often compromised by environmental factors such as distance, temperature, and humidity. However, their sensitivity limits their use in high-precision applications. Furthermore, the shift from wired to wireless monitoring has introduced new complexities in clinical settings. Wireless systems must ensure secure data transmission, avoid electromagnetic interference and comply with strict hospital interoperability standards. Power management is another critical challenge because continuous monitoring demands efficient low-power operation without frequent recharging or bulky power sources.
However, the scalability and manufacturing pose additional challenges. Physiological variability—resulting from differences in skin tone, tissue thickness, sweat levels, or movement patterns—can affect measurement accuracy, complicating both individualized performance and large-scale deployment. More extensive clinical validation is required, particularly among populations that would benefit most from these technologies, including neonates, elderly patients, and individuals with chronic diseases or mobility limitations.
Progress depends on the integration of multimodal sensing platforms that can cross-validate signals, AI-driven analytics that enhance the robustness in real-world environments, and low-power wireless communication protocols that ensure seamless connectivity. In particular, multimodal systems mitigate the limitations of individual sensing mechanisms, while offering richer physiological insights. By combining data from different sensor types, these systems can generate more robust and comprehensive physiological profiles. However, these synergies are accompanied by trade-offs between complexity and power demand. Addressing these technical, clinical, and scalability challenges is essential to achieving the full promise of wireless physiological monitoring in preventive, acute, and personalized medicine.
4. CONCLUSIONS
Although conventional wired physiological monitoring systems are accurate and reliable, they restrict mobility, cause skin irritation during prolonged use, and are poorly suited to out-of-hospital care. Recent advances in wireless sensing technology have addressed these limitations by enabling more flexible, comfortable, and patient-friendly monitoring methods.
This review categorizes wireless physiological sensors into five groups: thermoelectric, bioelectrical, optical, biochemical, and mechano-acoustic, reflecting the diversity of biological processes that can be monitored. Across these domains, the key innovations include the use of stretchable and biocompatible materials for skin conformity, low-power communication protocols such as BLE and NFC for real-time connectivity, and multimodal integration for improved accuracy. These advances have expanded physiological monitoring from hospital settings to homes, workplaces, and ambulatory environments, thereby supporting a shift toward proactive and personalized healthcare.
However, several significant challenges remain unresolved. Motion artifacts, unstable skin–sensor interfaces, and physiological variability complicate long-term real-world use. Many technologies are still in the proof-of-concept stage, with limited large-scale clinical validation. To achieve widespread adoption, future research should focus on developing sensor platforms that function reliably under real-life conditions while also pursuing clinical validation in populations that will benefit the most, such as neonates, older adults, and patients with limited mobility.
Advances in low-power communication, multimodal integration, and AI-driven signal processing are expected to play a key role in accelerate the practical deployment of wireless physiological monitoring technologies. In addition, the next stage of innovation is likely to involve closed-loop systems combining continuous sensing with therapeutic feedback, thereby enabling proactive health management. Establishing real-world evidence through large-scale, long-term studies is also critical for ensuring regulatory acceptance and clinical trust. Finally, the development of personalized algorithms that adapt to individual physiology can transform wireless sensors from generic monitoring devices into precision tools for tailored healthcare applications.
Acknowledgments
This work was supported by grants from the Korea University (K2509951 and K2509861). This research was supported by a grant of Korean ARPA-H Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: RS-2025-25454431).
References
-
I.J. Brekke, L.H. Puntervoll, P.B. Pedersen, J. Kellett, M. Brabrand, The value of vital sign trends in predicting and monitoring clinical deterioration: A systematic review, PloS one 14 (2019) e0210875.
[https://doi.org/10.1371/journal.pone.0210875]

-
T. Lu, S. Ji, W. Jin, Q. Yang, Q. Luo, T.L. Ren, Biocompatible and long-term monitoring strategies of wearable, ingestible and implantable biosensors: reform the next generation healthcare, Sensors 23 (2023) 2991.
[https://doi.org/10.3390/s23062991]

-
M.L. Morales-Botello, D. Gachet, M. de Buenaga, F. Aparicio, M.J Busto, J.R Ascanio, Chronic patient remote monitoring through the application of big data and internet of things, Health Inform. J. 27 (2021) 1–18.
[https://doi.org/10.1177/14604582211030956]

-
K.R. De Guzman, C.L. Snoswell, M.L. Taylor, L.C. Gray, L.J. Caffery, Economic evaluations of remote patient monitoring for chronic disease: a systematic review, Value Health 25 (2022) 897–913.
[https://doi.org/10.1016/j.jval.2021.12.001]

