Physiological Pressure Sensing in Bio-Integrated Systems
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License(https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Pressure is a crucial mechanical variable that is related to physiological processes, including perfusion, organ function, and tissue condition throughout the human body. Since pressures occur in various anatomical environments, including gas-filled alveoli, fluid-filled vascular spaces, soft tissues, and skin-contact areas, a single sensing method cannot satisfy all monitoring needs. This review provides a classification framework for physiological pressures based on their transmission media and interface depth, highlighting how the physical environment influences sensor design, accessibility, and operational limitations. We examine recent advances in soft, skin-interfaced sensors that enable noninvasive monitoring of vascular pressure and skin loading, as well as implantable and invasive platforms that directly measure pressure within cardiovascular and musculoskeletal compartments. These emerging systems utilize soft, biocompatible materials, miniaturized structures, and wireless communication to facilitate continuous monitoring of pressure and related physiological parameters. By integrating insights across organ systems, sensing modalities, and mechanical environments, this review outlines key design considerations and future opportunities for pressure-monitoring technologies that support early diagnosis, personalized medicine, and long-term health management.
Keywords:
Pressure sensors, Wireless biosensors, Biocompatibility, Health monitoring, Real-time sensing1. INTRODUCTION
Pressure is a crucial mechanical signal in human physiology, essential for functions such as cerebral perfusion, blood circulation, respiration, organ protection, and tissue health. Deviations from normal pressure levels can lead to or exacerbate severe conditions—including intracranial hypertension [1-8], respiratory issues [6,9], cardiovascular problems [10-13], gastroesophageal reflux [14,15], portal hypertension [16,17], pressure ulcers [18-21], and acute compartment syndrome [22]—making accurate pressure measurement vital for diagnosis, treatment, and prognosis. Despite its importance, monitoring physiological pressure remains challenging because signals originate from diverse sources, including fluid-filled deep organs and superficial tissues at the skin surface.
These complexities pose significant challenges for sensing technologies. No single sensor design can meet the requirements of diverse biological environments, where pressure is transmitted through gas, liquid, solid, or composite media and may originate within luminal, extraluminal, or surface-level compartments. The need for environment-specific sensing approaches necessitates a systematic understanding of how physiological pressures are generated, regulated, and transmitted across different organ systems.
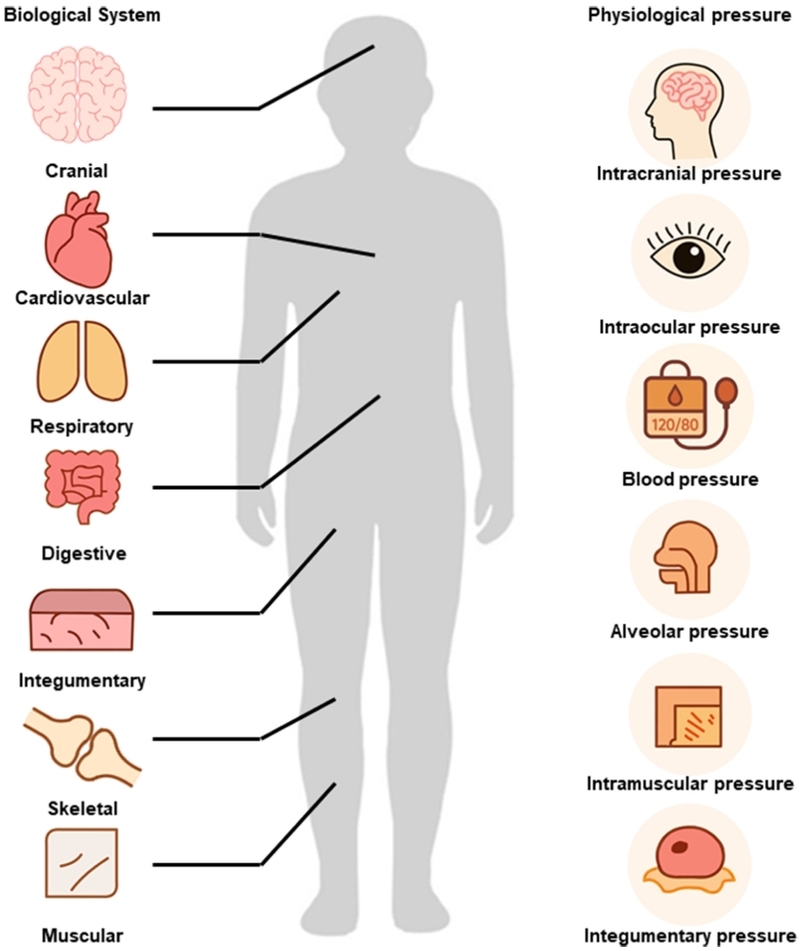
To address this need, Section 2 summarizes key physiological pressures across the cranial, cardiovascular, respiratory, gastrointestinal, hepatic, integumentary, musculoskeletal, and ocular systems. Existing classification approaches emphasize material properties such as elastic modulus, strain range, and sensing mechanisms, but offer a limited perspective on how physiological pressures vary with anatomical depth and interface conditions [23]. We organize these pressures using a classification framework that considers transmission media and interface depth, providing a foundation for linking biological constraints to sensor design principles. This framework also highlights the interconnected mechanics of vascular, muscular, and cutaneous tissues, which necessitate multimodal or multiscale sensing approaches to capture clinically meaningful signals.
