
Recent Trends in Conductive Metal–Organic Framework-Based Sensors for Hazardous and Environmental Gas Detection
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Conductive metal–organic frameworks (c-MOFs) have emerged as promising gas-sensing materials due to their intrinsic electrical conductivity at room temperature, high porosity, large surface area, and tunable chemical composition and structure. In particular, the presence of exposed catalytic sites—such as metal nodes and functionalized ligands—enables the binding of airborne chemical species. These interactions, through redox reactions and/or local polarity changes, induce measurable variations in electrical resistance. Common hazardous and environmental gases such as ammonia, hydrogen sulfide, nitrogen dioxide, and nitric oxide, which possess strong polarity and acid–base characteristics, are especially well-suited for detection by c-MOF-based sensors. However, the practical application of c-MOFs remains limited by their relatively low sensitivity and reversibility. These limitations can be addressed through rational compositional engineering to design fully reversible and sensitive c-MOF chemiresistors. In addition, various structural designs including the formation of hollow and hierarchical architectures in both thin films and nanoparticles have been employed to enhance sensitivity. Because many of these approaches are recent and represent first attempts within the c-MOF research field, this review aims to provide a comprehensive overview of the state-of-the-art strategies, offering design guidance for the development of room-temperature-operating gas sensors targeting hazardous and environmental gases.
Keywords:
Conductive metal-organic frameworks, Chemiresistor, Gas sensor, Hazardous gas, Environmental gas1. INTRODUCTION
Metal–organic frameworks (MOFs) are porous crystalline materials composed of highly ordered metal clusters and organic ligands [1]. These materials are characterized by extremely high surface areas and tunable porosities arising from their microporous structures. Owing to the presence of exposed metal centers and functional organic moieties within their pores, MOFs exhibit unique catalytic behaviors and demonstrate remarkable performance in gas storage [2], separation [3], and catalysis [4]. The vast design flexibility enabled by the diverse combinations of metals and ligands has facilitated the continuous discovery of new MOF materials tailored for specific applications.
Despite their advantages, conventional MOFs generally possess wide bandgaps and exhibit poor electrical conductivities, limiting their use in electronic applications. However, in recent years, significant progress has been made in understanding the charge transport mechanisms in MOFs [5], including through-bond, extended conjugation, through-space, redox hopping, and guest-promoted charge transfer pathways (Fig. 1), as well as ion conduction through the framework pores. These insights have led to the emergence of conductive MOFs (c-MOFs), a subclass of MOFs that exhibit appreciable electrical conductivity even at room temperature.
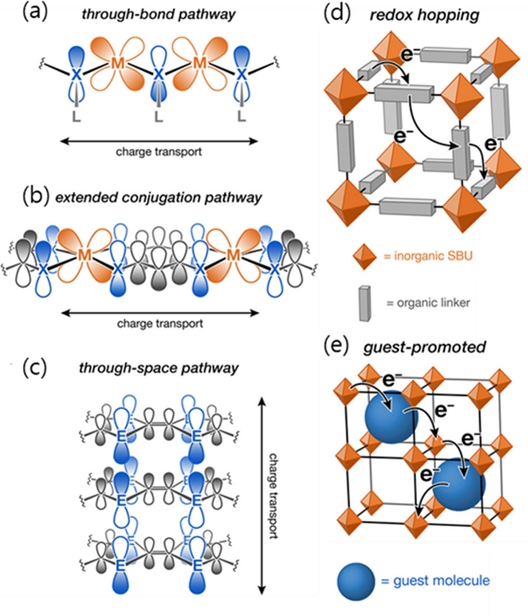
Schematic of electron transport mechanisms. (a) through-bond, (b) extended conjugation, (c) through-space, (d) redox hopping, and (e) guest-promoted pathways. Reprinted with permission from Ref. [5], Copyright (2020) American Chemical Society.
c-MOFs have attracted increasing interest for applications in which both electrical conductivity and porosity are desired, such as batteries [6], supercapacitors [7], organic electrochemical transistors [8], and more recently, gas sensors [9]. Among these, chemiresistive-type gas sensors based on c-MOFs are gaining attention because of their potential for the real-time, low-power, and miniaturized detection of hazardous and environmental gases. These sensors operate by monitoring changes in electrical resistance induced by charge transfer during interactions with target gas molecules. Their simple design and low-cost fabrication make them promising alternatives to conventional analytical methods, such as gas chromatography (GC) and proton transfer reaction–mass spectrometry (PTR-MS), especially for portable and integrated sensing platforms such as electronic noses.
Importantly, conventional chemiresistive gas sensors based on metal oxide semiconductors typically require high operating temperatures (200–400 °C), which result in high energy consumption and can cause thermal degradation of nearby components. By contrast, c-MOF-based gas sensors do not require external thermal activation. This offers distinct advantages for next-generation applications including autonomous vehicles, food quality monitoring, wearable healthcare devices, breath analysis for disease diagnosis, and smart agriculture. In addition, their ability to detect hazardous and environmental gases in real-time may significantly contribute to improving public health and environmental safety, making c-MOFs a promising platform for future sensor technologies.