-
D.G. Leo, B.J.R. Buckley, M. Chowdhury, S.L. Harrison, M. Isanejad, G.Y.H. Lip, et al., Interactive remote patient monitoring devices for managing chronic health conditions: systematic review and meta-analysis, J. Med. Internet Res. 24 (2022) e35508.
[https://doi.org/10.2196/35508]

-
D.K. Ming, S. Sangkaew, H.Q. Chanh, P.T.H. Nhat, S. Yacoub, P. Georgiou, et al., Continuous physiological monitoring using wearable technology to inform individual management of infectious diseases, public health and outbreak responses, Int. J. Infect. Dis. 2020 (2020) 648–654.
[https://doi.org/10.1016/j.ijid.2020.05.086]

-
C. Lund, Medical adhesives in the NICU, Newborn Infant Nurs. Rev. 14 (2014) 160–165.
[https://doi.org/10.1053/j.nainr.2014.10.001]

-
H.U. Chung, B.H. Kim, J.Y. Lee, J. Lee, Z. Xie, E.M. Ibler, et al., Binodal, wireless epidermal electronic systems with in-sensor analytics for neonatal intensive care, Science 363 (2019) eaau0780.
[https://doi.org/10.1126/science.aau0780]

-
H.U. Chung, A.Y. Rwei, A. Hourlier-Fargette, S. Xu, K.H. Lee, E.C. Dunne, et al., Skin-interfaced biosensors for advanced wireless physiological monitoring in neonatal and pediatric intensive-care units, Nat. Med. 26 (2020) 418–429.
[https://doi.org/10.1038/s41591-020-0792-9]

-
S.S. Kwak, S. Yoo, R. Avila, H.U. Chung, H. Jeong, C. Liu, et al., Skin‐integrated devices with soft, holey architectures for wireless physiological monitoring, with applications in the neonatal intensive care unit, Adv. Mater. 33 (2021) 2103974.
[https://doi.org/10.1002/adma.202103974]

-
D. Ryu, D.H. Kim, J.T. Price, J.Y. Lee, H.U. Chung, E. Allen, et al., Comprehensive pregnancy monitoring with a network of wireless, soft, and flexible sensors in high-and low-resource health settings, Proc. Natl. Acad. Sci. U.S.A. 118 (221) e2100466118.
[https://doi.org/10.1073/pnas.2100466118]

-
W. Zhang, L. Li, Y. Wang, X. Dong, C. Liu, L. Sun, et al., Continuous Core Body Temperature Monitoring for Heatstroke Alert via a Wearable In-Ear Thermometer, ACS Sens. 10 (2025) 1440–1449.
[https://doi.org/10.1021/acssensors.4c03470]

-
R. Kaveh, C. Schwendeman, L. Pu, A.C. Arias, R. Muller, Wireless ear EEG to monitor drowsiness, Nat. Commun. 15 (2024) 6520.
[https://doi.org/10.1038/s41467-024-48682-7]

-
Q. Xue, D. Nissanka, J.T. Yan, R. Wang, S. Patel, V. Iyer, PPG Earring: Wireless Smart Earring for Heart Health Monitoring, Proceedings of the 2025 CHI Conference on Human Factors in Computing Systems, Yokohama, Japan, 2025, pp. 1–16.
[https://doi.org/10.1145/3706598.3713856]

-
M. Zadan, A. Wertz, D. Shah, D.K. Patel, W. Zu, Y. Han, et al., Stretchable thermoelectric generators for self‐powered wearable health monitoring, Adv. Funct. Mater. 34 (2024) 2404861.
[https://doi.org/10.1002/adfm.202404861]

-
J. Yang, X. Gong, S. Chen, Y. Zheng, L. Peng, B. Liu, et al., Development of smartphone-controlled and microneedle-based wearable continuous glucose monitoring system for home-care diabetes management, ACS Sens. 8 (2023) 1241–1251.
[https://doi.org/10.1021/acssensors.2c02635]

-
T.T. Hoang, A.M. Cunio, S. Zhao, T.V. Nguyen, S. Peng, S. Liaw, et al., Flexible, Wearable Mechano‐Acoustic Sensors for Real‐Time, Wireless Monitoring of Low Frequency Body Sounds, Adv. Sens. Res. 3 (2024) 2400039.
[https://doi.org/10.1002/adsr.202400039]

-
J. Kim, A.S. Campbell, B.E.F. de Ávila, J. Wang, Wearable biosensors for healthcare monitoring, Nat. Biotechnol. 37 (2019) 389–406.
[https://doi.org/10.1038/s41587-019-0045-y]

