A Multilayered Paper-Based Biosensor with Rapid Analysis and Low Power Consumption for Detecting Drought Stress
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
This study presents a multilayer paper-based biosensor with low power consumption capable of rapidly detecting drought stress in plants under field conditions. Proline, a biomarker whose concentration increases during drought, was used to analyze drought stress in plants, and the proline concentration in the sample was determined using a colorimetric method based on the proline-ninhydrin reaction. A multilayer paper-based structure was fabricated using wax printing and origami techniques. To enable rapid sample analysis, the sensing unit was positioned on a waterproof layer, thereby reducing the power required for sensor operation. Additionally, the sensor was fabricated in a sealed configuration to prevent external contamination of the sample during analysis. Proline samples with concentrations ranging from 100 μM to 30 mM were analyzed using the fabricated sensor. The sensor successfully detected proline at concentrations as low as 500 μM, and the ΔRGB value exhibited a linear increase with rising proline concentration. Therefore, we anticipate that the proposed multilayer paper-based biosensor can be effectively applied for plant drought diagnosis in the field and contribute to improving crop productivity.
Keywords:
Plant, Drought stress, Proline, Colorimetric method, Paper-based biosensor1. INTRODUCTION
Plants grow and thrive under a wide range of environmental conditions, including drought and low temperatures. Among these, drought is known to adversely affect plant growth and development by limiting cell division and expansion, thus reducing leaf size, stem elongation, and root proliferation. It is a major abiotic stress that impacts approximately 70% of crops worldwide and decreases crop yields [1-3]. Therefore, it is crucial to detect drought promptly, as it is a key factor that reduces crop production and farm income.
Currently, there are two primary methods for diagnosing drought stress in crops: indirect methods, which evaluate drought through image analysis, and direct methods, which involve collecting crop samples. Indirect methods include remote measurements using infrared thermometry and approaches that employ spectral vegetation indices, such as those in the near-infrared range. Although these approaches enable rapid drought assessment over large areas using satellites and drones, they are costly, require skilled personnel, and often exhibit limited accuracy and precision, which slows the response to drought [4]. A direct method for diagnosing drought involves quantifying proline, a biomarker of drought stress in plant samples [5]. When plants are exposed to environmental stresses such as drought, they activate diverse metabolic and defense mechanisms to survive. Proline accumulates within the plant body to protect proteins from stress-induced damage that could lead to cell death [6,7]. In other words, when plants are affected by drought, they increase proline synthesis to ensure survival, leading to elevated proline concentrations within the plant. Analysis of the proline concentration can therefore be used to determine the degree of drought stress. Currently, methods for detecting proline in plants include high-performance liquid chromatography, isatin paper, and colorimetric analyses [8-12]. Among these, the ninhydrin-based colorimetric method is a simple, reliable, and quantitative approach that is widely employed in many laboratories owing to its simplicity, high measurement reliability, and low cost compared with other techniques. However, this method can only be performed by skilled personnel in a laboratory, as the expensive equipment required makes it impractical for field applications [13,14]. Consequently, there is a growing demand for a drought diagnostic sensor capable of accurately monitoring crop drought stress in the field with high sensitivity and at low cost, while being sufficiently simple for use by untrained personnel to enable rapid response.
To overcome these limitations, Choi et al. proposed two types of ninhydrin-based paper sensors for proline detection that were low-cost, easy to use, and simple to operate, even for unskilled personnel in the field. The first proposed paper sensor featured a two-layer design comprising a sensing layer and a waterproof layer, enabling rapid proline measurement. However, it suffered from low sensitivity and resolution [15]. The second proposed paper sensor, incorporating a four-layer structure with a mixing channel, addressed the sensitivity and resolution issues of the two-layer paper sensor. Nevertheless, it exhibited a threefold increase in reaction temperature and analysis time compared with the conventional method, thereby reducing the sample analysis speed. Therefore, further research is required to develop low-cost, paper-based sensors capable of rapidly analyzing proline concentrations with improved resolution [16].