2. TYPES AND SENSING MECHANISMS OF CONDUCTIVE MOF-BASED GAS SENSORS
To date, most c-MOFs achieve electrical conductivity through π–d conjugation, which arises from the periodic coordination between π-conjugated planar ligands and transition metal centers [10]. These c-MOFs commonly adopt two-dimensional layered structures, where interlayer π–π interactions facilitate the delocalization of charge carriers, resulting in intrinsic conductivity across the material. Typical π-conjugated ligands include benzene, triphenylene, and tetraazanaphthotetraphene derivatives, often functionalized with moieties such as hydroxy (-OH), amino (-NH2), or thiol (-SH) groups. The metal nodes coordinating these ligands vary widely and include Ni, Cu, Co, Zn, and Fe. The stacking mode of the resulting framework, and thus its conductivity, is heavily influenced by the specific metal-ligand combination, making the precise design of both the metal species and ligand functionality essential for tuning the electrical properties of c-MOFs.
The sensing mechanism of c-MOF-based gas sensors primarily relies on redox reactions facilitated by host–guest interactions [11]. Depending on the chemical nature of the metal nodes, the framework can offer Brønsted or Lewis acid sites (Fig. 2 (a)), which exhibit varying affinities for gases based on their basicity. For instance, ammonia (NH3), a strongly basic gas (pKb = 4.7), adsorbs more readily onto c-MOFs than weakly basic gases, such as CD3CN (pKb = 18.3) (Fig. 2 (b)), inducing greater changes in electrical resistance. Even in the absence of direct electron transfer via redox reactions, changes in local polarity caused by the dipole moment of gas molecules can modulate the electron density within the framework, leading to measurable resistive responses [12].
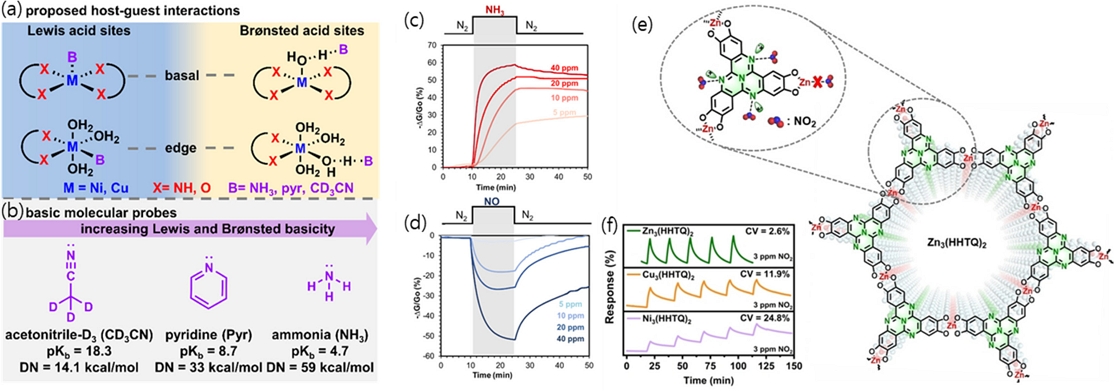
Sensing mechanisms of c-MOF chemiresistors. (a) Potential host sites of c-MOFs interacting with gases as either Brønsted or Lewis acid sites. (b) Three representative gases (acetonitrile-D3, pyridine, and ammonia) with their respective pKb and donor number (DN) values. Reprinted with permission from Ref. [11], Copyright (2020) American Chemical Society. Chemiresistive responses of devices integrated with Bi(HHTP) under an atmosphere of dry nitrogen to 15 min of exposure to (c) NH3 and (d) NO at 40, 20, 10, and 5 ppm. Reprinted with permission from Ref. [13], Copyright (2021) American Chemical Society. (e) Part of the structure of Zn3(HHTQ)2 and the potential gas reaction sites. (f) Response-recovery cycle plots of variation for Zn3(HHTQ)2, Cu3(HHTQ)2, Ni3(HHTQ)2 towards 3 ppm NO2. Reprinted with permission from Ref. [15], Copyright (2024) WILEY-VCH.
Aykanat et al. [13] recently demonstrated c-MOF chemiresistors based on Bi(HHTP) (where HHTP indicates hexahydroxytriphenylene) and evaluated their performance toward gases such as ammonia (NH3), nitric oxide (NO), acetone, ethanol, methanol, and isopropanol. In this system, the Bi nodes functioned as Lewis or Brønsted acid sites, interacting strongly with reducing and basic gases such as NH3, which donate electrons and consequently decrease the conductivity of the Bi(HHTP), indicating p-type semiconducting behavior (Fig. 2 (c)). Conversely, exposure to oxidizing gases such as NO led to an increase in conductivity (Fig. 2 (d)). Through a comparative analysis of various volatile organic compounds (VOCs), it was found that factors such as pKa, dipole moment, and dielectric constant were strongly correlated with the observed resistance changes. Gases capable of direct acid–base interactions, such as NH3 and NO, exhibited significantly higher sensitivities, even at low concentrations.
Moreover, the incorporation of heteroatoms such as nitrogen or boron into the ligand backbone creates chemically active sites that directly participate in gas sensing, thereby enabling novel sensing pathways [14]. Yan et al. [15] introduced Zn3(HHTQ)2 sensors based on the hexahydroxyltricycloquinazoline (HHTQ) ligand, which features uniformly distributed, highly reactive nitrogen atoms (Fig. 2 (e)). By coupling this ligand with relatively inert Zn metal nodes, they successfully prevented irreversible gas binding, which is commonly observed with more reactive metals [16], and achieved reversible NO2 sensing (Fig. 2 (f)). In this design, the nitrogen sites within the ligand act as primary reactive centers, providing both high sensitivity and excellent reversibility.
Environmental and hazardous gases, such as NO, NO2, NH3, and H2S, which exhibit diverse acid–base characteristics, interact with c-MOFs depending on their composition. These chemical interactions can be leveraged to design gas sensors with enhanced sensitivities and selectivities.