Sections 3 and 4 review recent technologies for detecting pressure originating from various locations. Soft, skin-interfaced pressure sensors consist of soft, biocompatible materials, thin, flexible structures, and wireless communication circuitry to monitor continuously in real time. These systems allow continuous tracking of superficial hemodynamics and skin loading, supporting applications such as cuffless blood pressure measurement and pressure ulcer prevention [24]. Conversely, invasive and implantable sensors are required for deep-tissue environments, where pressure changes cannot be accessed noninvasively. Miniaturized intravascular devices provide real-time hemodynamic monitoring within the heart and major vessels, while flexible multimodal probes enable simultaneous pressure, perfusion, and oxygenation measurements within muscle compartments.
Together, these advances demonstrate a broader shift toward continuous, multimodal pressure sensing across various body depths. By merging innovations in materials science, mechanics, microfabrication, and wireless communication, the potential for physiological monitoring expands, paving the way for early disease detection and personalized healthcare. The following sections provide a detailed overview of these physiological environments, sensor design strategies, and technological platforms, concluding with a discussion on future directions in biological pressure sensing.
2. PRESSURE SENSING IN BIOLOGICAL SYSTEMS
This section outlines considerations for designing pressure sensors for major organ systems with various mechanical characteristics. Physiological pressures arise from fluid flow, tissue deformation, organ motion, and external loading. Because each system exhibits a distinct combination of geometry, material composition, and mechanical constraints, understanding the underlying pressure environment is essential for developing optimal sensing strategies. Fig. 1 summarizes representative physiological pressure ranges and their associated biological functions.
2.1. Physiological pressures in biological systems.
Physiological pressures support essential biological processes—maintaining perfusion, regulating ventilation, sustaining digestive transport, stabilizing intracranial structures, and preserving musculoskeletal integrity [3,5,6]. Their magnitudes and mechanical behavior vary widely depending on anatomical location and the surrounding transmission medium. The following subsections highlight key pressure environments and emphasize features most relevant to sensor design.
Intracranial pressure (ICP) arises within a confined, fluid–solid composite environment consisting of cerebrospinal fluid (CSF), brain tissue, and the rigid skull [1,2,4,7,8]. Because the cranial cavity offers minimal compliance, small changes in CSF volume or cerebral blood flow produce nonlinear pressure responses [2,7,8]. Standard ICP typically ranges from 7–15 mmHg in adults, with values above ~20 mmHg requiring clinical intervention. Elevation beyond ~22 mmHg sustained for several minutes reflects refractory intracranial hypertension [1,2,7,8]. Conversely, abnormally low ICP (<5 mmHg) may indicate CSF leakage or intracranial hypotension. This closed compartment's low compliance and layered structure necessitate invasive access [1,2] or biomechanics-based indirect measurements [2].
