Development of a Colorimetric H2 Sensor Based on PtWO3 Nanoparticles
 ; Dong Geon Jung1, +
; Dong Geon Jung1, +
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Hydrogen (H2) is a promising clean-energy carrier; however, it poses safety concerns because of its high flammability and low ignition energy, necessitating the timely and reliable detection of leaks. In this study, a power-free colorimetric hydrogen sensor was developed by dispersing Pt-doped tungsten trioxide (PtWO3) nanoparticles into a polydimethylsiloxane (PDMS) matrix and depositing the composite onto an alumina substrate via spin coating. The spin speed (600–900 rpm) was optimized to achieve uniform membrane morphology and responsive optical behavior. Upon exposure to 1%–5% H2 (balanced with N2), the sensor exhibited a distinct and concentration-dependent color transition from light yellow to deep blue, corresponding to ΔE values exceeding 2.3, readily perceptible by the human eye under ambient conditions (23°C, 30% RH). The system demonstrated rapid response within minutes and complete color recovery upon re-exposure to air, confirming its reversibility. Furthermore, selectivity measurements against CO, NO, NO2, and CO2 indicated minimal interference. This power-independent, visually legible PtWO3/PDMS sensor offers a simple and practical platform for real-time hydrogen leak monitoring in energy and industrial safety applications.
Keywords:
Colorimetric gas sensor, H2 gas, PtWO3, PDMS1. INTRODUCTION
Hydrogen (H2) is a highly flammable and colorless gas with very low ignition energy, making early leakage detection essential for ensuring operational safety [1-3]. Conventional semiconductor and electrochemical hydrogen sensors have been extensively studied because of their high sensitivity and quantitative response characteristics [4-7]. However, these electronic sensors inherently require external power sources and signal processing circuits, which limit their applicability in power-restricted or visually monitored environments. Moreover, they often require high operating temperatures and exhibit degraded performance under fluctuating humidity and temperature conditions [8-10].
To overcome these limitations, colorimetric and gasochromic sensing techniques that rely on the visible color changes induced by chemical interactions between sensing materials and target gases have emerged as promising alternatives for hydrogen detection [11-16]. In such systems, the degree of visible color change can be quantified using the color difference (ΔE) parameter within the International Commission on Illumination (CIE) LAB color space, which represents variations in brightness (L*), chroma (a*), and hue (b*). According to the MacAdam limit, a ΔE value exceeding 2.3 is generally regarded as a color difference threshold perceptible by the human eye [17].
In this study, Pt-doped tungsten trioxide (PtWO3) nanoparticles were dispersed within a polydimethylsiloxane (PDMS) matrix and spin-coated onto an alumina (Al2O3) substrate to fabricate a membrane-type colorimetric hydrogen sensor. The coating uniformity, optical response, and gas diffusion behavior were optimized by systematically varying the spin speed (600–900 rpm). The fabricated sensor exhibited distinct and concentration-dependent color transitions upon exposure to 1%–5% H2, along with rapid recovery under ambient air. Because the sensor operates without an external power supply and allows real-time visual monitoring, this device provides a promising solution for hydrogen leak detection in power-limited or on-site industrial environments [18,19].
2. EXPERIMENTAL
2.1 Material and synthesis of PtWO3 nanoparticles