In addition to redox and acid–base sensing mechanisms, gas-induced structural deformations in flexible MOFs may serve as an additional sensing mechanism. Upon gas infiltration, flexible organic linkers can undergo tilting, twisting, or bending, which may perturb or enhance conductive pathways [17], thereby leading to a change in the overall resistance of the material. Jo et al. [18] demonstrated that Cu3(HHTT)2 c-MOFs exhibit structural distortions in response to increasing humidity levels. At approximately 25% relative humidity (RH), X-ray diffraction (XRD) patterns revealed widening of the interlayer d-spacing, along with in-plane twisting of the MOF layers induced by adsorbed water molecules. Although these observations were made under a relatively high humidity rather than in the presence of trace target gases, the results suggest that the structural flexibility of c-MOFs can be leveraged as a viable sensing mechanism in chemiresistive gas sensors.
Crystallinity plays a crucial role in maintaining the structural integrity and sensing behavior of c-MOF-based chemiresistors. Defect sites in sensing materials are often regarded as active gas adsorption sites because of their high chemical reactivity [19], In c-MOFs, a low crystallinity with abundant defect sites can lead to irreversible interactions with gas molecules, thereby hindering sensor reversibility. Noh et al. [20] synthesized highly crystalline Cu3(HIB)2 c-MOFs based on hexaiminobenzene ligands, achieving rod-shaped particles with widths of approximately 100 nm—two to three times larger than those of previously reported less-crystalline Cu3(HIB)2 materials (<40 nm) (Fig. 3 (a)). Owing to the refined synthetic conditions and improved crystallinity, the resulting Cu3(HIB)2 sensors exhibited a remarkable response of over 2,000% to 25 ppm NO gas, along with complete signal recovery, which was not observed in the less-crystalline control groups. Additionally, the sensor showed six-fold higher responses to NO than to various interfering gases, including NO2, H2S, SO2, CO, CO2, and NH3 (Fig. 3 (b)). These results highlight that highly crystalline Cu3(HIB)2 is a promising candidate material for the selective and reversible sensing of the hazardous environmental pollutant NO.
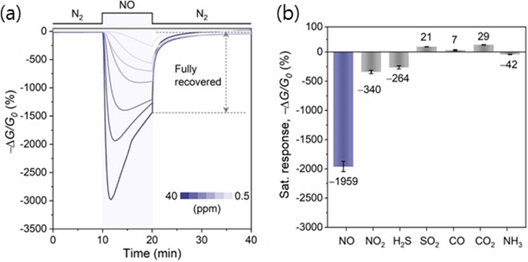
Gas sensing properties of Cu3(HIB)2 sensors. (a) Saturation sensing traces of Cu3(HIB)2 after 10 min of exposure to 40, 20, 10, 5, 2,1, and 0.5 ppm of NO. (b) Saturated responses of Cu3(HIB)2 to 25 ppm of NO, NO2, H2S, SO2, CO, and NH3, and 5000 ppm of CO2. Reprinted with permission from Ref. [20], Copyright (2024) WILEY-VCH.
Nevertheless, the inherently low reactivity and slow response rates at room temperature remain significant obstacles in achieving high sensitivity, underscoring the need for continued advancements in material design and sensor engineering.
3. STRATEGIES TO ENHANCE SENSITIVITY VIA THE FORMATION OF c-MOF GAS-SENSING FILMS
Enhancing gas accessibility through c-MOF sensing films is a crucial approach for maximizing sensitivity by ensuring that the target gas molecules can efficiently reach the overall active sites in the sensing materials. One effective strategy involves the fabrication of thin MOF films that facilitate rapid diffusion and efficient interactions between the gas molecules and sensing material. These films can be synthesized directly onto substrates via in situ growth [21], where parameters such as the reaction time and temperature are precisely controlled to tailor the film thickness and, consequently, its gas-sensing performance.
For example, c-MOFs have been grown directly on the surfaces of fibrous or yarn-like templates to form highly accessible sensing-film architectures. Smith et al. [22] fabricated electronic textiles by integrating Ni3(HHTP)2 and Ni3(HITP)2 (HITP = hexaiminotriphenylene) via direct solution-phase self-assembly onto fabric substrates (Fig. 4 (a)). The resulting c-MOF-coated textiles exhibited highly porous and gas-permeable structures, which contributed to their strong chemiresistive responses. Specifically, the response of Ni3(HHTP)2 to H2S reached 98%, whereas Ni3(HITP)2 showed a comparable response of 97% (Fig. 4 (b)). These substantial responses are attributed to the synergistic effect of the porous fiber network and the uniform deposition of c-MOFs on the fabric surface. In addition, the inherent flexibility of textile substrates enables sensor operation under mechanically dynamic conditions, such as twisting, bending, and wrapping, which broadens their applicability in wearable and flexible sensing platforms (Fig. 4 (c)). However, the direct in situ coating of c-MOFs can sometimes lead to the formation of undesired precipitated nanoparticles, which may act as barriers to gas diffusion and hinder sensor performance.
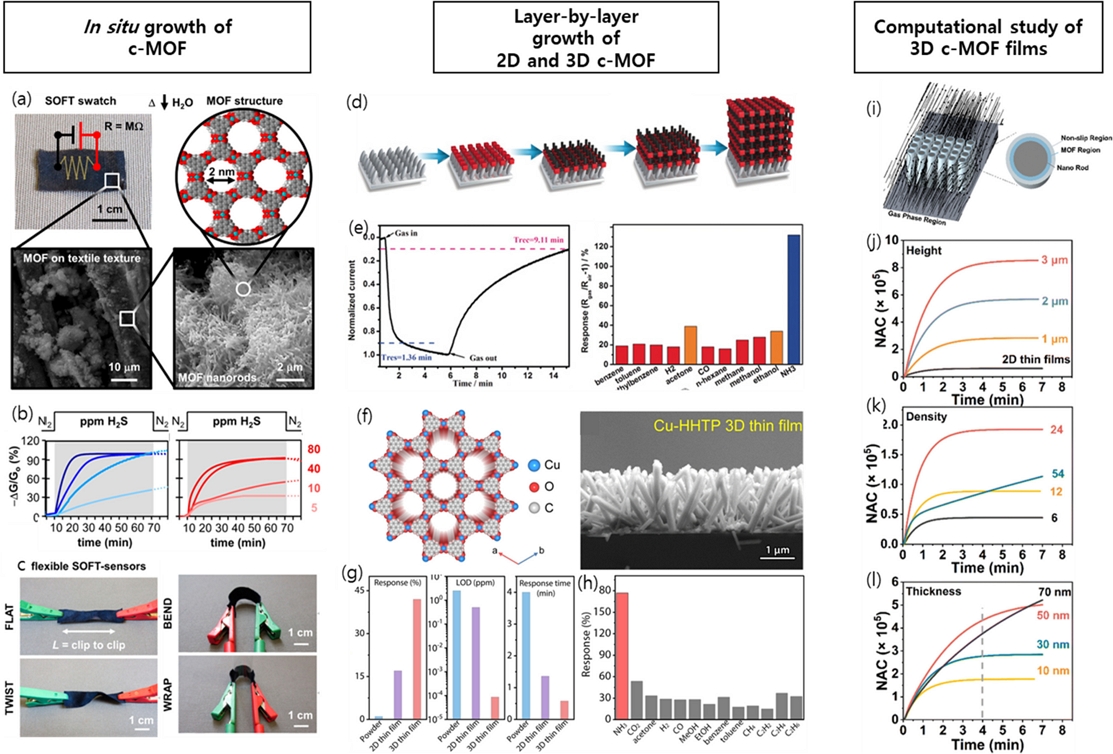
