
Study on the Enhancement of Dry-Etching Characteristics of z-cut LiNbO3 by Proton Exchange
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Dry etching of lithium niobate (LiNbO3), a key material for integrated optics, is hindered by nonvolatile lithium fluoride (LiF) byproducts that degrade pattern quality. This study investigates the effect of proton exchange (PE) pretreatment and mask type (PR/Cr vs. Cr-only) on the CF4/He plasma etching of z-cut LiNbO3. PE pretreatment was found to double the etch rate while dramatically suppressing the sidewall redeposition and “fencing” to produce clean, near-vertical profiles. This improvement is attributed to hydrogen ions (H⁺) from the PE process acting as scavengers reacting with fluorine radicals to form volatile hydrogen fluoride, thereby inhibiting LiF formation, which was confirmed by the absence of fluorine residues on PE-treated surfaces. While a Cr-only mask yielded the highest profile quality, a PR/Cr composite mask provided the maximum etch rate. These findings establish PE pretreatment as a highly effective method for enhancing the efficiency and quality of LiNbO3 etching, offering a foundational technology for fabricating high-performance optical devices.
Keywords:
Lithium niobate, Proton exchange, Plasma etching, Waveguide, Integrated optics1. INTRODUCTION
The rapid advancement of modern information society demands innovative optical technologies in diverse fields such as optical communications, quantum computing, and high-precision sensing [1]. At the core of these technologies is the integrated optical circuit (IOC), which precisely controls and integrates light. The choice of platform material for implementing IOCs is a critical issue that directly affects device performance. Among various optical materials, lithium niobate (LiNbO3) has held a unique position as the ‘‘workhorse material’’ in the field of optics for several decades [2].
LiNbO3 is a ferroelectric crystal with a perovskite-like structure that possesses unparalleled physical properties [3]. Its strong electro-optic effect allows linear modulation of the refractive index through an external electric field, forming the basis for ultra-high-speed optical modulators and switches [4]. In addition, its excellent nonlinear optical properties are essential for wavelength converters that alter the wavelengths of lasers and generate quantum-entangled photon sources. Its piezoelectric and photoelastic properties are widely applied in communication and sensor fields such as surface acoustic wave filters and acousto-optic devices [5]. Among the various crystal orientations of LiNbO3, the z-cut substrate exhibits the highest electro-optic coefficient (r33), which enables efficient phase modulation with a low driving voltage, making it the most widely used material for fabricating high-performance optical waveguide devices [6]. Owing to these unique advantages, the importance of LiNbO3 as a core material for next-generation integrated optical platforms is increasing.
Forming an optical waveguide that confines and guides light on a LiNbO3 substrate is a fundamental step in fabricating IOCs. One of the representative techniques is the proton exchange (PE) process, in which a LiNbO3 substrate is immersed in a proton source, such as benzoic acid, to exchange lithium ions (Li⁺) in the crystal with hydrogen ions (H⁺, or protons) [7]. This process forms a region with a higher refractive index on the substrate surface, which functions as a waveguide. However, the PE process has inherent limitations. The penetration of an excessive amount of hydrogen ions into the LiNbO3 crystal lattice forms various crystalline phases of HxLi1-xNbO3, leading to severe lattice distortion