Arterial blood pressure reflects pulsatile forces generated by ventricular contraction and relaxation [25]. Typical systolic and diastolic pressures range from 90–120 mmHg and 60–80 mmHg, respectively, with deviations indicating systemic hemodynamic imbalance [11]. Because arterial lumens are fluid-filled, cylindrical, and dynamically deforming, pressure propagates rapidly along the vascular tree, influencing perfusion across all organs. Accurate waveform reconstruction requires direct intraluminal access [11] or calibrated surface-based methods capable of capturing subtle pulse-induced deformations [11].
Breathing mechanics rely on pressure differences between the thoracic cavity, pleural space, and alveoli [6,9]. Inspiration creates negative intrapleural pressure that pulls air into the lungs [5], while expiration reverses this process through elastic recoil [6,9]. Disruptions in these pressure patterns—such as excessive negative pressure during hyperventilation—can affect gas exchange efficiency and acid–base balance [9]. Since direct measurements are limited in the gas-filled alveolar environment without tissue disruption, clinical assessments use surrogate measurements (e.g., airway pressure, pulmonary wedge pressure) that reflect alveolar mechanics.
Gastric and esophageal pressures control food movement and barrier function. Loss of normal lower esophageal sphincter (LES) pressure (~10–30 mmHg) due to gastric distension can cause reflux [15]. In the liver, portal pressure, measured by the hepatic venous pressure gradient (HVPG), is typically below 5 mmHg. When it exceeds 10 mmHg, it signals clinically significant portal hypertension and is linked to complications like variceal formation [16,17]. Sensing luminal pressures in soft, deformable organs is influenced by peristalsis, fluid load, and body position, requiring sensors that can withstand dynamic, compressive, and chemical conditions.
At the skin interface, external forces compress microvascular networks and soft tissues [18-21]. Sustained pressure above ~32 mmHg for ≥2 hours can lead to ischemia and ulcer formation, particularly at bony prominences [18-21]. Because superficial hemodynamics and tissue deformation occur within millimeters of the surface, these signals propagate effectively to wearable sensors. In this case, sensors must maintain conformal contact, minimize perturbation of the intrinsic pressure field, and remain reliable under high normal and shear loads.
Muscle compartments in the limbs are enclosed by relatively inelastic fascia. Intramuscular pressure typically remains below ~10 mmHg but rises markedly with swelling, trauma, or ischemia [22,26]. Pressures above ~30 mmHg raise concern for acute compartment syndrome, a rapidly evolving condition that compromises perfusion [22,26]. Given the need for pressure sensing deep within thick, heterogeneous soft tissues, surface measurements are insufficient and require invasive access for accurate assessment.
Intraocular pressure (IOP) reflects fluid dynamics within the anterior chamber and must remain within a narrow physiological range to protect retinal structure and function [26]. Corneal deformation under external load is commonly used to infer internal pressure. Sensing requires highly compliant interfaces that quantify minute curvature changes without disturbing ocular biomechanics [27].
2.2. Design of sensors across biological systems
Biological pressures interact with tissues through gas-, liquid-, solid-, or composite-mediated transmission, each imposing distinct constraints on sensor architecture, material selection, and measurement strategy. Fig. 2 presents a classification framework based on transmission medium and interface depth, connecting physiological mechanisms to engineering requirements.