-
L.P.J. Teunissen, J. Klewer, A. De Haan, J.J. De Koning, H.A.M. Daanen, Non-invasive continuous core temperature measurement by zero heat flux, Physiol. Meas. 32 (2011) 559.
[https://doi.org/10.1088/0967-3334/32/5/005]

-
H.A.M. Daanen, V. Kohlen, L.P.J. Teunissen, Heat flux systems for body core temperature assessment during exercise, J. Therm. Biol. 112 (2023) 103480.
[https://doi.org/10.1016/j.jtherbio.2023.103480]

-
S.L. Lauronen, M.L. Kalliomäki, J. Kalliovalkama, A. Aho, H. Huhtala, A.M. Yli-Hankala, et al., Comparison of zero heat flux and double sensor thermometers during spinal anaesthesia: a prospective observational study, J. Clin. Monit. Comput. 36 (2022) 1547–1555.
[https://doi.org/10.1007/s10877-021-00799-6]

-
N. Panunzio, G. Marrocco, Flexible and wireless multi-sensor thermometer based on dual-heat-flux model, Proceedings of the 2022 IEEE 12th International Conference on RFID Technology and Applications (RFID-TA), Cagliari, Italy, 2022, pp. 29–32.
[https://doi.org/10.1109/RFID-TA54958.2022.9924064]

-
M. Lazaro, A. Lazaro, R. Villarino, D. Girbau, Smart face mask with an integrated heat flux sensor for fast and remote people’s healthcare monitoring, Sensors 21 (2021) 7472.
[https://doi.org/10.3390/s21227472]

-
Y. Hashimoto, S. Tada, Y. Nishida, Reference-free calibration for wearable core body temperature sensor based on single heat flux method, IEEE Sens. Lett. 8 (2024) 1–4.
[https://doi.org/10.1109/LSENS.2024.3435965]

-
I. Kato, H. Watanabe, K. Nagashima, Evaluation of newly developed wearable ear canal thermometer, mimicking the application to activities on sports and labor fields, J. Physiol. Sci. 73 (2023) 15.
[https://doi.org/10.1186/s12576-023-00874-4]

-
Y. Du, C. Zhou, Y. Feng, L. Qiu, Flexible, multimodal device for measurement of body temperature, core temperature, thermal conductivity and water content, npj Flex. Electron. 8 (2024) 85.
[https://doi.org/10.1038/s41528-024-00373-5]

-
S. Oh, J.Y. Yoo, W.Y. Maeng, S. Yoo, T. Yang, S.M. Slattery, et al., Simple, miniaturized biosensors for wireless mapping of thermoregulatory responses, Biosens. Bioelectron. 237 (2023) 115545.
[https://doi.org/10.1016/j.bios.2023.115545]

-
E.S. Dahiya, A.M. Kalra, A. Lowe, G. Anand, Wearable technology for monitoring electrocardiograms (ECGs) in adults: a scoping review, Sensors 24 (2024) 1318.
[https://doi.org/10.3390/s24041318]

-
L. Neri, M.T. Oberdier, K.C.J. Van Abeelen, L. Menghini, E. Tumarkin, H. Tripathi, et al., Electrocardiogram monitoring wearable devices and artificial-intelligence-enabled diagnostic capabilities: a review, Sensors 23 (2023) 4805.
[https://doi.org/10.3390/s23104805]

-
Q. Zhuang, K. Yao, X. Song, Q. Zhang, C. Zhang, H. Wang, et al., An ICU-grade breathable cardiac electronic skin for health, diagnostics, and intraoperative and postoperative monitoring, Sci. Adv. 11 (2025) eadu3146.
[https://doi.org/10.1126/sciadv.adu3146]

-
S. Rauf, R.M. Bilal, J. Li, M. Vaseem, A.N. Ahmad, A. Shamim, Fully screen-printed and gentle-to-skin wet ECG electrodes with compact wireless readout for cardiac diagnosis and remote monitoring, ACS Nano 18 (2024) 10074–10087.
[https://doi.org/10.1021/acsnano.3c12477]

-
G.D. Clifford, I. Silva, J. Behar, G.B Moody, Non-invasive fetal ECG analysis, Physiol. Meas. 35 (2014) 1521.
[https://doi.org/10.1088/0967-3334/35/8/1521]

-
D.D. Kanjoor Nair, S. Gilvaz, B. Menon, P. Singh, Accuracy of a Noninvasive, Wearable, Wireless, ECG-Based, Intrapartum Monitoring Tool Against the Conventional Ultrasound-Based CTG, J. Obstet. Gynaecol. India 75 (2024) 340–347.
[https://doi.org/10.1007/s13224-024-01998-2]