and defects [8]. This degradation of crystallinity can significantly weaken or even eliminate the intrinsic electro-optic properties of LiNbO3 [9]. Furthermore, the region with a high concentration of substituted hydrogen exists in a thermodynamically unstable metastable phase that causes the refractive-index profile to change with temperature variations or over time, thereby undermining the long-term reliability of the device [10]. To mitigate these problems, an annealing process is typically required after the PE process. Annealing diffuses the hydrogen ions concentrated on the surface deeper into the substrate, lowering the surface concentration, and restoring the distorted crystal lattice (lattice recovery) to recover the degraded electrooptic properties [11]. This facilitates the transition from an unstable phase to a more stable α-phase, improving the overall stability of the waveguide.
Although the annealing process significantly improves the characteristics of PE:LiNbO3 waveguides, it has limitations in the complete removal or control of the high-concentration hydrogen-exchanged layer at the top surface of the waveguide. This microscopic residual layer can be a source of optical scattering loss or a factor that impairs long-term stability [12]. In this paper, we propose a dry-etching process as a post-treatment technology for PE:LiNbO3 waveguides. The core idea was to precisely remove the surface layer formed by the PE and annealing processes at the nanometer scale to physically eliminate the source of potential defects to reduce optical loss and maximize device stability.
However, LiNbO3 is known to be a very difficult material to etch owing to its chemical stability and mechanical hardness. In particular, significant problems arise in reactive ion etching processes using fluorine (F)-based gases (e.g., CF4 and SF6) [13]. When fluorine radicals in the plasma react with LiNbO3, the niobium compound NbF5 becomes volatile and easily removed. In contrast, the lithium compound LiF is a nonvolatile byproduct with a very high boiling point. This LiF redeposits on the etched surface, acting as a micromask, or adheres to the sidewalls of the etched pattern (a phenomenon known as “fencing”), which reduces the etch rate and results in a very rough surface and nonvertical sidewalls [14].
This study proposes a critical hypothesis to address this challenge: the PE process can positively influence the dry-etching characteristics of LiNbO3. It is expected that the abundant hydrogen ions (H+) on the substrate surface owing to the PE process can preferentially react with the fluorine radicals in the plasma to form volatile hydrogen fluoride (HF) gas. This reaction is anticipated to act as a “scavenger” that chemically suppresses the formation of nonvolatile LiF to mitigate the redeposition problem and enable cleaner and more efficient etching. To verify this hypothesis and explore optimal process conditions, this study was designed as a comparative experiment with four different conditions by varying the mask composition (photoresist (PR)/Cr composite mask vs. Cr-only mask) and PE treatment of the substrate. Furthermore, as an engineering approach to maximize the LiF removal effect, a time-multiplexed plasma etching method—which periodically alternates between the etching gas, CF4, and physically sputtering gas He—was introduced to evaluate its effectiveness.
2. EXPERIMENTAL
To systematically analyze the effects of the PE process and mask type on the dry-etching characteristics of z-cut LiNbO3 (Gooch & Housego) substrates, experiments were designed based on commercial 3 in. z-cut LiNbO3 single-crystal wafers (500 μm thick). All samples were prepared by ultrasonic cleaning in acetone, isopropyl alcohol, and deionized water, respectively, to remove organic contaminants from the surface, followed by drying with nitrogen gas.