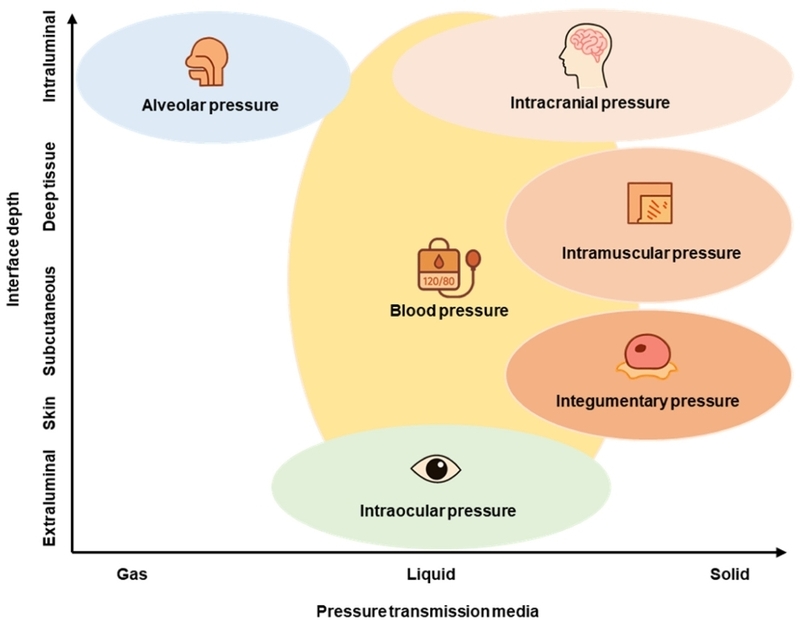
Classification of physiological pressure related to the pressure transmission media and the interface depth.
In gas-filled spaces such as the alveoli, direct access is infeasible due to tissue fragility. Consequently, sensing relies on indirect indicators—such as airway or pulmonary wedge pressure—that reflect downstream mechanics rather than the local pressure itself [5]. Sensors must interface with upstream or downstream structures and incorporate biomechanical modeling to reconstruct alveolar dynamics.
Intravascular and intraocular systems transmit pressure efficiently through incompressible fluids. Sensing strategies must account for rapid propagation, pulsatility, and fluid–wall interactions. Intraocular measurements, for example, use corneal deformation as a mechanical proxy for internal pressure. Design form factors must permit minimally invasive deployment, withstand cyclic loading, and capture transient waveforms with high fidelity.
Soft tissues—including muscle, dermis, and fascia—form composite mechanical domains where pressure arises from swelling, external loading, or fluid shifts. In the intracranial and musculoskeletal systems, solid matrices constrain pressure propagation, producing local, nonlinear responses dependent on tissue stiffness and geometry. Deep-tissue environments require invasive sensors capable of operating within confined, mechanically heterogeneous compartments.
Although vascular, intramuscular, and integumentary pressures stem from different physiological processes, they form a mechanical continuum across tissue layers. Blood pressure causes vessel-wall expansion, intramuscular pressure indicates swelling-induced tension within fascia, and integumentary pressure results from external compression that gradually influences deeper tissues. Understanding this continuum necessitates combining pressure with perfusion, deformation, or oxygenation signals to detect clinically significant patterns. Considering them together enables a more integrated understanding of how pressure manifests across tissue layers and how these environments impose different requirements on sensor design. This framework provides a basis for discussing pressure-sensing platforms that operate across multiple anatomical depths—from soft, skin-interfaced systems for surface loading to miniaturized implantable devices for deep-tissue monitoring.
3. SKIN-INTERFACED PRESSURE MONITORING SYSTEM
Pressures at the body surface, including those arising from superficial hemodynamics and external mechanical loading, serve as clinically meaningful signals that can be captured through soft, skin-interfaced sensors [25,28-30]. Unlike intravascular or deep-tissue pressures—which require catheter-based or implantable devices—surface and near-surface pressures propagate with minimal attenuation through soft tissue and can therefore be detected reliably using thin, flexible platforms [31]. These skin-interfaced systems facilitate continuous, non-invasive monitoring of mechanical stress, pulse-induced skin deformation, and other cardio-mechanical signatures in both daily and clinical environments [25,28-33]. While skin-interfaced pressure sensors enable continuous, non-invasive monitoring, long-term epidermal contact requires careful consideration of safety [34,35], particularly adhesive-induced skin irritation and localized heat accumulation in wireless or powered systems. These risks can be mitigated through ultrathin, low-modulus device designs combined with skin-friendly [36] or switchable adhesives [36,37] and thermal management strategies [38,39] that enhance biocompatibility and long-term wearability.