-
X.Y. Liu, W.L. Wang, M. Liu, M.Y. Chen, T. Pereira, D.Y. Doda, et al., Recent applications of EEG-based brain-computer-interface in the medical field, Mil. Med. Res. 12 (2025) 14.
[https://doi.org/10.1186/s40779-025-00598-z]

-
G. Buzsáki, C.A. Anastassiou, C. Koch, The origin of extracellular fields and currents—EEG, ECoG, LFP and spikes, Nat. Rev. Neurosci. 13 (2012) 407–420.
[https://doi.org/10.1038/nrn3241]

-
Q. Zhao, E. Gribkova, Y. Shen, J. Cui, N. Naughton, L. Liu, et al., Highly stretchable and customizable microneedle electrode arrays for intramuscular electromyography, Sci. Adv. 10 (2024) eadn7202.
[https://doi.org/10.1126/sciadv.adn7202]

-
C. Zhou, G. Yao, X. Gan, K. Chai, P. Li, J. Peng, et al., Modulus-adjustable and mechanically adaptive dry microneedle electrodes for personalized electrophysiological recording, npj Flex. Electron. 9 (2025) 77.
[https://doi.org/10.1038/s41528-025-00458-9]

-
T. Kim, I. Park, Skin-interfaced wearable biosensors: a mini-review, J. Sens. Sci. Technol. 31 (2022) 71–78.
[https://doi.org/10.46670/JSST.2022.31.2.71]

-
D. Im, B.J. Kim, G.H. Kim, T. An, Effect of Adhesion Strength Between Flexible Substrates and Electrodes on the Durability of Electrodes, J. Sens. Sci. Technol. 33 (2024) 86–92.
[https://doi.org/10.46670/JSST.2024.33.2.86]

- A. John, B. Cardiff, D. John, A Review on Multisensor Data Fusion for Wearable Health Monitoring, arXiv., arXiv:2412.05895, (2024).
-
E. Gkintoni, A. Aroutzidis, H. Antonopoulou, C. Halkiopoulos, From neural networks to emotional networks: A systematic review of EEG-based emotion recognition in cognitive neuroscience and real-world applications, Brain Sci. 15 (2025) 220.
[https://doi.org/10.3390/brainsci15030220]

-
L. Zhou, M. Guess, K.R. Kim, W.H. Yeo, Skin-interfacing wearable biosensors for smart health monitoring of infants and neonates, Commun. Mater. 5 (2024) 72.
[https://doi.org/10.1038/s43246-024-00511-6]

-
J.N. Wong, J.R. Walter, E.C. Conrad, D.R. Seshadri, J.Y. Lee, H. Gonzalez, et al., A comprehensive wireless neurological and cardiopulmonary monitoring platform for pediatrics, PLOS Digit. Health 2 (2023) e0000291.
[https://doi.org/10.1371/journal.pdig.0000291]

- N.L. Wickramasinghe, D.S. Udayantha, A. Abeyratne, K. Weerasinghe, K. Wickremasinghe, A. De Silva, et al., Improving Neonatal Care: An Active Dry-Contact Electrode-based Continuous EEG Monitoring System with Seizure Detection, arXiv., arXiv:2503.23338, (2025).
-
Y. Jiang, M. Tian, J. Zhang, J. Li, C. Tan, C. Ren, et al., IEMS: An IoT-empowered wearable multimodal monitoring system in neurocritical care, IEEE Internet Things J. 10 (2022) 1860–1875.
[https://doi.org/10.1109/JIOT.2022.3210930]

-
S.J. van Bohemen, J.M. Rogers, A. Alavanja, A. Evans, N. Young, P.C. Boughton, et al., Safety, feasibility, and acceptability of a novel device to monitor ischaemic stroke patients, J. Med. Eng. Technol. 48 (2024) 173–185.
[https://doi.org/10.1080/03091902.2024.2409115]

-
X. Tang, W. Chen, S. Mandal, K. Bi, T. Özdemir, High-sensitivity electric potential sensors for non-contact monitoring of physiological signals, IEEE Access 10 (2022) 19096–19111.
[https://doi.org/10.1109/ACCESS.2022.3150587]

-
W. Huang, W. Wu, M.V. Lucas, H. Huang, Z. Wen, Y. Li, Neurofeedback training with an electroencephalogram-based brain-computer interface enhances emotion regulation, IEEE Trans. Affect. Comput. 14 (2021) 998–1011.
[https://doi.org/10.1109/TAFFC.2021.3134183]