All chemicals were of analytical grade, purchased from Sigma-Aldrich (USA), and used as received without further purification. The precursors included tungsten hexachloride (WCl6, 99.9%) as the tungsten source and hexachloroplatinic acid as the platinum source. Ethanol (99.9%) and deionized (DI) water were used as solvents throughout the synthesis.
The Pt-doped tungsten trioxide (PtWO3) nanoparticles were synthesized via controlled thermal decomposition under a nitrogen atmosphere.
First, 17.4 mmol of WCl6 and 11.9 mmol of hexachloroplatinic acid were dissolved in 36 mL of ethanol under vigorous stirring until a homogeneous suspension was obtained. The mixed solution was then dried at 120°C for 12 h under a nitrogen atmosphere to remove the residual solvent and initiate precursor decomposition.
Subsequently, the dried powder was calcined at 300°C for 1 h to reach the crystalline PtWO3 phase, optimizing its color-change behavior toward hydrogen gas. A slow heating rate (approximately 2°C min⁻¹) was maintained to prevent agglomeration and promote uniform Pt incorporation into the WO3 lattice.
After cooling naturally to room temperature, the resulting grayish-blue PtWO3 powder was finely ground in an agate mortar to yield well-dispersed nanoparticles (20–50 nm). The powders were stored in airtight glass vials to prevent moisture absorption and were directly used to fabricate the PDMS-based colorimetric membranes described in Section 2.2.
2.2 Fabrication of colorimetric membrane on alumina substrates
The PtWO3/PDMS composite solution was prepared by dispersing the as-synthesized PtWO3 nanoparticles in a pre-polymerized PDMS matrix (Sylgard 184, base:curing agent = 10:1 wt ratio). The mixture was magnetically stirred for 1 h to achieve a uniform dispersion of the nanoparticles, followed by vacuum degassing for 30 min to remove entrapped air bubbles. The absence of air voids is critical for ensuring film uniformity and optical transparency, which directly affects the reproducibility of the colorimetric response.
After degassing, approximately 100 µL of the composite solution was drop-cast onto a pre-cleaned alumina (Al2O3) substrate (10 × 10 mm2 ). The substrates were ultrasonically cleaned in ethanol and DI water for 10 min each and dried under nitrogen flow before coating. The film was then spin-coated at four different rotation speeds (600, 700, 800, and 900 rpm) for 60 s to investigate the influence of coating thickness and surface uniformity on optical performance.
Following spin coating, the samples were thermally cured at 80°C for 2 h in a convection oven to complete the PDMS cross-linking and immobilize the PtWO3 nanoparticles within the elastomeric network. The resulting membranes exhibited smooth and flexible surfaces with good adhesion to the alumina substrate. The film thickness decreased from approximately 120 µm at 600 rpm to 60 µm at 900 rpm, as estimated using a surface profilometer. The coating prepared at 800 rpm showed the best balance between uniformity and mechanical robustness.
The fully cured membranes were subsequently stored in a desiccator prior to the gas-sensing experiments to prevent ambient moisture adsorption, which can alter the baseline color of PDMS.
2.3 Gas exposure and colorimetric measurement
All test gases, including H2, CO, NO, NO2, and CO2, were supplied by Korea Standard Gas (Republic of Korea) as N2-balanced certified calibration gases. A 10% H2 (N2-balanced) cylinder was used as the parent source and was diluted with high-purity N2 and dry air using mass flow controllers (MFCs) to achieve final hydrogen concentrations of 1%, 2%, 3%, 4%, and 5%. The total flow rate was maintained at 1000 sccm, which consisted of 500 sccm of dry air and 500 sccm of a mixed stream of N2 and the target gas. Before each run, all gas lines were purged with pure N2 for at least 10 min to remove any residual species.