3.1 Vascular pressure monitoring sensor
Recent soft 3D piezoresistive pressure sensors—particularly those designed with buckled table-like microarchitectures and complete Wheatstone bridge configurations—demonstrate substantial improvements in sensitivity by amplifying strain in both tensile and compressive directions (Fig. 3(a)) [25]. Such microstructures increase the gauge factor, while the full-bridge layout enhances the output signal by up to fourfold and stabilizes linearity across physiologic pressure ranges (R² ≈ 0.995). Furthermore, multilayer ultrathin PI/metal stacks position the metal gauge far from the neutral plane, magnifying strain even under low pressure and enabling high sensitivity in the sub-kPa regime relevant to blood-flow monitoring.
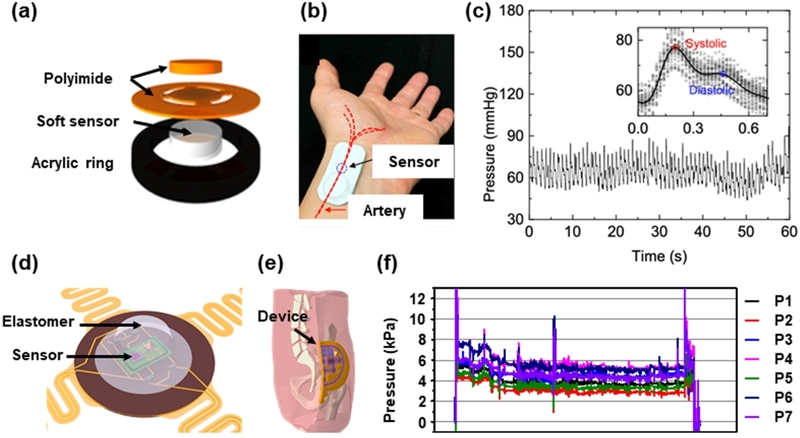
Skin-interfaced pressure monitoring system. (a-c) Skin-interfaced vascular pressure monitoring at the wrist artery. Adapted from Ref. [25]. (d-f) Skin-interfaced integumentary pressure monitoring at the sacral area. Adapted from Ref. [21].
To satisfy these requirements, wearable pressure sensors increasingly employ material and structural strategies that stabilize performance during movement. Serpentine polyimide layouts help redistribute shear and rotational forces away from the sensing core, reducing motion-induced artifacts. In addition, ultrathin, low-modulus elastomers (≈ 60–100 kPa) match the mechanical properties of skin, allowing the device to follow natural deformations without micro-slip or delamination. These features enhance contact stability and facilitate continuous waveform acquisition even during daily activity. Soft wireless systems also minimize inertial effects by incorporating compact BLE modules and lightweight power architectures, reducing noise during movement (Fig. 3(b)) [25].
Blood pressure estimation from skin-interfaced measurements relies on transforming surface pressure waveforms into hemodynamic equivalents. A calibration model maps the superficial pressure signal to the underlying arterial pressure, accounting for mechanical coupling between the sensor, the skin, and the vessel wall (Fig. 3(c)) [25]. Reconstructed waveforms enable the extraction of systolic and diastolic features through peak-detection algorithms. This computational pipeline—calibration, waveform reconstruction, and systolic/diastolic extraction—underpins many recent approaches for cuffless, continuous blood pressure monitoring.
3.2 Integumentary pressure monitoring sensor
Integumentary pressures occur when external mechanical forces—such as posture, recumbency, medical devices, or everyday activities—are transferred to the skin and underlying soft tissues. Continuous monitoring of these pressures is crucial because sustained loads on bony prominences can decrease blood flow and cause tissue damage, leading to pressure ulcers and other complications [18,40-42]. Monitoring such pressures requires sensors that remain durable under high normal loads, maintain mechanical strength during repeated deformation, and operate reliably across different parts of the body.