-
A.A. Alian, K.H. Shelley, Photoplethysmography, Best Pract. Res. Clin. Anaesthesiol. 28 (2014) 395–406.
[https://doi.org/10.1016/j.bpa.2014.08.006]

-
J. Koseeyaporn, P. Wardkein, A. Sinchai, P. Kainan, P. Tuwanut, Pulse oximetry based on quadrature multiplexing of the amplitude modulated photoplethysmographic signals, Sensors 23 (2023) 6106.
[https://doi.org/10.3390/s23136106]

-
Y. Lee, J.W. Chung, G.H. Lee, H. Kang, J.Y. Kim, C. Bae, et al., Standalone real-time health monitoring patch based on a stretchable organic optoelectronic system, Sci. Adv. 7 (2021) eabg9180.
[https://doi.org/10.1126/sciadv.abg9180]

-
S. Kumar, R. Kedia, A. Bisht, A. Soni, R.K. Sharma, S. Yadav, et al., Novel Flexible Organic Photoplethysmogram Sensor for Continuous Cardiovascular Monitoring, ACS Sens. 10 (2025) 4297–4306.
[https://doi.org/10.1021/acssensors.5c00552]

-
S. Li, J.H. Jang, W. Chung, H. Seung, S.I. Park, H. Ma, et al., Ultrathin self-powered heavy-metal-free Cu–In–Se quantum dot photodetectors for wearable health monitoring, ACS Nano 17 (2023) 20013–20023.
[https://doi.org/10.1021/acsnano.3c05178]

-
P. Ahmmed, E. Garceau, T. Latif, A. Brewer, J. Dieffenderfer, J.M. Valero-Sarmiento, et al., Preclinical Evaluation of a Wearable Wristband With Compressed-Sensing Based Photoplethysmography, IEEE Trans. Biomed. Eng. 72 (2024) 1596–1604.
[https://doi.org/10.1109/TBME.2024.3513462]

-
T. Li, Q. Wang, L. Lei, Y. An, L. Guo, L. Ren, et al., Improvement of Non-invasive Glucose Estimation Accuracy through Multi-wavelength PPG, IEEE J. Biomed. Health Inform. 29 (2025) 5465–5478.
[https://doi.org/10.1109/JBHI.2025.3556666]

-
M. Cui, X. Dong, Y. Zhuang, S. Li, S. Yin, Z. Chen, et al., ACNN-BiLSTM: A Deep Learning Approach for Continuous Noninvasive Blood Pressure Measurement Using Multi-Wavelength PPG Fusion, Bioengineering 11 (2024) 306.
[https://doi.org/10.3390/bioengineering11040306]

-
I. Cusini, R. Rinaldi, P. Castiglioni, A. Faini, F. Villa, Multi-wavelength SPAD photoplethysmography for cardio-respiratory monitoring, Front. Phys. 11 (2023) 952103.
[https://doi.org/10.3389/fphy.2023.952103]

-
D. Franklin, A. Tzavelis, J.Y. Lee, H.U. Chung, J. Trueb, H. Arafa, et al., Synchronized wearables for the detection of haemodynamic states via electrocardiography and multispectral photoplethysmography, Nat. Biomed. Eng. 7 (2023) 1229–1241.
[https://doi.org/10.1038/s41551-023-01098-y]

-
R.J. Lee, S. Sivakumar, K.H. Lim, Review on remote heart rate measurements using photoplethysmography, Multimed. Tools Appl. 83 (2024) 44699–44728.
[https://doi.org/10.1007/s11042-023-16794-9]

-
Z. Tang, H. Hu, C. Xu, K. Zhao, Exploring an efficient remote biomedical signal monitoring framework for personal health in the COVID-19 pandemic, Int. J. Environ. Res. Public Health 18 (2021) 9037.
[https://doi.org/10.3390/ijerph18179037]

-
A.R. Watson, R. Wah, R. Thamman, The value of remote monitoring for the COVID-19 pandemic, Telemed. e-Health 26 (2020) 1110–1112.
[https://doi.org/10.1089/tmj.2020.0134]

-
X. Liu, Y. Zhang, Z. Yu, H. Lu, H. Yue, J. Yang, rppg-mae: Self-supervised pretraining with masked autoencoders for remote physiological measurements, IEEE Trans. Multimed. 26 (2024) 7278–7293.
[https://doi.org/10.1109/TMM.2024.3363660]

-
J. Zhang, H. Sun, Y. Hu, G. Zhu, F. Liu, B. Yan, et al., Channel attention pyramid network for remote physiological measurement, Sci. Rep. 15 (2025) 22495.
[https://doi.org/10.1038/s41598-025-06107-5]