In this study, we fabricated and characterized a multilayer paper-based biosensor capable of rapid and quantitative proline analysis in plant samples with excellent resolution. The proposed sensor features a four-layer structure that includes a mixing channel. Unlike existing sensors, the sensing unit is integrated into two layers, providing enhanced sensor resolution and enabling fast, low-power quantitative analysis of proline concentration. The proline concentration in the sample is quantified by analyzing the ΔRGB value of the color change resulting from the proline-ninhydrin reaction within the sensing window.
2. EXPERIMENTAL
2.1 Fabrication of multilayer paper-based biosensors
The biosensor designed for measuring proline concentration in a sample features a multilayer structure comprising four layers of stacked paper, as shown in Fig. 1. The sensor consists of a sample loading area (4th or top layer) for sample introduction, a ninhydrin area (3rd and 4th layer) where ninhydrin is deposited at a defined concentration, a mixing channel (2nd layer) in which proline and ninhydrin are combined, a sensing window (2nd layer) for detecting the colorimetric change produced by the reaction between proline and ninhydrin, and a waterproof layer (1st or bottom layer) that prevents sample leakage during the sample analysis process. Fig. 1 (a) illustrates a schematic of the sensor. The sample volume used in the experiment was 80 μL, and the ninhydrin components were prepared on two layers, with equal surface areas and identical components, to ensure sufficient interaction with the sample. The reaction time between proline and ninhydrin must remain constant to ensure precise proline analysis. Accordingly, ninhydrin regions with constant concentrations were fabricated independently.
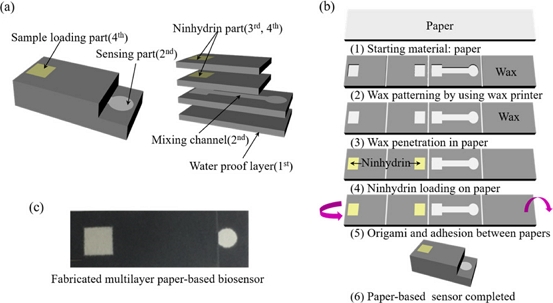
Schematic diagram and fabrication process of a multilayer paper-based biosensor. (a) Schematic of the multilayer paper-based biosensor for high-resolution proline detection. (b) Fabrication process for a multilayer paper-based biosensor. (c) Optical image of the fabricated multilayer paper-based biosensor.
The position of the sensing window in the proposed sensor was modified from four layers (as in previously reported multilayer sensors) to two layers deep, allowing for more rapid heating.
The sensing window operates with lower power consumption compared with previously proposed multilayer structure sensors. This modification reduces both the reaction temperature and response time. Additionally, to protect the sample from evaporative loss and contamination, thereby ensuring precision analysis, all regions except the sample loading area and sensing window were sealed with a wax layer. A wax-coated waterproof layer was also incorporated beneath the mixing channel to minimize sample loss.
Fig. 1 (b) presents the fabrication process using wax printing and origami techniques. The biosensor was printed on Whatman Grade 1 cellulose chromatography paper (GE Healthcare, UK) using a wax printer. The wax-printed paper was then placed on a hotplate heated to 120℃ for 3 min to allow the wax to penetrate the paper thoroughly. Subsequently, 160 μL of ninhydrin was evenly applied to the two designated ninhydrin-containing regions and dried at a low temperature. Finally, the four-layered paper was assembled by double-sided tape, glued, and then folded sequentially to complete the fabrication of a multilayered paper-based biosensor. Fig. 1 (c) presents an optical image of the fabricated multilayer paper-based biosensor.
2.2 Operating principles and analysis of multilayer paper-based biosensors
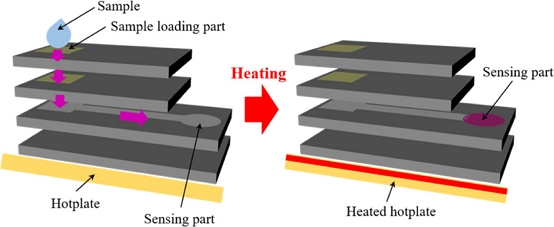
The operating principle of the fabricated multilayer paper-based biosensor is illustrated in Fig. 2. When 80 μL of a proline-containing sample is introduced onto the ninhydrin-coated sample loading area in the fourth (top) layer, the ninhydrin in the sample loading area binds to the proline sample. The proline-ninhydrin mixture descends to the ninhydrin-coated region on the third layer (second from the top) under the influence of gravity, where any unreacted proline completes its reaction with ninhydrin. The proline-ninhydrin mixture continues moving into the mixing channel on the second layer under the influence of gravity, after which it is transported by capillary action along the channel to the sensing window. Finally, the sensor containing the sample is transferred onto a hotplate and heated to 120℃ for 3 min to initiate the reaction. The sensing region develops a purple coloration as a result of the proline-ninhydrin reaction; the higher the proline concentration, the more intense the purple color becomes.