Each prepared gas mixture was collected in a Tedlar bag to ensure compositional accuracy and flow stability. Gas exposure tests were conducted in a custom-built acrylic chamber (6 × 4 × 8 cm3 ) that could be evacuated to a near-vacuum state before gas introduction. After evacuation, the pre-filled Tedlar bag gas was injected through a controlled inlet until the chamber pressure reached the ambient level, ensuring a homogeneous gas distribution around the sensor.
All measurements were performed at a controlled temperature of 23 ± 1°C and relative humidity of 30 ± 2%, with continuous monitoring using a temperature and humidity sensor (BLUETEC, BO-807). The PtWO3/PDMS-coated alumina sensor was placed at the center of the chamber and exposed to the target gas for 15 min (reaction phase).
Immediately after the exposure phase, the recovery behavior was evaluated by opening the chamber directly in ambient air, allowing natural desorption and color restoration under atmospheric conditions. The color change process during both exposure and recovery was recorded using a Galaxy Note 8 smartphone camera, which provided real-time visual documentation under ambient lighting.
The captured images were analyzed using ImageJ software (version 1.54, NIH, USA) to extract the corresponding RGB intensity values, which were then converted to CIELAB (L*, a*, b*) coordinates. The overall color difference (ΔE) was calculated as follows:
| (1) |
where the subscripts 1 and 2 represent the RGB values before and after gas exposure, respectively. A ΔE > 2.3 was defined as the threshold for a color change perceptible by the human eye [17]. Each experiment was repeated thrice to verify the reproducibility and stability of the colorimetric response. To ensure consistent image capture, all photographs were captured under controlled lighting conditions with fixed white balance and exposure settings.
3. RESULTS AND DISCUSSIONS
3.1 Spin-coating speed optimization
The effect of the spin coating rate on the optical response of the PtWO3/PDMS colorimetric membrane was systematically investigated, as illustrated in Fig. 1.
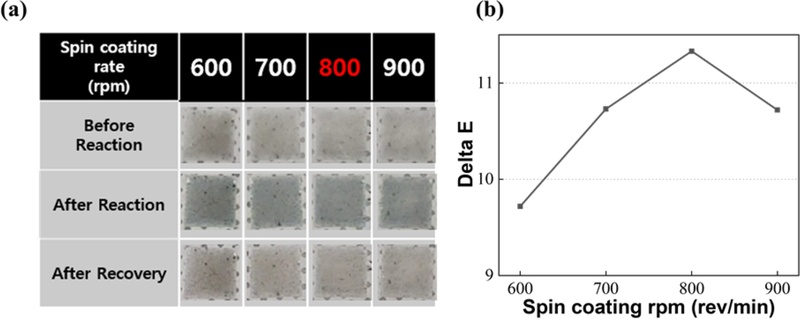
Optimization of spin-coated PtWO3/PDMS membranes for H2 sensing. (a) Colorimetric images of PtWO3/PDMS membranes before/after 3% H2 exposure and after recovery at different spin rates (600–900 rpm), (b) ΔE variations showing the highest contrast at 800 rpm.
Fig. 1(a) shows the photographic images of the sensor surfaces before/after exposure to 3% H2, and after the recovery phase at four different coating speeds (600, 700, 800, and 900 rpm).
At lower speeds (600 and 700 rpm), the films appeared relatively thick and slightly opaque, resulting in weaker and slower color transitions during H2 exposure.
In contrast, the film coated at 800 rpm exhibited the most distinct and rapid color change from light gray to deep blue, indicating optimal film thickness and uniform nanoparticle dispersion.
When the spin speed was further increased to 900 rpm, the coating became too thin and partially nonuniform, reducing the active volume for gas interaction and thus diminishing the visible color contrast.
The quantitative colorimetric responses corresponding to these visual results are shown in Fig. 1(b).
The ΔE value increased with the spin speed up to 800 rpm and then decreased beyond that point, forming a bell-shaped curve, which is a characteristic of diffusion-limited thin-film systems.
The sensor fabricated at 800 rpm achieved the highest ΔE of approximately 11.3 upon exposure to 3% H2, which is well above the perceptibility threshold (ΔE > 2.3), confirming a clearly visible color change to the human eye.
This optimal behavior can be attributed to a balanced combination of light transmittance, gas permeability through the PDMS matrix, and effective optical path length within the PtWO3-dispersed layer.