-
B. Li, P. Zhang, J. Peng, H. Fu, Non-contact PPG signal and heart rate estimation with multi-hierarchical convolutional network, Pattern Recognit. 139 (2023) 109421.
[https://doi.org/10.1016/j.patcog.2023.109421]

-
W. Gao, S. Emaminejad, H.Y.Y. Nyein, S. Challa, K. Chen, A. Peck, et al., Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis, Nature 529 (2016) 509–514.
[https://doi.org/10.1038/nature16521]

-
W. He, C. Wang, H. Wang, M. Jian, W. Lu, X. Liang, et al., Integrated textile sensor patch for real-time and multiplex sweat analysis, Sci. Adv. 5 (2019) eaax0649.
[https://doi.org/10.1126/sciadv.aax0649]

-
Y. Zhao, B. Wang, H. Hojaiji, Z. Wang, S. Lin, C. Yeung, et al., A wearable freestanding electrochemical sensing system, Sci. Adv. 6 (2020) eaaz0007.
[https://doi.org/10.1126/sciadv.aaz0007]

-
H. Cao, R. Lin, Z. Long, L. Xing, X. Xue, A self-powered wireless sweat-analysis patch for real-time monitoring physiological status, Nano Energy 123 (2024) 109411.
[https://doi.org/10.1016/j.nanoen.2024.109411]

-
A.J. Bandodkar, P. Gutruf, J. Choi, K.H. Lee, Y. Sekine, J.T. Reeder, et al., Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat, Sci. Adv. 5 (2019) eaav3294.
[https://doi.org/10.1126/sciadv.aav3294]

-
Y. Wu, Q. Wang, X. Li, K. Li, D. Huang, K. Zou, et al., Olfactory-Inspired Separation-Sensing Nanochannel-Based Electronics for Wireless Sweat Monitoring, ACS Nano 19 (2025) 3781–3790.
[https://doi.org/10.1021/acsnano.4c14660]

-
J. Tu, J. Min, Y. Song, C. Xu, J. Li, J. Moore, et al., A wireless patch for the monitoring of C-reactive protein in sweat, Nat. Biomed. Eng. 7 (2023) 1293–1306.
[https://doi.org/10.1038/s41551-023-01059-5]

-
K. Kwon, J.U. Kim, Y. Deng, S.R. Krishnan, J. Choi, H. Jang, et al., An on-skin platform for wireless monitoring of flow rate, cumulative loss and temperature of sweat in real time, Nat. Electron. 4 (2021) 302–312.
[https://doi.org/10.1038/s41928-021-00556-2]

-
H. Choi, S.W. Lee, Metal Oxide-based Electrochemical Non-enzymatic Glucose Biosensors: A Mini-Review, J. Sens. Sci. Technol. 34 (2025) 105–115.
[https://doi.org/10.46670/JSST.2025.34.2.105]

-
Z. Lv, J. Wang, G. Li, An Antenna Loaded with Complementary Split Ring Resonator for Non-Invasive Blood Glucose Measurement. Proceedings of the 2024 IEEE International Instrumentation and Measurement Technology Conference (I2MTC), Glasgow, United Kingdom, 2024, pp. 1–6.
[https://doi.org/10.1109/I2MTC60896.2024.10561044]

-
Z. Hamouda, J.L. Wojkiewicz, A.A. Pud, L. Kone, S. Bergheul, T. Lasri, Magnetodielectric nanocomposite polymer-based dual-band flexible antenna for wearable applications, IEEE Trans. Antennas Propag. 66 (2018) 3271–3277.
[https://doi.org/10.1109/TAP.2018.2826573]

-
Y. Chen, S. Lu, S. Zhang, Y. Li, Z. Qu, Y. Chen, et al., Skin-like biosensor system via electrochemical channels for noninvasive blood glucose monitoring, Sci. Adv. 3 (2017) e1701629.
[https://doi.org/10.1126/sciadv.1701629]

-
L. Lipani, B.G.R. Dupont, F. Doungmene, F. Marken, R.M. Tyrrell, R.H. Guy, et al., Non-invasive, transdermal, path-selective and specific glucose monitoring via a graphene-based platform, Nat. Nanotechnol. 13 (2018) 504–511.
[https://doi.org/10.1038/s41565-018-0112-4]

-
Z. Pu, X. Zhang, H. Yu, J. Tu, H. Chen, Y. Liu, et al., A thermal activated and differential self-calibrated flexible epidermal biomicrofluidic device for wearable accurate blood glucose monitoring, Sci. Adv. 7 (2021) eabd0199.
[https://doi.org/10.1126/sciadv.abd0199]