The degree of discoloration observed in the sensing window can be quantitatively evaluated by determining the ΔRGB value of an image captured with a camera in a dark room under constant light brightness. The RGB values of the captured image are quantified using ImageJ software. The ΔRGB value is then calculated as the difference between the average RGB value (Rn, Gn, Bn) obtained in this manner and the RGB value (R0, G0, B0) of the sensing window prior to sample loading (eq. 1) [17].
| (1) |
2.3 Reagents and materials
To quantitatively analyze proline concentration using the multilayer paper-based biosensor, purified L-proline and ninhydrin were obtained from Sigma-Aldrich(St. Louis, USA). The ninhydrin reagent employed in the experiment was prepared by dissolving 1.25% ninhydrin in an 80% glacial acetic acid solution (Duksan Pure Chemicals Co., Ltd, Ansan, Korea). Proline standards of various concentrations were prepared for calibration by completely dissolving L-proline in a 1% sulfosalicylic acid solution (Bio Basic Inc., Canada). Proline concentrations of 0.1, 0.5, 1, 5, 10, 20, and 30 mM were employed in the experiment.
3. RESULTS AND DISCUSSIONS
3.1 Optimization of multilayer paper-based biosensors
To ensure the optimal performance of the multilayer paper-based biosensor, the proline-ninhydrin reaction must be optimized, and the sensor design and fabrication must account for factors that affect this reaction. Key parameters affecting the proline-ninhydrin reaction include reaction temperature and time, the ratio of the sample volume to the ninhydrin volume, and the degree of mixing between the sample and the ninhydrin reagent. Based on results from previously reported paper-based proline sensors, the optimal sample-to-ninhydrin ratio is 1:2 [15,16]. Therefore, in this study, two key factors were investigated to optimize sensor performance: the reaction temperature and time, as well as the length of the mixing channel, which can enhance the degree of mixing between proline and ninhydrin.
Determining the optimal reaction temperature and time is essential for maximizing the efficiency of the proline-ninhydrin reaction. The temperature range for this study was selected based on previously reported references. There are two types of paper-based biosensors: those with a two-layer sensing structure and those with a four-layer sensing structure. The optimal reaction temperature and time for the proline-ninhydrin reaction in a sensor with the sensing window located on the second layer are 3 min at 110℃; when the sensing window is on the fourth layer, the optimal reaction temperature and time are 9 min at 150℃ [15,16]. Based on these findings, and given that the sensing window of the paper-based biosensor proposed in this study is positioned on the second layer, the optimal reaction temperature was expected to be approximately 110℃. Thus, the range of reaction temperatures for optimization was set to 100, 110, and 120℃. The proline concentration of the sample in the experiment was 30 mM, the volume was 80 μL, and a ninhydrin volume of 160 μL was used. The proline-ninhydrin reaction time was measured over 4 min at the three temperatures. Fig. 3 presents the ΔRGB values as a function of reaction temperature and time. As the reaction temperature increased at a given time point, the color of the sensing window became a deeper purple, indicating a corresponding increase in the ΔRGB value. Moreover, after 90 s, the ΔRGB value increased irrespective of the reaction temperature, after which it either plateaued or decreased with extended reaction time. The observed decrease in ΔRGB values after a specific reaction time is attributed to excessive reaction temperatures, which cause deterioration of the sensing window, subsequently affecting the RGB values.
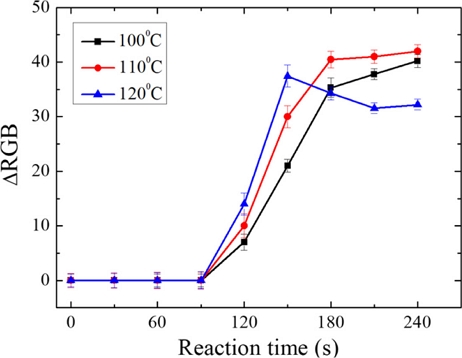
Response of ΔRGB values to reaction temperature and time of the proline-ninhydrin reaction in the proposed multilayer paper-based biosensor.
In our experiment, the ΔRGB value reached its maximum at a reaction time of 3 min and a temperature of 110℃, indicating excellent sensor resolution. Compared with previously reported four-layer paper-based biosensors, the sensor developed and evaluated in this study demonstrated the advantage of detecting proline with minimal power consumption, as the reaction temperature and time were reduced. Based on the experimental results, the optimized reaction temperature and reaction time of the paper-based biosensor fabricated for proline detection were determined to be 110℃ and 3 min, respectively.
In addition to the reaction temperature and time, the extent of mixing between proline and ninhydrin plays a crucial role in determining the sensor’s sensitivity. Existing multilayer paper-based biosensors for proline detection incorporate mixing channels, enabling the sample to flow along the porous paper and ensuring the proper mixing of proline and ninhydrin [16]. The proposed biosensor similarly incorporates a mixing channel that facilitates the mixing of proline and ninhydrin. However, its structural design differs from previously reported sensors, necessitating optimization of the mixing channel length. Therefore, the influence of mixing channel length on sensor sensitivity was systematically analyzed.