The samples were broadly divided into two groups: those that underwent PE and those that did not. For the PE process group, a hydrogen-exchanged layer was formed on the surface by immersing the substrates in a benzoic acid melt containing 0.8 mol% of lithium benzoate (LB, Merck) at 300oC for 3 h. The addition of LB serves to control the proton activity in the melt, inducing the formation of a stable α-phase waveguide. After the PE process, a post-annealing treatment was performed in an atmospheric oven at 400oC for 2 h to repair the crystal lattice damaged during the process and optimize the concentration profile of the exchanged hydrogen ions.
Next, a mask pattern for dry etching was formed. To compare the effects of mask materials on the etching characteristics, two types of masks were prepared: a PR/Cr double-layer mask and Cr-only mask, as shown in Fig. 1.
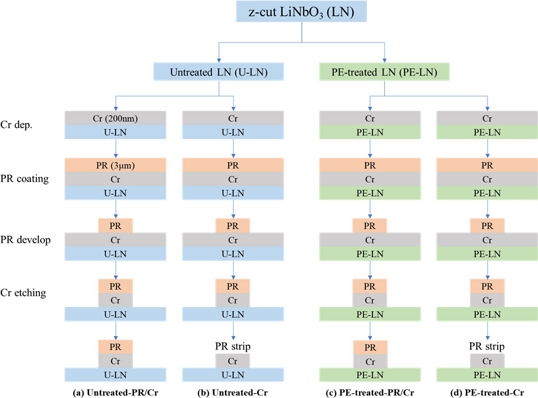
Flowchart illustrating the fabrication steps for U-LN and PE-LN samples with PR/Cr and Cr-only masks. (a) fabrication of a PR/Cr mask on U-LN, (b) fabrication of a C-only mask on U-LN, (c) fabrication of a Pr/Cr mask on PE-LN, and (d) fabrication of a Cr-only mask on PE-LN.
First, a Cr thin film with a thickness of approximately 200 nm was uniformly deposited on all the samples using a DC sputtering system (Sunic system, IS 3000). A PR (AZ GXR601) layer of approximately 3 μm thick was then spin-coated on top, and a pattern was defined using a contact aligner (SUSS MicroTec Lithograph GmbH, MA8) with an exposure energy of 240 mJ/cm2 and subsequently developed. This PR pattern was used as an etching mask to etch the underlying Cr film with an inductively coupled plasma (ICP, Oxford Instruments, PlasmaLab System 133) etching system to form the PR/Cr double-layer mask. To analyze the effect of the Cr-only mask, the top PR layer was completely removed from some samples using a PR stripper. Through this process, four sample conditions were ultimately prepared: untreated-PR/Cr, untreated-Cr, PE-treated-PR/Cr, and PE-treated-Cr.
The dry etching of LiNbO3 for all four conditions was performed using the same ICP etching equipment at a substrate temperature of 20oC. A time-multiplexed etching technique—which periodically alternates between chemical etching and physical surface cleaning—was employed to effectively suppress the redeposition of nonvolatile byproducts such as LiF. This technique consisted of a cycle of a chemical etching step with CF4 gas (5 s) and a physical sputtering/surface cleaning step with He gas (10 s), repeated for a total of 45 cycles.
Finally, after all etching processes were completed, the etch depth, cross-sectional profile, sidewall verticality and roughness, and the presence of byproduct residues were comprehensively evaluated using field-emission scanning electron microscopy (FE-SEM, FEI Company, Quanta 200 FEG). This allowed the qualitative and quantitative analysis of the effects of the two key variables on the dry-etching results of LiNbO3: the PE process and mask type.
3. RESULTS AND DISCUSSIONS
This study aimed to elucidate the effects of PE pretreatment and type of etch mask (PR/Cr composite or Cr-only) on the etch rate and profile quality during the dry etching of z-cut LiNbO3 substrates. After etching the four different samples (a–d) under identical time-multiplexed CF4/He plasma conditions, the cross-sectional profiles and depths were analyzed using FE-SEM. Fig. 2 shows representative cross-sectional SEM images for each condition. Table 1 quantitatively summarizes the measured etch depths and calculated etch rates.