-
J. Yang, S. Zheng, D. Ma, T. Zhang, X. Huang, S. Huang, et al., Masticatory system–inspired microneedle theranostic platform for intelligent and precise diabetic management, Sci. Adv. 8 (2022) eabo6900.
[https://doi.org/10.1126/sciadv.abo6900]

-
X. Xiao, Q. Li, A noninvasive measurement of blood glucose concentration by UWB microwave spectrum, IEEE Antennas Wirel. Propag. Lett. 16 (2016) 1040–1043.
[https://doi.org/10.1109/LAWP.2016.2618946]

-
J. Hanna, Y. Tawk, S. Azar, A.H. Ramadan, B. Dia, E. Shamieh, et al., Wearable flexible body matched electromagnetic sensors for personalized non-invasive glucose monitoring, Sci. Rep. 12 (2022) 14885.
[https://doi.org/10.1038/s41598-022-19251-z]

-
A.M. Sabatini, Estimating three-dimensional orientation of human body parts by inertial/magnetic sensing, Sensors 11 (2011) 1489–1525.
[https://doi.org/10.3390/s110201489]

-
P. Picerno, 25 years of lower limb joint kinematics by using inertial and magnetic sensors: A review of methodological approaches, Gait Posture 51 (2017) 239–246.
[https://doi.org/10.1016/j.gaitpost.2016.11.008]

-
M. Airaksinen, A. Gallen, A. Kivi, P. Vijayakrishnan, T. Häyrinen, E. Ilén, et al., Intelligent wearable allows out-of-the-lab tracking of developing motor abilities in infants, Commun. Med. 2 (2022) 69.
[https://doi.org/10.1038/s43856-022-00131-6]

-
C. Voisard, N. De l'Escalopier, D. Ricard, L. Oudre, Automatic gait events detection with inertial measurement units: healthy subjects and moderate to severe impaired patients, J. Neuroeng. Rehabil. 21 (2024) 104.
[https://doi.org/10.1186/s12984-024-01405-x]

-
L.M. Baker, A. Yawar, D.E. Lieberman, C.J. Walsh, Predicting overstriding with wearable IMUs during treadmill and overground running, Sci. Adv. 14 (2024) 6347.
[https://doi.org/10.1038/s41598-024-56888-4]

-
C. Holloway, W. Bhot, K.X.X. Yong, I. McCarthy, T. Suzuki, A. Carton, et al., STEP-UP: Enabling low-cost IMU sensors to predict the type of dementia during everyday stair climbing, Front. Comput. Sci. 3 (2022) 804917.
[https://doi.org/10.3389/fcomp.2021.804917]

-
C.K. Tseng, S.J. Huang, L.J. Kau, Wearable Fall Detection System with Real-Time Localization and Notification Capabilities, Sensors 25 (2025) 3632.
[https://doi.org/10.3390/s25123632]

- K. Kim, Paddling Posture Correction System Using IMU Sensors, J. Sens. Sci. Technol. 27 (2018) 86–92.
-
T. Unger, R. de Sousa Ribeiro, M. Mokni, T. Weikert, J. Pohl, A. Schwarz, et al., Upper limb movement quality measures: comparing IMUs and optical motion capture in stroke patients performing a drinking task, Front. Digit. Health 6 (2024) 1359776.
[https://doi.org/10.3389/fdgth.2024.1359776]

-
G. Di Lazzaro, M. Ricci, M. Al-Wardat, T. Schirinzi, S. Scalise, F. Giannini, et al., Technology-based objective measures detect subclinical axial signs in untreated, de novo Parkinson’s disease, J. Parkinsons Dis. 10 (2020) 113–122.
[https://doi.org/10.3233/JPD-191758]

-
D. Rai, H.K. Thakkar, S.S. Rajput, J. Santamaria, C. Bhatt, F. Roca, A comprehensive review on seismocardiogram: current advancements on acquisition, annotation, and applications, Mathematics 9 (2021) 2243.
[https://doi.org/10.3390/math9182243]

-
Z. Zhou, Y. Jin, J. Fu, S. Si, M. Liu, Y. Hu, et al., Smart wireless flexible sensing system for unconstrained monitoring of ballistocardiogram and respiration, npj Flex. Electron. 9 (2025) 15.
[https://doi.org/10.1038/s41528-025-00388-6]