After the exposure phase, all samples showed reversible color recovery when the chamber was opened to ambient air.
However, the 800 rpm specimen produced the strongest color contrast and exhibited the fastest and most complete recovery within approximately 5 min, demonstrating the high sensitivity and stable structural integrity of the membrane.
These results establish 800 rpm as the optimal spin-coating condition for the fabrication of reproducible and responsive PtWO3/PDMS colorimetry.
3.2 Colorimetric behavior of PtWO3/PDMS membranes at different H2 concentrations
The overall colorimetric response of the PtWO3/PDMS membrane to various hydrogen concentrations is shown in Fig. 2(a).
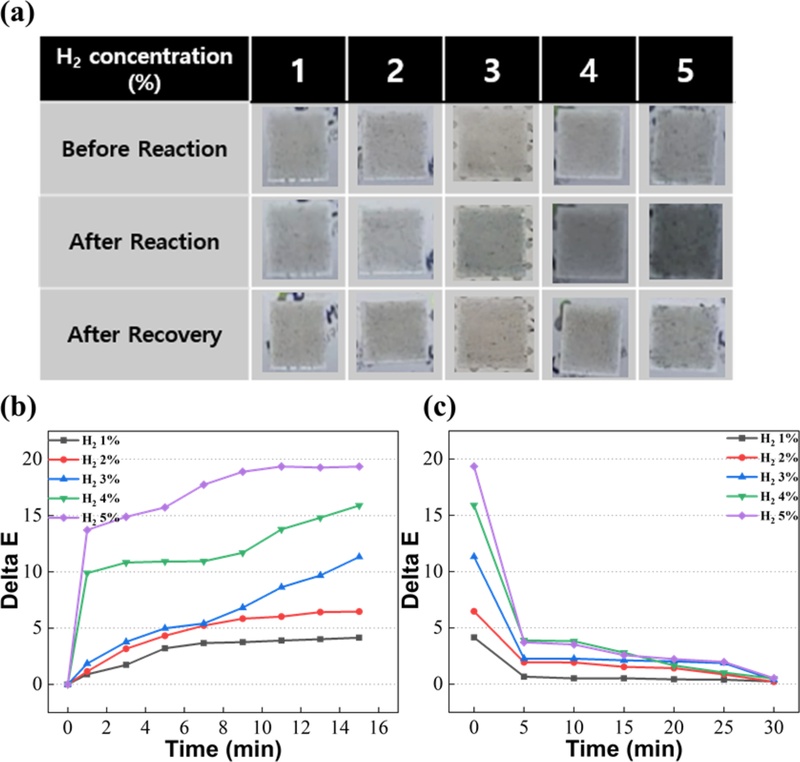
Colorimetric response of the PtWO3/PDMS membrane to different H2 concentrations. (a) Membrane photographs before/after exposure and after recovery at various H2 concentrations (1%–5%), (b) ΔE–time profiles during reaction and (c) recovery, showing rapid, reversible color changes.
When exposed to H2 gas (1%–5%), the initially light-gray surface gradually turned blue, and the coloration intensity increased proportionally with the gas concentration.
A noticeable color transition occurred even at 1% H2, with the film becoming distinctly deep blue at concentrations above 3% H2, thus confirming high optical sensitivity.
The temporal evolution of ΔE during exposure is presented in Fig. 2(b).
A rapid increase in ΔE was observed within the first few minutes, followed by a gradual saturation behavior as equilibrium was reached between H2 adsorption and surface reduction of WO3.
The membrane reached 90% of its final ΔE value within approximately 4 min, demonstrating fast response kinetics.
The maximum ΔE values were approximately 3.2, 7.8, 11.3, 14.2, and 19.7 for 1%, 2%, 3%, 4%, and 5% H2, respectively, indicating a clear concentration-dependent response.
This coloration originates from the catalytic dissociation of H2 on Pt nanoparticles and subsequent proton spillover to the WO3 lattice, leading to the formation of tungsten bronze (HxWO3). The resulting intervalence charge transfer between W5+ and W6+ ions generates the characteristic blue color [20]. This mechanism provides both high sensitivity and rapid optical transition under ambient conditions without requiring external power.
Immediately after gas exposure, the chamber was opened to ambient air to evaluate the recovery performance.
As shown in Figs. 2(a) (bottom row) and 2(c), the blue color gradually fades as the adsorbed hydrogen species desorb and the lattice is re-oxidized.
The sharp ΔE decrease within the first 5 min returns close to its baseline level after approximately 10 min, confirming efficient self-recovery of the sensor under room-temperature air.
The recovery process is attributed to the reoxidation of W⁵⁺ to W⁶⁺ by ambient oxygen a desorption of hydrogen species from the Pt/WO3 interface.
No irreversible color shift or film degradation was observed after repeated exposure–recovery cycles, proving the chemical and structural stability of the PtWO3/PDMS membrane.
This reversible and rapid colorimetric behavior demonstrates the potential of the developed membrane as a power-free, visually readable hydrogen leakage sensor capable of repeated operations under ambient conditions.
3.3 Selectivity against interfering gases
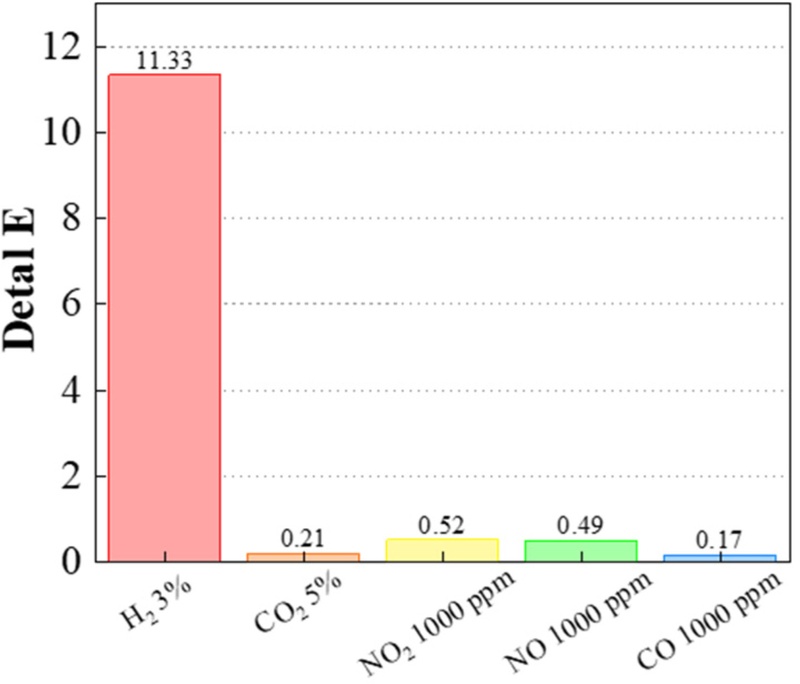
The selectivity of the PtWO3/PDMS membrane toward hydrogen gas was investigated by comparing its colorimetric response to various potentially interfering gases under identical experimental conditions (Fig. 3). The gas tested included CO2 (5%), CO (1000 ppm), NO2 (1000 ppm), and NO (1000 ppm), with all mixtures balanced in nitrogen and supplied by Korea Standard Gas (Republic of Korea).