Recent systems embed barometric MEMS sensors within soft silicone elastomer structures to enhance durability and ensure dependable operation under high normal loads (Fig. 3(d)) [21]. Elastomer deformation distributes stress evenly to the MEMS diaphragm, as confirmed by FEA, leading to low device-to-device variability, drift within ±2%, and mechanical endurance over thousands of cycles. Shear perturbations generally cause less than 5% error. This robustness is essential, as integumentary environments involve large normal forces and complex loading conditions during movement or repositioning.
Accurate measurement of skin-interface pressure across diverse anatomical curves requires that the sensor does not distort the underlying pressure field. To address this, systems utilize soft multilayer cushions, modulus-engineered foams, or stretchable frames (Fig. 3(e)) [21]. These features maintain sensing accuracy by distributing the load uniformly and preserving the device–skin coupling. FEA optimization confirms that these interfaces minimize perturbations to the intrinsic pressure fields, ensuring an accurate estimation of true tissue loading. Empirical measurements across multiple sensing nodes show negligible differences in pressure distributions between measurements with and without the device in place, confirming minimal disruption to the natural load profile.
Multi-node soft sensor arrays enable real-time mapping of pressure distributions across multiple anatomical sites (Fig. 3(f)) [21]. Nodes located at weight-bearing regions register consistently higher loads than those in lightly loaded areas, reflecting natural redistribution patterns associated with posture, micro-movements, and repositioning. Stable long-term baselines with minimal drift support extended monitoring periods, providing clinically actionable insights into ulcer risk and load management strategies.
4. INVASIVE PRESSURE MONITORING SYSTEM
Pressures that develop within deep anatomical compartments—such as intravascular spaces, cardiac chambers, and muscle compartments—play essential roles in regulating perfusion, organ function, and tissue health [17]. Unlike surface or near-surface pressures, which can be measured with skin-interfaced sensors with minimal signal loss, these internal pressures are situated within mechanically complex, fluid-filled, or tightly confined environments. Consequently, accurately assessing these pressures requires sensing systems capable of operating in regions inaccessible to noninvasive methods. Traditional catheter-based measurements offer high accuracy but are limited by their temporary nature, procedural invasiveness, and restrictions on wired connections [10]. These limitations have driven the development of implantable platforms that enable continuous, real-time monitoring of pressure and related physiological parameters within deep tissues [10,43]. Such systems expand pressure sensing from surface biomechanics to internal organ function, supporting early diagnosis, improved clinical decision-making, and new opportunities for long-term physiological monitoring.
4.1 Intravascular pressure monitoring sensor
Pressures generated within the cardiac chambers and major blood vessels are fundamental to cardiovascular function, directly governing perfusion, ventricular workload, and systemic circulation. Because these pressures originate deep within fluid-filled lumens and undergo rapid cyclical variations, they cannot be captured through surface-based sensing methods. Conventional assessments rely on wired catheter systems, which provide accurate waveforms but are restricted to short-term measurements, introduce procedural risks, and limit patient mobility due to their tethered configuration. These constraints have motivated the development of miniaturized, wirelessly powered implants that can operate directly within the intravascular environment and provide continuous access to hemodynamic information [10].
Recent device architectures address these requirements through compact multimodal platforms that measure pressure, flow, and temperature within a single integrated system (Fig. 4(a)) [10]. Their millimeter-scale dimensions facilitate delivery via minimally invasive catheters and seamless integration into existing cardiovascular devices, such as stents or valve prostheses. By anchoring the sensor within the luminal space or combining it with an expandable metallic scaffold, these systems maintain stable positioning even under pulsatile flow conditions (Fig. 4(b)) [10].