To evaluate the effect of the mixing channel length on the sensitivity of the paper sensor, a proline sample with a concentration of 30 mM and a volume of 80 μL was used along with 160 μL of ninhydrin reagent. The reaction temperature and time were set to 110℃ and 3 min, respectively. The length of the mixing channel was varied from 10 to 40 mm in 5 mm increments, and the resulting color change of the sensing window corresponding to each channel length was measured and analyzed. Fig. 4 presents the ΔRGB of a 30 mM proline sample as a function of the mixing channel length. As the mixing channel length increased (up to 30 mm), the ΔRGB value of the sensing window also increased. The ΔRGB value of the sensing window was highest at a channel length of 30 mm, confirming the optimal sensor sensitivity. The ΔRGB value remained constant or decreased slightly when the mixing channel lengths exceeded 30 mm. This phenomenon is likely attributable to insufficient delivery of the mixed proline-ninhydrin solution to the sensing window, despite improved mixing efficiency.
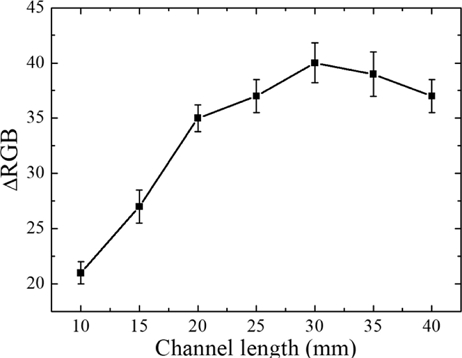
Response of ΔRGB values for a 30 mM proline sample as a function of the mixing channel length in the multilayer paper-based biosensor.
Therefore, based on these experimental results, the mixing channel length of the multilayer paper-based biosensor developed in this study was designed and fabricated to be 30 mm.
3.2 Quantitative analysis of proline using the optimized biosensor
Experiments were conducted to analyze various proline samples quantitatively using the optimized paper-based biosensor. The proline concentrations of the samples tested ranged from 0 to 30 mM, and the corresponding ΔRGB values were determined; the results are presented in Fig. 5. The experimental findings demonstrated that the minimum detectable concentration of proline using the proposed sensor was 500 μM. Furthermore, it was observed that the color of the sensing window progressively changed to a dark purple as the proline concentration increased. At a proline concentration of 500 μM, the ΔRGB value was 15.99, whereas at 30 mM, the ΔRGB value increased to 40.46.
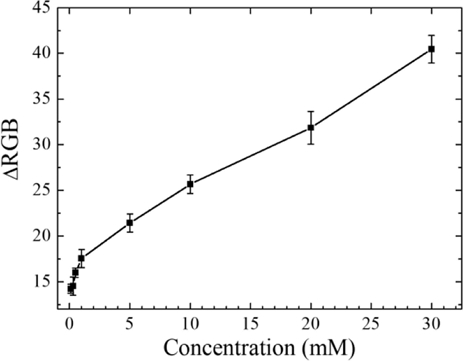
ΔRGB response of the sensing window in the multilayer paper-based biosensor for analyzing various concentrations of proline.
The results indicate that the resolution of the proposed sensor is approximately six times higher than that of the previously reported two-layer paper sensor, which is capable of detecting a minimum proline concentration of 3 mM [15]. Although the sensing window of the proposed biosensor is located on the same layer as that of existing sensors, the superior resolution of the proposed multilayer sensor can be attributed to the effective mixing of proline and ninhydrin within the sample, which is facilitated by the mixing channel integrated within the multilayer structure.
4. CONCLUSIONS
In this study, we developed a multilayer paper-based biosensor capable of quantitatively and rapidly measuring proline with low power consumption, enabling the detection of drought stress in plants under field conditions. The proposed biosensor can be conveniently fabricated using wax printing and origami techniques. To minimize errors caused by external contaminants during sample analysis, only the sample loading area and sensing window are exposed to the environment. Furthermore, to enhance the sensor’s resolution, a mixing channel was incorporated to facilitate efficient mixing between the proline sample and ninhydrin. In addition, a waterproof layer was integrated beneath the mixing channel and sensing window to reduce sample loss.
Using the fabricated multilayer paper-based biosensor, color changes in the sensing window resulting from the proline-ninhydrin reaction were photographed for samples with various proline concentrations, and quantitatively analyzed through ΔRGB analysis. The proposed biosensor successfully detected proline concentrations as low as 500 μM, improving the resolution of the previously proposed two-layer paper sensor by approximately six times. Moreover, as the proline concentration in the samples increased, the color of the sensing window became progressively darker purple, and the corresponding ΔRGB values showed a consistent increase. Therefore, the multilayer paper-based biosensor developed in this study offers a practical approach for diagnosing drought stress directly in the field, thereby enabling improved crop productivity.
Acknowledgments
This study was conducted with research funds from Gwangju University in 2025.
References
-
M. Farooq, A. Wahid, N. Kobayashi, D. Fukita, S.M.A. Basra, Plant drought stress: effects, mechanisms and management, In: E. Lichtfouse, M. Navarrete, P. Debaeke, S. Véronique, C. Alberola (Eds.), Sustainable Agriculture, Springer Nature, Dordrecht, 2009, pp. 153-188.
[https://doi.org/10.1007/978-90-481-2666-8_12]