As shown in Fig. 3, the membrane exhibited a strong optical response only to hydrogen exposure, with negligible color changes from the other gases. The corresponding ΔE values were approximately 11.3 for 3% H2, whereas the other gases—CO2, CO, NO2, and NO—produced ΔE values of only 0.21, 0.52, 0.49, and 0.17, respectively. All of these were far below the perceptible color-difference threshold (ΔE = 2.3), demonstrating excellent selectivity for hydrogen.
This selective response originated from the unique redox chemistry of the PtWO3 composite. Under hydrogen exposure, the Pt nanoparticles catalyze the dissociation of H2 molecules, followed by proton spillover to the WO3 lattice. The resulting reduction process leads to the formation of tungsten-bronze (HxWO3) species, causing the pronounced blue coloration via intervalence charge transfer (W⁶⁺ ↔ W⁵⁺). In contrast, other test gases (CO2, CO, NO, and NO2) neither dissociated effectively from Pt at room temperature nor induced sufficient reduction of WO3, resulting in minimal color variation.
Moreover, the hydrophobic PDMS matrix served as a gas-permeable yet chemically inert host that limited the adsorption of polar or oxidizing species, further enhancing the hydrogen-specific response. The combination of Pt-catalyzed dissociative activation and WO3 optical transition provided a composite film with high selectivity, chemical stability, and repeatable colorimetric performance for hydrogen leak visualization in ambient environments.
4. CONCLUSIONS
A power-free colorimetric hydrogen sensor based on PtWO3 nanoparticles dispersed in a PDMS matrix was successfully fabricated on an alumina substrate using a simple spin-coating process. The effects of the coating parameters and gas concentration on optical performance were systematically investigated. The optimized membrane prepared at 800 rpm exhibited the highest colorimetric contrast, yielding a ΔE value of 11.3 upon exposure to 3% H2, with a clear and rapid color transition from light gray to blue that was easily perceptible by the naked eye (ΔE > 2.3).
The sensor demonstrated a strong concentration-dependent response across the range of 1%–5% H2, reaching saturation within 4 min and recovering its initial color within 5 min under ambient air without an external power supply. Moreover, the selectivity tests confirmed that the PtWO3/PDMS membrane was highly specific to hydrogen, showing negligible responses (ΔE < 1) to other interfering gases, such as CO2, CO, NO2, and NO.
These results highlight the synergistic effect of Pt catalytic activity and WO3 photochromic transition, effectively enabling rapid and reversible hydrogen-induced coloration.
The simple, low-cost fabrication route and power-free operation of the PtWO3/PDMS colorimetric sensor make it a promising candidate for real-time hydrogen leak visualization in safety-critical environments such as fuel cell vehicles, hydrogen storage facilities, and industrial process monitoring.
Acknowledgments
This study has been conducted with the support of the Korea Institute of Industrial Technology (KITECH) as “Development of artificial intelligence-based hydrogen sensor to ensure fuel cell vehicle safety in real driving environments (Kitech UR-25-0005).”
This work was also supported by the Technological Innovation R&D Program (RS-2024-00487867) funded by the Ministry of SMEs and Startups (MSS, Korea).
This work was supported by a KOITA grant funded by the Ministry of Science and ICT (MSIT 1711199734).
This work was supported by a New Faculty Research Grant of Pusan National University, 2025.
REFERENCES
-
G. Korotcenkov, S.-D. Han, J.R. Stetter, Review of electrochemical hydrogen sensors, Chem. Rev. 109 (2009) 1402–1433.
[https://doi.org/10.1021/cr800339k]