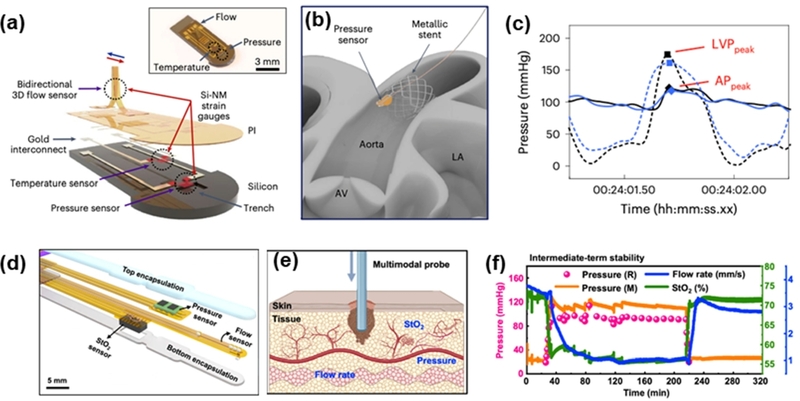
Invasive pressure monitoring system. (a-c) Implantable intravascular pressure monitoring inside the cardiac chamber. Adapted from Ref. [10]. (d-f) Invasive intramuscular pressure monitoring within the muscle compartment. Adapted from Ref. [22].
Once implanted, wireless devices can capture high-fidelity left ventricular and aortic pressure waveforms, enabling direct comparison with clinical catheter-based measurements (Fig. 4(c)) [10]. This capability is particularly valuable for quantifying transvalvular pressure gradients, which are essential diagnostic indicators of conditions such as aortic stenosis. Progressive narrowing of the valve increases flow resistance and, correspondingly, elevates the pressure difference between the ventricle and the aorta. Continuous monitoring of these gradients—both peak and mean—provides quantitative insight into disease severity and offers a platform for real-time evaluation of therapeutic interventions.
4.2 Intramuscular pressure monitoring sensor
Intramuscular pressure reflects the mechanical state of muscle compartments, where increased tissue swelling or fluid accumulation can restrict perfusion and threaten tissue viability. Accurate and continuous assessment of intramuscular pressure is critical in acute compartment syndrome, a condition in which elevated pressure within the fascial boundary can rapidly lead to ischemia and irreversible muscle damage. Because these pressures develop deep within solid tissues and evolve dynamically with movement, hydration, and perfusion, they cannot be reliably captured with skin-surface sensors. Conventional needle-based measurements provide only intermittent snapshots, are highly operator-dependent, and cannot characterize evolving pathophysiology over time [22,26].
These limitations have motivated the development of minimally invasive, multimodal probes engineered to measure pressure alongside perfusion-related parameters within the muscle compartment itself [22]. Thin, flexible substrates incorporating pressure, oxygen saturation (StO2), and flow sensors enable simultaneous characterization of mechanical and hemodynamic responses at a single anatomical location (Fig. 4(d)) [22]. The soft probe geometry facilitates insertion through a small incision while minimizing disruption to surrounding tissues, allowing each sensing element to interface directly with muscle or microvascular structures.
Following implantation, the probe conforms to the fascial compartment, where the tightly coupled relationships among pressure, blood flow, and tissue oxygenation can be observed in real time (Fig. 4(e)) [22]. Initial changes in the pressure trace typically reflect tissue relaxation and probe stabilization, after which the signals converge to a steady operating baseline. As compartment pressure rises, reductions in perfusion and declines in StO2 occur in parallel, reflecting the physiological cascade underlying ischemic injury. This multimodal sensing capability provides diagnostic information that cannot be obtained from pressure alone, thereby enhancing early detection capabilities for pathological progression.
Long-term recordings demonstrate that these probes maintain stable performance over extended periods, with minimal drift across pressure, flow, and oxygenation channels even under repeated loading or movement (Fig. 4(f)) [22]. Such stability is critical for monitoring conditions that evolve gradually and require continuous surveillance rather than episodic measurement. Collectively, minimally invasive multimodal probes represent a promising direction for deep-tissue pressure sensing, enabling early diagnosis and improved physiological interpretation in scenarios such as compartment syndrome, postoperative recovery, and rehabilitative monitoring.