- Y. Fu, K. Sun, R. Wang, X. Li, J. Ren, Y. Zhang, et al., Enabling Cardiac Monitoring using In-ear Ballistocardiogram on COTS Wireless Earbuds, arXiv., arXiv:2501.06744, (2025).
-
R.A. Iacobescu, B.D. Florescu, A.M. Dimache, L.E. Corneanu, E.A. Coman, Pearls and pitfalls of cuffless blood pressure monitoring devices, Rom. Med. J. 70 (2023) 118–122.
[https://doi.org/10.37897/RMJ.2023.3.2]

-
J.-R. Hu, G. Martin, S. Iyengar, L.C. Kovell, T.B. Plante, N. van Helmond, et al., Validating cuffless continuous blood pressure monitoring devices, Cardiovasc. Digit. Health J. 4 (2023) 9–20.
[https://doi.org/10.1016/j.cvdhj.2023.01.001]

-
K.H. Lee, X. Ni, J.Y. Lee, H. Arafa, D.J. Pe, S. Xu, et al., Mechano-acoustic sensing of physiological processes and body motions via a soft wireless device placed at the suprasternal notch, Nat. Biomed. Eng. 4 (2020) 148–158.
[https://doi.org/10.1038/s41551-019-0480-6]

-
M. Guess,I. Soltis, B. Rigo, N. Zavanelli, S. Kapasi, H. Kim, et al., Wireless batteryless soft sensors for ambulatory cardiovascular health monitoring, Soft Sci. 3 (2023) 24.
[https://doi.org/10.20517/ss.2023.17]

-
G. N, C.R. Bhat, M. T.R, T.E. Yimer, Enhancing visual seismocardiography in noisy environments with adaptive bidirectional filtering for Cardiac Health Monitoring, BMC Med. Inform. Decis. Mak. 24 (2024) 1–14.
[https://doi.org/10.1186/s12911-024-02690-1]

-
Y. Zhao, Q. Sun, S. Mei, L. Gao, X. Zhang, Z. Yang, et al., Wearable multichannel-active pressurized pulse sensing platform, Microsyst. Nanoeng. 10 (2024) 77.
[https://doi.org/10.1038/s41378-024-00703-7]

-
J. Li, H. Jia, J. Zhou, X. Huang, L. Xu, S. Jia, et al., Thin, soft, wearable system for continuous wireless monitoring of artery blood pressure, Nat. Commun. 14 (2023) 5009.
[https://doi.org/10.1038/s41467-023-40763-3]

-
S. Zhou, G. Park, K. Longardner, M. Lin, B. Qi, X. Yang, et al., Clinical validation of a wearable ultrasound sensor of blood pressure, Nat. Biomed. Eng. 9 (2025) 865–881.
[https://doi.org/10.1038/s41551-024-01279-3]


Eung Soo Lim is currently an undergraduate student in the Division of Environmental and Ecological Engineering at Korea University with a double major in Medical Convergence Engineering. As an undergraduate researcher in Professor Ha Uk Chung’s laboratory, his research interests include wireless physiological monitoring and wearable sensor technologies.

So Hyun Lee is an undergraduate student at the School of Biomedical Engineering at Korea University, pursuing a double major in Medical Convergence Engineering. She is conducting undergraduate research under the supervision of Professor Ha Uk Chung. Her research interests include wireless physiological-signal monitoring and wearable biosensors.

Jieun Choi is an undergraduate student at the School of Biomedical Engineering at Korea University, where she will enroll in 2023. She is also pursuing a double major in Materials Science and Engineering. As an undergraduate researcher at Professor Ha Uk Chung’s laboratory, her research interests include wireless physiological monitoring and wearable biosensor technology.

Yeon Jun Lim is currently an undergraduate student at the School of Biomedical Engineering at Korea University, with a double major in Electrical Engineering. Since the summer of 2025, he has been working as an undergraduate researcher at the WISE Medicine Laboratory under the supervision of Professor Ha Uk Chung. His research interests include biosensors and artificial neural networks.

Ha Uk Chung is an Assistant Professor of Biomedical Engineering at Korea University and a co-founder of Sibel Health, a medical device company with FDA-cleared wireless platforms. His research aims to transform healthcare through innovative bio-integrated wireless electronic systems by integrating advances in biomedical instrumentation, wireless technology, flexible mechanics, and artificial intelligence. Drawing on his experience of successfully launching multiple FDA-cleared Class II medical products, he focused on developing solutions that touch patients by addressing important clinical needs. His work spans the entire continuum of care, benefiting diverse patient populations—including fetuses, neonates, pregnant women, patients with cancer, and older adults with neurodegenerative disorders—and translating these innovations into commercialized products.