-
H. Gu, Z. Wang, Y. Wang, Hydrogen gas sensors based on semiconductor oxide nanostructures, Sensors 12 (2012) 5517–5550.
[https://doi.org/10.3390/s120505517]

-
L. Boon-Brett, J. Bousek, G. Black, P. Moretto, P. Castello, T. Hübert, et al., Identifying performance gaps in hydrogen safety sensor technology for automotive and stationary applications, Int. J. Hydrogen Energy 35 (2010) 373–384.
[https://doi.org/10.1016/j.ijhydene.2009.10.064]

-
L. Li, Z. Wang, Y. Chang, Microscale chemiresistive hydrogen sensors: Current status and recent developments, Nanotechnol. Precis. Eng. 7 (2024) 045002.
[https://doi.org/10.1063/10.0028735]

-
C. Wang, J. Yang, J. Li, C. Luo, X. Xu, F. Qian, Solid-state electrochemical hydrogen sensors: A review, Int. J. Hydrogen Energy 48 (2023) 31377–31391.
[https://doi.org/10.1016/j.ijhydene.2023.04.167]

-
S. Yan, Y. Cao, Y. Su, B. Huang, C. Chen, X. Yu, et al., Hydrogen sensors based on Pd-based materials: A review, Sensors 25 (2025) 3402.
[https://doi.org/10.3390/s25113402]

-
I. Darmadi, A. Nugroho, A.B. Dahlin, High-performance nanostructured palladium-based hydrogen sensors—current limitations and strategies for their mitigation, ACS Sens. 5 (2020) 3306–3327.
[https://doi.org/10.1021/acssensors.0c02019]