Cross-sectional SEM images for the four sample conditions: (a) untreated-PR/Cr, (b) untreated-Cr, (c) PE-treated-PR/Cr, and (d) PE-treated-Cr.

Summary of etch depth and etch rate for the four sample conditions: (a) untreated-PR/Cr, (b) untreated-Cr, (c) PE-treated-PR/Cr, and (d) PE-treated-Cr.
The results show that the PE pretreatment dramatically enhanced the etch rate of LiNbO3. As listed in Table 1, the etch rates of the untreated samples (a) and (b) were 0.25 nm/s and 0.22 nm/s, respectively. In contrast, the PE-treated samples (c) and (d) showed a significant increase in the etch rate of 0.47 nm/s and 0.43 nm/s, respectively, representing an improvement of approximately 1.9–2.1 times. This phenomenon can be directly attributed to the formation of nonvolatile byproducts—a persistent problem in LiNbO3 dry etching. In a typical CF4 plasma environment, the lithium (Li) component of LiNbO3 reacts with fluorine (F) radicals to form LiF, which has a very high boiling point. This LiF redeposits on the etching surface, acting as a micromask and physically hindering subsequent etching reactions, which is the primary cause of reduced etching rates. However, when Li+ ions on the substrate surface are replaced by H+ ions through the PE process, a new chemical reaction pathway opens up in which fluorine radicals in the plasma preferentially react with H+ instead of Li+ to form volatile HF gas. In other words, the introduced hydrogen acted as a scavenger, suppressing the formation of nonvolatile LiF to maintain a clean etching surface and promoting continuous etching reactions.
This difference in chemical mechanism is qualitatively observed in the SEM images shown in Fig. 2. For the untreated samples (a) and (b), a significant amount of redeposited material was observed across the etched surface and sidewalls. In particular, a distinct ‘‘fencing’’ phenomenon, resembling grass or whiskers, is evident on the sidewalls of sample (a). This showed that the LiF byproducts generated during the etching process were not effectively removed and had accumulated. In contrast, for PE-treated samples (c) and (d), such redeposited materials or fencing phenomena were almost nonexistent, and very clean and steep profiles were formed. This suggests that the PE process not only increases the etch rate but also plays a crucial role in determining the quality and precision of the etch profile.
Secondly, the mask composition had a significant effect on the etching results. Regardless of whether PE treatment was applied, the samples using the PR/Cr composite mask (a, c) showed a greater etch depth than those using the Cr-only mask (b, d). This is presumably because the carbon (C) components from the PR, which decompose during the etching process, contribute to additional chemical reactions within the plasma. For example, carbon components might react with nonvolatile byproducts, such as LiF, to form more volatile compounds or adjust the F/C ratio in the plasma, positively influencing the etching characteristics. However, this advantage in etch depth has a drawback. Looking at the top of the images for Figs. 2 (a) and (c), the PR/Cr mask was partially etched by the plasma, resulting in a rough surface. In contrast, sample (d), which used a Cr-only mask, had a slightly lower etch depth than (c) but achieved the best overall profile quality with an undamaged, very clean top surface and vertical sidewalls.
To support the physical and morphological observations of the enhanced etch rate and improved profile quality presented in the previous section, and to quantitatively prove the underlying chemical mechanism, SEM and energy-dispersive X-ray spectroscopy (EDS) analysis was performed. Elemental compositions were measured on the etched bottom surface of each sample after the process, and the results are summarized in Table 2. A significant fluorine signal was detected in untreated samples (a) and (b). Specifically, the etched surface of sample (a) contained 9.2% fluorine, while sample (b) contained a much higher concentration of 20.9%. As this fluorine can only exist in the form of nonvolatile LiF, the quantitative data directly and numerically proves the hypothesis that LiF byproducts accumulate on the etched surface during LiNbO3 dry etching. The high fluorine residue acts as a micromask, physically hindering subsequent etching reactions and serving as a direct cause of the low etching rate and rough surface profile. The fact that sample (b) has a higher fluorine residue than sample (a) supports the observation that the etch rate (0.22 nm/s) of sample (b) was slightly lower than that of sample (a) (0.25 nm/s). A crucial finding was the difference in fluorine residue content between the two untreated samples. Sample (a) had a significantly lower fluorine content compared with (b) (9.2% vs. 20.9%); the only difference between the two samples is the mask composition (PR/Cr vs. Cr-only). The high carbon (C) content (61.7%) in the EDS data of sample (a) indicated that the PR mask was decomposing during the etching process. It is highly probable that these carbon radicals reacted with fluorine radicals in the plasma to form volatile CFx species to act as a secondary scavenger that consumes some of the fluorine radicals. Owing to this secondary scavenger effect, the formation of nonvolatile LiF in sample (a) was partially suppressed, resulting in a higher etching rate than that in sample (b). This provides evidence that the carbon component of the PR mask is not merely a cause of mask damage, but can actively participate in the etching chemistry to have a positive effect.