- H.M. Akram, A. Ali, A. Sattar, H.S.U. Rehman, A. Bibi, Impact of water deficit stress on various physiological and agronomic traits of three basmati rice (Oryza sativa L.) Cultivars, J. Anim. Plant Sci. 23 (2013) 1415-1423.
-
P. Thakur, S. Kumar, J.A. Malik, J.D. Berger, H. Nayyar, Cold stress effects on reproductive development in grain crops: An overview, Environ. Exp. Bot. 67 (2010) 429-443.
[https://doi.org/10.1016/j.envexpbot.2009.09.004]

-
V. Alchanatis, Y. Cohen, S. Cohen, M. Moller, M. Sprinstin, M. Meron, et al., Evaluation of different approaches for estimating and mapping crop water status in cotton with thermal imaging, Precis. Agric. 11 (2010) 27-41.
[https://doi.org/10.1007/s11119-009-9111-7]

-
M. Yamada, H. Morishita, K. Urano, N. Shiozaki, K. Yamaguchi-Shinozaki, K. Shinozaki, et al., Effects of free proline accumulation in petunias under drought stress, J. Exp. Bot. 56 (2005) 1975-1981.
[https://doi.org/10.1093/jxb/eri195]

-
P.B.K. Kishor, P.H. Kumari, M.S.L. Sunita, N. Sreenivasulu, Role of proline in cell wall synthesis and plant development and its implications in plant ontogecy, Front. Plant Sci. 6 (2015) 00544.
[https://doi.org/10.3389/fpls.2015.00544]