-
A. Mirzaei, J.-H. Kim, H.-W. Kim, S.-S. Kim, Gasochromic WO₃ nanostructures for the detection of hydrogen gas: an overview, Appl. Sci. 9 (2019) 1775.
[https://doi.org/10.3390/app9091775]

-
S.H. Cho, J.M. Suh, B. Jeong, T.H. Lee, K.S. Choi, T.H. Eom, et al., Fast responding and highly reversible gasochromic H₂ sensors using Pd–aWO₃ film, Chem. Eng. J. 446 (2022) 136862.
[https://doi.org/10.1016/j.cej.2022.136862]

-
S.-H. Hua, T.-T. Bui, D.-C. Nguyen, Y.-B. Cho, H. Chun, Y.-S. Kim, Enhanced colorimetric detection of hydrogen using PdO-decorated ZnO covered with a metal–organic framework membrane, Int. J. Hydrogen Energy 47 (2022) 39687–39699.
[https://doi.org/10.1016/j.ijhydene.2022.09.117]

-
H. Liu, Z. Yang, L. Wang, P. Yu, Z. Kang, Q. Wu, et al., Gasochromic hydrogen sensors: Fundamentals, recent advances, and perspectives, Sens. Mater. 35 (2023) 41–64.
[https://doi.org/10.18494/SAM4244]

-
S.H. Cho, J.M. Suh, B. Jeong, T.H. Lee, K.S. Choi, T.H. Eom, et al., Substantially accelerated response and recovery in Pd-decorated WO₃ nanorods gasochromic hydrogen sensor, Small 20 (2024) 2309744.
[https://doi.org/10.1002/smll.202309744]

-
X. Pan, D. Wu, Q. Xie, X. Zang, C. Zhang, M. Hua, et al., WO₃ gasochromic materials for hydrogen leakage detection: Synthesis, properties, and applications, Chem. Eng. Technol. 48 (2025) e70005.
[https://doi.org/10.1002/ceat.70005]

-
C. Gao, X. Guo, L. Nie, X. Wu, L. Peng, J. Chen, A review on WO₃ gasochromic film: Mechanism, preparation and properties, Int. J. Hydrogen Energy 48 (2023) 2442–2465.
[https://doi.org/10.1016/j.ijhydene.2022.10.100]

-
S.H. Cho, J.M. Suh, T.H. Eom, T. Kim, H.W. Jang, Colorimetric sensors for toxic and hazardous gas detection: A review, Electron. Mater. Lett. 17 (2021) 1–17.
[https://doi.org/10.1007/s13391-020-00254-9]

-
C. Kim, J.-W. Yoon, Colorimetric sensor based on Pd–MoO₃ nanowires for hydrogen gas leak detection, J. Sens. Sci. Technol. 33 (2024) 259–264.
[https://doi.org/10.46670/JSST.2024.33.5.259]

-
D.L. MacAdam, Visual sensitivities to color differences in daylight, J. Opt. Soc. Am. 32 (1942) 247–274.
[https://doi.org/10.1364/JOSA.32.000247]

-
W.J. Buttner, M.B. Post, R. Burgess, C. Rivkin, An overview of hydrogen safety sensors and requirements, Int. J. Hydrogen Energy 36 (2011) 2462–2470.
[https://doi.org/10.1016/j.ijhydene.2010.04.176]

-
A.H. Anwer, M. Saadaoui, A.T. Mohamed, N. Ahmad, A. Benamor, State-of-the-art advances and challenges in wearable gas sensors for emerging applications: Innovations and future prospects, Chem. Eng. J. 502 (2024) 157899.
[https://doi.org/10.1016/j.cej.2024.157899]

-
S. Matsuura, N. Yamasaku, Y. Nishijima, S. Okazaki, T. Arakawa, Characteristics of highly sensitive hydrogen sensor based on Pt–WO₃/Si microring resonator, Sensors 20 (2020) 96.
[https://doi.org/10.3390/s20010096]