Compositional distribution of the etched surfaces for four sample conditions: (a) untreated-PR/Cr, (b) untreated-Cr, (c) PE-treated-PR/Cr, and (d) PE-treated-Cr.
The most notable result is that the EDS analysis of the PE-treated samples (c) and (d) showed no fluorine signal, indicating that fluorine was not detected. This quantitatively proves that the scavenger role of hydrogen ions (H+) proposed in this study is not just a hypothesis but a highly effective mechanism for completely suppressing the formation of nonvolatile fluorine residues. By preferentially reacting with fluorine radicals in the plasma, the H+ ions exchanged through the PE process were removed as volatile HF gas so no LiF accumulated on the etched surface. This complete removal of fluorine directly explains all the observed results, including the approximately two-fold increase in the etch rate and formation of a clean, vertical sidewall profile. The clean surface composition of samples (c) and (d) in Table 2 shows that no obstacles causing etch-rate degradation were present. Notably, sample (d) without fluorine residues had a very high O K content of 86.2% and a relatively low Nb L content of 13.8%. This suggests that the PE treatment contributes to a more efficient and cleaner removal of Li and Nb from the LiNbO3 crystal lattice. The etched surface maintains the clean state of the bulk material, which is primarily composed of oxygen.
In conclusion, these experimental results demonstrate that PE pretreatment is a highly effective method for maximizing both the efficiency and quality of LiNbO3 dry etching. By chemically suppressing the formation of nonvolatile LiF byproducts through hydrogen exchange, an approximately two-fold increase in the etch rate and a clean sidewall profile were achieved simultaneously. Furthermore, a tradeoff between the etch rate and profile quality was identified depending on the mask type. The PR/Cr composite mask provided the highest etch rate but at the cost of mask surface damage, while the Cr-only mask was advantageous for forming the most precise and clean patterns but with a slightly lower etch rate. Therefore, the strategic selection of the PE treatment process and mask type, based on the target device structure and required precision, will be a key process variable for fabricating high-quality LiNbO3-based optical devices.
4. CONCLUSIONS
This study investigated a PE pretreatment process to address the persistent issue of nonvolatile LiF byproduct formation during the dry etching of z-cut LiNbO3 substrates. The PE pretreatment proved highly effective, nearly doubling the etch rate and achieving a high-quality, clean, and near-vertical etch profile by suppressing the sidewall redeposition and “fencing” phenomenon. The mechanism underlying this enhancement was elucidated by EDS compositional analysis. It confirmed that hydrogen ions introduced via the PE process acted as powerful scavengers, preferentially reacting with fluorine radicals in the plasma. This chemical pathway suppressed the formation of nonvolatile LiF, as verified by the absence of fluorine residues on etched surfaces of PE-treated samples.
A tradeoff between the etch rate and profile quality was identified, depending on the mask composition. A PR/Cr composite mask yielded the highest etch rate but resulted in mask surface damage, while a Cr-only mask with superior plasma resistance was advantageous for fabricating the most precise and clean patterns. In conclusion, this study demonstrates that the strategic combination of PE pretreatment and mask selection is a critical parameter for the optimal process design of high-quality and high-efficiency LiNbO3-based integrated optical circuits.
Acknowledgments
This study has been conducted with the support of the Korea Institute of Industrial Technology as “Development of a single-mobility for transportation vulnerables (KITECH UR-25-0103)”.
References
-
Y. Hu, D. Zhu, S. Lu, X. Zhu, Y. Song, D. Renaud, et al., Integrated electro-optics on thin-film lithium niobate, Nat. Rev. Phys. 7 (2025) 237–254.
[https://doi.org/10.1038/s42254-025-00825-5]