Design of various structured c-MOF chemiresistors. (a) Ni3(HHTP)2-integrated electronic textiles, and (b) representative responses for Ni3(HITP)2 (blue) and Ni3(HHTP)2 (red) electronic textile sensors when exposed to H2S. (c) Mechanical properties of electronic c-MOF fabric sensor devices. Reprinted with permission from Ref. [22], Copyright (2017) American Chemical Society. (d) Illustration of layer-by-layer coating process. (e) Response–recovery time curves of 2D Cu3(HHTP)2 sensors to 100 ppm NH3 and responses of 2D Cu3(HHTP)2 sensors toward various reducing gases. Reprinted with permission from Ref. [23], Copyright (2017) WILEY-VCH. (f) SEM image of 3D Cu3(HHTP)2. (g) Comparison of the response (1 ppm), limit of detection (ppm) and response time (min) of Cu3(HHTP)2 powder-, 2D thin film- and 3D thin film-based RT NH3 sensors. (i) Response toward NH3 and interfering gases of 100 ppm. Reprinted with permission from Ref. [25], Copyright (2021) WILEY-VCH. (i) Illustration of the model used in the computational simulation. (j-l) Time-dependent molecular capture quantities in the system with NAC as a function of the (j) film height, (k) density, and (l) thickness. Reprinted with permission from Ref. [26], Copyright (2024) Royal Society of Chemistry.
To address this issue, epitaxial growth methods have been developed to enable the controlled and oriented growth of MOFs, thereby minimizing nanoparticle precipitation and improving film uniformity. Given the well-defined coordination between the metal ions and organic ligands in c-MOFs, the layer-by-layer (LBL) coating technique is particularly suitable for constructing ultrathin and uniform films [23]. In the LBL process, the substrate is alternately exposed to a metal ion solution and a ligand solution, with intermediate rinsing steps in fresh deionized water or ethanol to remove unbound species. Each coating cycle contributes several distinct layers of the c-MOF, and the total thickness of the sensing film can be precisely controlled by varying the number of cycles (Fig. 4 (d)).[24] This technique enables the fine-tuning of film morphology and facilitates the formation of homogeneous nanoscale c-MOF layers, making it ideal for high-performance gas-sensing applications.
Yao et al. [23] first demonstrated the use of LBL-assembled c-MOF films for chemiresistive gas sensing. In their work, Cu3(HHTP)2 was employed as the active sensing material and a spray-assisted LBL technique was used, wherein metal and ligand precursor solutions were alternately sprayed onto the electrode substrate. The spraying process was repeated for a defined number of cycles to control the film thickness. This study revealed that as the thickness of the Cu3(HHTP)2 film decreased, the sensor response increased disproportionately.
Notably, the film assembled with 10 LBL cycles exhibited the highest response—an impressive 129% change in conductance (−ΔG/G₀ upon exposure to 100 ppm NH3 (Fig. 4 (e)). Moreover, the sensor exhibited superior selectivity for NH3 over a wide range of potential interference gases, including benzene, toluene, ethylbenzene, H2, acetone, CO, n-hexane, methane, methanol, and ethanol. These results indicate that optimizing the film thickness is crucial for maximizing gas sensitivity. However, it should also be noted that extremely thin films, while yielding high sensitivity, tend to suffer from a significantly increased film resistance owing to the reduced overall electrical conductivity. Therefore, selecting an appropriate number of LBL cycles is essential to strike a balance between the sensitivity and practical electrical performance for real-world sensing applications.
Further enhancement of gas accessibility can be achieved by applying an LBL coating onto three-dimensional (3D) hierarchical insulating scaffold templates. These porous architectures promote molecular diffusion and significantly increase the number of accessible active sites. Lin et al. [25] demonstrated this approach by integrating LBL-grown Cu3(HHTP)2 films with TiO2 nanorod scaffold substrates (Fig. 4 (f)). The thickness of the Cu3(HHTP)2 films was precisely controlled by varying the number of LBL cycles from 8 to 36, and the film thickness increased linearly at a rate of approximately 2 nm per cycle.
The sensor fabricated on the 3D scaffold exhibited a remarkable response of 42% to 1 ppm NH3, which was 16100%, and 250% higher than those of the powder-based and 2D planar thin-film sensors, respectively (Fig. 4 (g)). Furthermore, the limit of detection (LOD) was as low as 87 ppt—approximately 5,000 and 1,000 times more sensitive than the powder and 2D film counterparts, respectively. The response time was also significantly improved to 35 s for the 3D thin film compared to 82 and 240 s for the 2D thin film and powder, respectively. The selectivity of the 3D Cu3HHTP2 sensors was higher than that of other interfering gases, including CO2, acetone, H2, CO, methanol, ethanol, benzene, toluene, CH4, C2H2, C2H4, and C2H6 (Fig. 4 (h)).
Interestingly, during the initial stages of LBL growth, Cu3(HHTP)2 did not immediately form a continuous film but instead developed as isolated island-like structures on the TiO2 nanorod surfaces. This suggests that a minimum number of coating cycles is insufficient to achieve a fully connected and electrically conductive film. Consistently, gas-sensing tests revealed that films with thicknesses of 6 and 12 nm exhibited lower responses than the 20-nm-thick film. These observations imply a trade-off between the number of accessible active sites and gas diffusivity, indicating that there exists an optimal film thickness for maximizing performance. Therefore, careful optimization of the film thickness is crucial for achieving a proper balance between gas accessibility, film connectivity, and overall sensing efficiency.