-
S. Hayat, Q. Hayat, M.N. Alyemeni, A.S. Wani, J. Pichtel, A. Ahmad, Role of proline under changing environments, Plant Signal. Behav. 7 (2012) 1456-1466.
[https://doi.org/10.4161/psb.21949]

-
E. Abrahám, C. Hourton-Cabassa, L. Erdei, L. Szabados, Methods for determination of proline in plants, In: J.M. Walker (Ed.), Methods in Molecular Biology, Humana Press (Springer Nature), New Jersey, 2010, pp. 317-331.
[https://doi.org/10.1007/978-1-60761-702-0_20]

-
G. Noctor, C.H. Foyer, Simultaneous measurement of foliar glutathione, gamma-glutamylcysteine, and amino acids by high-performance liquid chromatography: comparison with two other assay methods for glutathione, Anal. Biochem. 264 (1998) 98-110.
[https://doi.org/10.1006/abio.1998.2794]

-
I. Smith, Colour reactions on paper chromatograms by a dipping technique, Nature 171 (1953) 43-44.
[https://doi.org/10.1038/171043a0]

-
R.J. Elliott, D.L. Grander, Proline determination with isatin, in the presence of amino acids, Anal. Biochem. 70 (1976) 268-273.
[https://doi.org/10.1016/S0003-2697(76)80068-1]

-
F.N. Boctor, An improved method for colorimetric determination of proline with isatin, Anal. Biochem. 43 (1971) 66-70.
[https://doi.org/10.1016/0003-2697(71)90108-4]

-
F.P. Chinard, Photometric estimation of proline and ornithine, J. Biol. Chem. 199 (1952) 91-95.
[https://doi.org/10.1016/S0021-9258(18)44814-4]

-
A.T. Singh, D. Lantigua, A. Meka, S. Taing, M. Pandher, G. Camci-Unal, Paper-based sensors: emerging themes and applications, Sensors 18 (2018) 2838.
[https://doi.org/10.3390/s18092838]

-
Y.S. Choi, M.R. Lee, C.S. Kim, K.H. Lee, Detection of proline using a novel paper-based analytical device for on-site diagnosis of drought stress in plants, Rev. Sci. Instrum. 90 (2019) 045002.
[https://doi.org/10.1063/1.5055798]

-
Y.S. Choi, M.K. Im, M.R. Lee, C.S. Kim, K.H. Lee, Highly sensitive enclosed multilayer paper-based microfluidic sensor for quantifying proline in plants, Anal. Chim. Acta 1105 (2020) 169-177.
[https://doi.org/10.1016/j.aca.2020.01.038]

-
R.C. Murdock, L. Shen, D.K. Griffin, N. Kelley-Loughnane, I. Papautsky, J.A. Hagen, Optimization of a paper-based ELISA for a human performance biomarker, Anal. Chem. 85 (2013) 11634-11642.
[https://doi.org/10.1021/ac403040a]