-
N. Montaut, A. George, M. Monika, F. Nosrati, H. Yu, S. Sciara, et al., Progress in integrated and fiber optics for time-bin based quantum information processing, Adv. Opt. Technol. 14 (2025) 1560084.
[https://doi.org/10.3389/aot.2025.1560084]

-
Y. Wang, Y. Xu, B. Zhang, Y. Dong, Y. Ni, High-performance thin-film lithium niobate electro-optic modulator based on etching slot and ultrathin silicon film, Appl. Opt. 62 (2023) 1858–1864.
[https://doi.org/10.1364/AO.482202]

-
C. Wang, M. Zhang, X. Chen, M. Bertrand, A. Shams-Ansari, S. Chandrasekhar, et al., Integrated lithium niobate electro-optic modulators operating at CMOS-compatible voltages, Nature 562 (2018) 101–104.
[https://doi.org/10.1038/s41586-018-0551-y]

-
Y. Yang, L. Gao, S. Gong, Surface-acoustic-wave devices based on lithium niobate and amorphous silicon thin films on a silicon substrate, IEEE Trans. Microw. Theory Tech. 70 (2022) 5185–5194.
[https://doi.org/10.1109/TMTT.2022.3194554]

-
N. Courjal, M.P. Bernal, G. Ulliac, J. Dahdah, S. Benchabane, J-M. Merolla, LiNbO3 acousto-optical and electro-optical micromodulators, J. Eur. Opt. Soc.-Rapid Publ. 4 (2009) 09018.
[https://doi.org/10.2971/jeos.2009.09018]

-
C. Canali, A. Carnera, G. Della Mea, P. Mazzoldi, S.M. Al Shukri, A.C.G. Nutt, et al., Structural characterization of proton exchanged LiNbO3 optical waveguides, J. Appl. Phys. 59 (1986) 2643–2649.
[https://doi.org/10.1063/1.336968]

-
Y.N. Korkishko, V.A. Fedorov, O.Y. Feoktistova, LiNbO3 optical waveguide fabrication by high-temperature proton exchange, J. Lightwave Technol. 18 (2000) 562–570.
[https://doi.org/10.1109/50.838131]

-
L. Wan, Z. Yang, W. Zhou, M. Wen, T. Feng, S. Zeng, et al., Highly efficient acousto-optic modulation using nonsuspended thin-film lithium niobate-chalcogenide hybrid waveguides, Light Sci. Appl. 11 (2022) 145.
[https://doi.org/10.1038/s41377-022-00840-6]

-
A. Sosunov, R. Ponomarev, O. Semenova, I. Petukhov, A. Volyntsev, Effect of pre-annealing of lithium niobate on the structure and optical characteristics of proton-exchanged waveguides, Opt. Mater. 88 (2019) 176–180.
[https://doi.org/10.1016/j.optmat.2018.11.018]

-
F. Lenzini, S. Kasture, B. Haylock, M. Lobino, Anisotropic model for the fabrication of annealed and reverse proton exchanged waveguides in congruent lithium niobate, Opt. Express 23 (2015) 1748–1756.
[https://doi.org/10.1364/OE.23.001748]

-
L. Cai, Y. Wang, H. Hu, Low-loss waveguides in a single-crystal lithium niobate thin film, Opt. Lett. 40 (2015) 3013–3016.
[https://doi.org/10.1364/OL.40.003013]

-
A.A. Osipov, S.E. Alexandrov, G.A. Iankevich, The effect of a lithium niobate heating on the etching rate in SF6 ICP plasma, Mater. Res. Express 6 (2019) 046306.
[https://doi.org/10.1088/2053-1591/aafa9d]

-
G. Ulliac, V. Calero, A. Ndao, F.I. Baida, M.P. Bernal, Argon plasma inductively coupled plasma reactive ion etching study for smooth sidewall thin film lithium niobate waveguide application, Opt. Mater. 53 (2016) 1–5.
[https://doi.org/10.1016/j.optmat.2015.12.040]