Pan et al. [26] attempted to elucidate the interplay among several factors affecting the gas-sensing behavior of 3D c-MOF thin films using computational simulations (Fig. 4 (i)). Specifically, this study analyzed how the gas transport dynamics are influenced by the coating thickness of c-MOFs when 3D rod-shaped pillars are arranged with defined lengths and spatial densities (Figs. 4 (j–l)). Their study revealed that the effective diffusion surface area increased with film thickness; however, when the c-MOF coating layers became excessively thick, individual 3D structural units began to merge, reducing the available diffusion pathways. When simulating the number of atoms captured (NAC) under various conditions to understand the time-dependent molecular capture quantities in this system, it was observed that increasing the height of the 3D pillar consistently led to higher NAC values.
Moreover, the simulations showed that both increased thickness and density resulted in a higher NAC. However, in cases where the film was too dense or thick, gas diffusion was hindered, leading to a plateau or a slower rate of increase in NAC over time. These findings suggest that, while denser and thicker films can enhance the initial gas capture, there is a threshold beyond which gas transport becomes limited, emphasizing the need to optimize both the density and thickness for efficient sensing performance.
Huang et al. [27] developed hierarchical c-MOF film structures that exhibit superior gas-sensing performance by facilitating multiscale diffusion pathways. Hierarchical films were fabricated using dense sacrificial ZIF-8 films as templates (Fig. 5 (a)). Upon immersion in an HHTP ligand solution, the ZIF-8 films underwent partial transformation into Cu3(HHTP)2 via a thermodynamically driven process. Instead of being fully replaced, ZIF-8 served as a structural scaffold, with the c-MOF growing primarily on its surface (Fig. 5 (b)). This partial conversion resulted in a hierarchically porous and hollow architecture (denoted as Zn-HHTP-H). The Zn-HHTP-H films were compared with bulk-type Zn-HHTP films (denoted as Zn-HHTP-B).
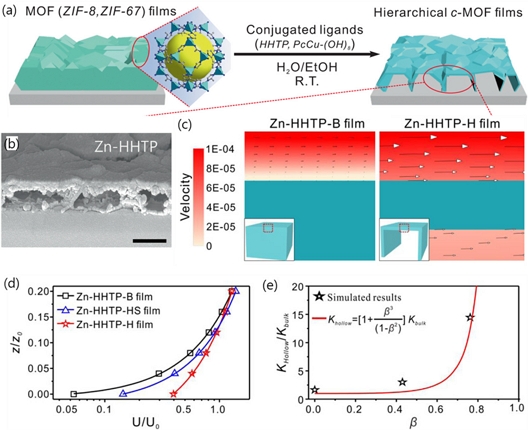
Gas accessible c-MOF film structures. (a) Schematic of the transformation of insulating 3D MOF film precursors to hierarchical c-MOF films. (b) Cross-sectional scanning electron microscopy (SEM) images of Zn-HHTP-H. (c) Vectors and magnitudes of velocities in the plane of symmetry. The inset images show the models for two different Zn-HHTP films. (d) Average surface velocities U in different planes above the surface of the Zn-HHTP films. U0 and z0 represent the average inlet velocity and the thickness of the films, respectively. (e) Theoretical permeability KHollow for the Zn-HHTP films with varying hollow volume ratios β. Reprinted with permission from Ref. [27], Copyright (2023) Springer Nature.
Computational simulations demonstrated that the masstransfer efficiency and velocity improved significantly when Zn-HHTP-H films with cavity sizes exceeding 500 nm were used. The enlarged pore structures enhanced both the gas permeability and transport velocity of the gas molecules (Figs. 5 (c–e)), contributing to faster response times in chemiresistive sensing at RT. These findings highlight the potential of the hierarchically engineered c-MOF films for high-performance gas-sensing applications under ambient conditions. In addition, these findings underscore the importance of film morphology in c-MOF-based chemiresistors, with 3D, hollow, and hierarchical architectures offering substantial advantages in terms of mass transport and gas accessibility, ultimately leading to improved sensing performance.
4. APPROACHES FOR IMPROVING THE SENSITIVITY THROUGH THE MORPHOLOGICAL CONTROL OF c-MOFs NANOPOWDERS
In addition to thin-film formation strategies, engineering c-MOFs into nanostructured particles can significantly enhance gas accessibility [28]. The inherently high surface-area-to-volume ratio of nanoparticles enables greater exposure of the active sites and promotes more efficient interactions with the analyte gases. Many 2D c-MOFs favor anisotropic crystal growth, resulting in morphologies such as nanorods and nanoflakes. Wrogenmann et al. [29] synthesized rod- and flake-like Cu3(HHTP)2 particles by precisely controlling the reaction conditions. Structural characterization revealed that the stacking sequences differed between the morphologies: the rods exhibited a tilted stacking mode, whereas the flakes adopted an eclipsed stacking configuration. The N2 adsorption isotherm data showed that the flake-like particles possessed a significantly higher surface area (424 m2/g) than the rod-like particles (86 m2/g). These results highlight the importance of selecting morphologies with maximum surface areas to ensure more efficient exposure to target gases.
Jo et al. [16] further explored the effect of the particle size on the gas-sensing performance of Cu3(HHTP)2 nanoflakes. The as-synthesized Cu3(HHTP)2 consisted of nanoflakes with a broad size distribution, which were subsequently size-fractionated via differential-speed centrifugation. By gradually decreasing the centrifugation speed, they successfully collected suspensions of larger and smaller particles ranging from approximately 400 to 150 nm. Gas sensing tests revealed a clear size-dependent behavior: the smallest nanoflakes exhibited a response of 89.4%, whereas the largest nanoflakes showed a reduced response of 61.1%. This trend confirms that smaller particles provide greater exposure to gas-reactive sites, which is attributed to the nano-size effect of secondary particles [30], thereby enhancing the sensor sensitivity.
Huang et al. [31] further advanced this concept by employing insulating MOF templates and the Kirkendall effect to fabricate various hierarchical c-MOF nanostructures. In their study, rhombohedral ZIF-8 crystals were used as templates and continuous exposure to the ligand solution induced the following sequence of processes: (i) formation of a secondary amorphous layer, (ii) dissolution and recrystallization of the framework, and (iii) evolution into a hollow c-MOF structure (Fig. 6 (a)).
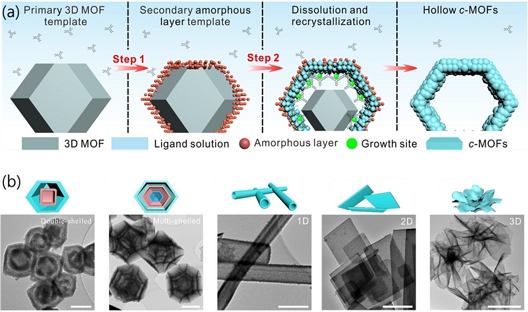
Gas accessible structures of c-MOF nanoparticles. (a) Schematic of the two-step transformation mechanism of ZIF-8 to hollow Zn-HHTP nanoparticles. (b) TEM images of double-shelled hollow Co-HHTP@Zn-HHTP nanoparticles, hollow multishelled CuBHT nanoparticles, Zn-HHB nanotubes, CuBHT nanosheets, Cu-HHB nanoflowers, and Cu-BHT nanosheets-assembled frame nanofilm. Reprinted with permission from Ref. [31], Copyright (2023) WILEY-VCH.
The resulting morphologies could be finely tuned by adjusting parameters such as the water content in the solvent mixture and the reaction temperature. Furthermore, by varying the compositions of the template and ligand precursors, a wide range of nanostructures, including double-shelled, multishelled, nanoflower-like, nanotubular, and nanoplate morphologies, were successfully synthesized (Fig. 6 (b)). These innovative 3D structural engineering strategies led to a substantial enhancement in the chemiresistive gas-sensing performance compared to conventional bulk c-MOF particles. These architectures contributed to enhanced gas accessibility and adsorption efficiency.
Collectively, these strategies demonstrate that tuning the dimensionality and morphology of c-MOF sensing films is essential for achieving high sensitivity and fast response in gas-detection applications. Tunable 3D structuring and nanoscale engineering provide versatile toolboxes for the rational design of next-generation c-MOF gas sensors.
5. OUTLOOK AND CONCLUSION
Recent advances in c-MOF-based gas-sensing research range from the development of novel material compositions through new combinations of metal nodes and organic ligands to the design of highly gas-accessible structures with tailored morphologies in both thin films and particulate forms. Among these, the construction of hierarchical and hollow c-MOF architectures has demonstrated particularly promising results, yielding significant improvements in the hazardous and environmental gas-sensing performance. These structures have enabled exceptional sensitivity and ultralow detection limits, highlighting their potential for use in next-generation gas-sensing technologies.
From the perspective of conventional gas sensor design, the strategy of integrating heterogeneous catalytic materials, such as metals, metal oxides, and MOFs, into precisely engineered nanostructured c-MOF systems has not yet been fully explored. This represents a promising research direction for the development of next-generation high-performance gas sensors.
In addition, the design of c-MOF compositions that maintain measurable electrical conductivities remains essential. Leveraging one of the inherent advantages of MOFs, namely their gas sieving and filtration capabilities [32], will also be crucial for improving selectivity in complex gas environments and should be regarded as a key focus for the future development of c-MOF-based gas sensors.
By integrating various advanced strategies currently being developed, c-MOFs have the potential to emerge as optimal materials for the RT detection of hazardous and environmental gases. This review aimed to enhance the understanding of researchers in the sensor community and provide new insights that may guide future directions for c-MOF-based gas sensing research.
Acknowledgments
This work was supported by the Kyungpook National University Research Fund (2025).
References
-
S.L. James, Metal-organic frameworks, Chem. Soc. Rev. 32 (2003) 276–288.
[https://doi.org/10.1039/b200393g]

-
Y. He, W. Zhou, G. Qian, B. Chen, Methane storage in metal–organic frameworks, Chem. Soc. Rev. 43 (2014) 5657–5678.
[https://doi.org/10.1039/C4CS00032C]

-
X. Zhao, Y. Wang, D.-S. Li, X. Bu, P. Feng, Metal–Organic Frameworks for Separation, Adv. Mater. 30 (2018) 1705189.
[https://doi.org/10.1002/adma.201705189]

-
J. Lee, O.K. Farha, J. Roberts, K.A. Scheidt, S.T. Nguyen, J.T. Hupp, Metal–organic framework materials as catalysts, Chem. Soc. Rev. 38 (2009) 1450–1459.
[https://doi.org/10.1039/b807080f]

-
L.S. Xie, G. Skorupskii, M. Dincă, Electrically Conductive Metal–Organic Frameworks, Chem. Rev. 120 (2020) 8536–8580.
[https://doi.org/10.1021/acs.chemrev.9b00766]

-
R. Zhao, Z. Liang, R. Zou, Q. Xu, Metal-Organic Frameworks for Batteries, Joule 2 (2018) 2235–2259.
[https://doi.org/10.1016/j.joule.2018.09.019]

-
R.R. Salunkhe, Y.V. Kaneti, Y. Yamauchi, Metal–Organic Framework-Derived Nanoporous Metal Oxides toward Supercapacitor Applications: Progress and Prospects, ACS Nano 11 (2017) 5293–5308.
[https://doi.org/10.1021/acsnano.7b02796]

-
G. Wu, J. Huang, Y. Zang, J. He, G. Xu, Porous Field-Effect Transistors Based on a Semiconductive Metal–Organic Framework, J. Am. Chem. Soc. 139 (2017) 1360–1363.
[https://doi.org/10.1021/jacs.6b08511]

-
Y.-M. Jo, Y.K. Jo, J.-H. Lee, H.W. Jang, I.-S. Hwang, D.J. Yoo, MOF-Based Chemiresistive Gas Sensors: Toward New Functionalities, Adv. Mater. 35 (2022) 2206842.
[https://doi.org/10.1002/adma.202206842]

-
D. Sheberla, L. Sun, M.A. Blood-Forsythe, S. Er, C.R. Wade, C.K. Brozek, et al., High Electrical Conductivity in Ni3(2,3,6,7,10,11-hexaiminotriphenylene)2, a Semiconducting Metal–Organic Graphene Analogue, J. Am. Chem. Soc. 136 (2014) 8859–8862.
[https://doi.org/10.1021/ja502765n]

-
R.M. Stolz, A. Mahdavi-Shakib, B.G. Frederick, K.A. Mirica, Host–Guest Interactions and Redox Activity in Layered Conductive Metal–Organic Frameworks, Chem. Mater. 32 (2020) 7639–7652.
[https://doi.org/10.1021/acs.chemmater.0c01007]

-
M. Wang, Z. Zhang, H. Zhong, X. Huang, W. Li, M. Hambsch, et al., Surface-Modified Phthalocyanine-Based Two-Dimensional Conjugated Metal–Organic Framework Films for Polarity-Selective Chemiresistive Sensing, Angew. Chem. 133 (2021) 18814–18820.
[https://doi.org/10.1002/ange.202104461]

-
A. Aykanat, C.G. Jones, E. Cline, R.M. Stolz, Z. Meng, H.M. Nelson, et al., Conductive Stimuli-Responsive Coordination Network Linked with Bismuth for Chemiresistive Gas Sensing, ACS Appl. Mater. Interfaces 13 (2021) 60306–60318.
[https://doi.org/10.1021/acsami.1c14453]

-
L.S. Panchakarla, K.S. Subrahmanyam, S.K. Saha, A. Govindaraj, H.R. Krishnamurthy, U.V. Waghmare, et al., Synthesis, Structure, and Properties of Boron- and Nitrogen-Doped Graphene, Adv. Mater. 21 (2009) 4726–4730.
[https://doi.org/10.1002/adma.200901285]

-
X. Yan, J. Chen, X. Su, J. Zhang, C. Wang, H. Zhang, et al., Redox Synergy: Enhancing Gas Sensing Stability in 2D Conjugated Metal–Organic Frameworks via Balancing Metal Node and Ligand Reactivity, Angew. Chem. Int. Ed. 63 (2024) e202408189.
[https://doi.org/10.1002/anie.202408189]

-
Y.-M. Jo, K. Lim, J.W. Yoon, Y.K. Jo, Y.K. Moon, H.W. Jang, et al., Visible-Light-Activated Type II Heterojunction in Cu3(hexahydroxytriphenylene)2/Fe2O3 Hybrids for Reversible NO2 Sensing: Critical Role of π–π* Transition, ACS Cent. Sci. 7 (2021) 1176–1182.
[https://doi.org/10.1021/acscentsci.1c00289]

-
D. Fairen-Jimenez, S.A. Moggach, M.T. Wharmby, P.A. Wright, S. Parsons, T. Düren, Opening the Gate: Framework Flexibility in ZIF-8 Explored by Experiments and Simulations, J. Am. Chem. Soc. 133 (2011) 8900–8902.
[https://doi.org/10.1021/ja202154j]

-
Y.-M. Jo, D.-H. Kim, J. Wang, J.J. Oppenheim, M. Dincă, Humidity-Mediated Dual Ionic–Electronic Conductivity Enables High Sensitivity in MOF Chemiresistors, J. Am. Chem. Soc. 146 (2024) 20213–20220.
[https://doi.org/10.1021/jacs.4c05343]

-
M.-W. Ahn, K.-S. Park, J.-H. Heo, J.-G. Park, D.-W. Kim, K. J. Choi, et al., Gas sensing properties of defect-controlled ZnO-nanowire gas sensor, Appl. Phys. Lett. 93 (2008) 263103.
[https://doi.org/10.1063/1.3046726]

-
H.-J. Noh, D.L. Pennington, J.-M. Seo, E. Cline, G. Benedetto, J.B. Baek, et al., Reversible and Ultrasensitive Detection of Nitric Oxide Using a Conductive Two-Dimensional Metal–Organic Framework, Angew. Chem. Int. Ed. 64 (2024) e202419869.
[https://doi.org/10.1002/anie.202419869]

-
M.-S. Yao, W.X. Tang, G.-E. Wang, B. Nath, G. Xu, MOF Thin Film-Coated Metal Oxide Nanowire Array: Significantly Improved Chemiresistor Sensor Performance, Adv. Mater. 28 (2016) 5229–5234.
[https://doi.org/10.1002/adma.201506457]

-
M.K. Smith, K.A. Mirica, Self-Organized Frameworks on Textiles (SOFT): Conductive Fabrics for Simultaneous Sensing, Capture, and Filtration of Gases, J. Am. Chem. Soc. 139 (2017) 16759–16767.
[https://doi.org/10.1021/jacs.7b08840]

-
M.-S. Yao, X.-J. Lv, Z.-H. Fu, W.-H. Li, W.-H. Deng, G.-D. Wu, et al., Layer-by-Layer Assembled Conductive Metal–Organic Framework Nanofilms for Room-Temperature Chemiresistive Sensing, Angew. Chem. Int. Ed. 56 (2017) 16510–16514.
[https://doi.org/10.1002/anie.201709558]

-
R. Zheng, Z.-H. Fu, W.-H. Deng, Y. Wen, A.-Q. Wu, X.-L. Ye, G. Xu, The Growth Mechanism of a Conductive MOF Thin Film in Spray-based Layer-by-layer Liquid Phase Epitaxy, Angew. Chem. Int. Ed. 61 (2022) e202212797.
[https://doi.org/10.1002/anie.202212797]

-
Y. Lin, W.-H. Li, Y. Wen, G.-E. Wang, X.L. Ye, et al., Layer-by-Layer Growth of Preferred-Oriented MOF Thin Film on Nanowire Array for High-Performance Chemiresistive Sensing, Angew. Chem. Int. Ed. 60 (2021) 25758–25761.
[https://doi.org/10.1002/anie.202111519]

-
Y. Pan, W. Sun, J. Chen, Y. Lin, Y.-J. Chen, Z. Wen, et al., Mass transport control over a conductive MOF 3D thin film to improve gas sensing, J. Mater. Chem. A 12 (2024) 26902–26908.
[https://doi.org/10.1039/D4TA04855E]

-
C. Huang, X. Shang, X. Zhou, Z. Zhang, X. Huang, Y. Lu, et al., Hierarchical conductive metal-organic framework films enabling efficient interfacial mass transfer, Nat. Commun. 14 (2023) 3850.
[https://doi.org/10.1038/s41467-023-39630-y]

-
J.-H. Lee, Gas sensors using hierarchical and hollow oxide nanostructures: Overview, Sens. Actuators B Chem. 140 (2009) 319–336.
[https://doi.org/10.1016/j.snb.2009.04.026]

-
J.M. Wrogemann, M.J. Lüther, P. Bärmann, M. Lounasvuori, A. Javed, M. Tiemann, et al., Overcoming Diffusion Limitation of Faradaic Processes: Property-Performance Relationships of 2D Conductive Metal-Organic Framework Cu3(HHTP)2 for Reversible Lithium-Ion Storage, Angew. Chem. Int. Ed. 62 (2023) e202303111.
[https://doi.org/10.1002/anie.202303111]

-
E. Comini, Metal oxide nano-crystals for gas sensing, Anal. Chim. Acta 568 (2006) 28–40.
[https://doi.org/10.1016/j.aca.2005.10.069]

-
C. Huang, W. Sun, Y. Jin, Q. Guo, D. Mücke, X. Chu, et al., A General Synthesis of Nanostructured Conductive Metal–Organic Frameworks from Insulating MOF Precursors for Supercapacitors and Chemiresistive Sensors, Angew. Chem. Int. Ed. 63 (2023) e202313591.
[https://doi.org/10.1002/anie.202313591]

-
S.M. Lee, Y.J. Kim, S.J. Park, W.S. Cheon, J. Kim, G.B. Nam, et al., In-Situ Growth of 2D MOFs as a Molecular Sieving Layer on SnS2 Nanoflakes for Realizing Ultraselective H2S Detection, Adv. Funct. Mater. 35 (2025) 2417019.
[https://doi.org/10.1002/adfm.202417019]


Young-Moo Jo is an Assistant Professor in the School of Materials Science and Engineering at Kyungpook National University (KNU). He received his B.S. and PhD degrees in Materials Science and Engineering from Korea University in 2016 and 2022, respectively. After serving as a BK21 research professor at Korea University for six months, he joined the Department of Chemistry at the Massachusetts Institute of Technology (MIT) as a postdoctoral associate through 2025. His research focuses on the synthesis and functionalization of inorganic conductive materials, including electrically conductive metal–organic frameworks and metal oxides, with an emphasis on their application in chemiresistive gas sensor devices.