5. CONCLUSIONS
Physiological pressure arises across multiple anatomical depths and material environments, spanning gas-filled alveoli, fluid-driven cardiovascular lumens, deformable soft tissues, and skin–interface contact regions. These diverse pressure conditions play essential roles in regulating perfusion, organ mechanics, and tissue health, underscoring the importance of sensing technologies that can accurately capture the mechanical states of biological systems. The classification framework presented in this review—based on pressure-transmission media and interface depth—highlights the breadth of design constraints that emerge when translating fundamental physiological requirements into sensing strategies.
Advances in soft and biocompatible materials, miniaturized structures, and low-power, wireless electronics have enabled the development of skin-interfaced pressure sensors capable of capturing near-surface mechanical signals, including blood-flow-related dynamics and externally applied loads. These wearable systems offer noninvasive, continuous monitoring suitable for daily activity and clinical environments, providing early indicators of hemodynamic instability or pressure-induced tissue injury.
Complementary advances in implantable and invasive technologies have extended pressure sensing to deep physiological compartments inaccessible via surface-based approaches. Miniaturized intravascular platforms enable real-time measurement of cardiac and vascular pressures, while flexible multimodal probes allow integrated assessments of pressure, perfusion, and oxygenation within muscle compartments. Together, these systems provide insights into internal physiological processes with spatial and temporal precision that surpasses traditional catheter-based or intermittent measurement techniques.
Across both wearable and invasive modalities, a unifying theme is the shift toward continuous, wireless, and accurate pressure monitoring, supported by advances in materials, mechanics, wireless communication, and computational analysis. In parallel, emerging AI- and data-driven approaches are expected to play an increasingly important role in extracting clinically meaningful information from continuous pressure data, enabling improved signal interpretation, personalized risk assessment, and long-term monitoring across both wearable and implantable systems. Future research will require further integration of pressure sensing with complementary physiological signals, improved long-term biocompatibility, and personalized calibration frameworks that account for anatomical and mechanical variability across users. These directions will be essential for transitioning pressure monitoring from discrete clinical assessments to pervasive, real-time physiological surveillance that can inform diagnosis, guide therapy, and prevent disease progression.
At the same time, each pressure-sensing strategy involves inherent trade-offs that extend beyond demonstrated device performance. Skin-interfaced systems are susceptible to motion-induced artifacts, long-term drift, and calibration dependence, whereas invasive platforms introduce implantation-related risks, biocompatibility concerns, and constraints on long-term stability and deployment. Although the pressure-sensing systems discussed in this review largely remain at the proof-of-concept stage, they are designed around clinically relevant physiological targets and thus hold substantial potential for future clinical and commercial translation.
Acknowledgments
S.Y. acknowledges support from the National Research Foundation of Korea (NRF) grant (No. RS-2025-23525124), funded by the Korea government (MSIT), and the BK21 FOUR program (Digital Anti-aging Convergence Research Group, Inje University). This research was supported by the University Innovation Support Project Research Fund of Hoseo University in 2024 (2024-0015-01). This work was partly supported by the Institute of Information & Communications Technology Planning & Evaluation(IITP)-Innovative Human Resource Development for Local Intellectualization program grant funded by the Korea government (MSIT)(IITP-2025-RS-2024-00436765).
REFERENCES
-
G. Cucciolini, V. Motroni, M. Czosnyka, Intracranial pressure for clinicians: it is not just a number, J. Anesth. Analg. Crit. Care 3 (2023) 31.
[https://doi.org/10.1186/s44158-023-00115-5]

-
M. Harary, R.G.F. Dolmans, W.B. Gormley, Intracranial Pressure Monitoring-Review and Avenues for Development, Sensors 18 (2018) 465.
[https://doi.org/10.3390/s18020465]

-
V. Kukreti, H. Mohseni-Bod, J. Drake, Management of raised intracranial pressure in children with traumatic brain injury, J. Pediatr. Neurosci. 9 (2014) 207–215.
[https://doi.org/10.4103/1817-1745.147572]

- J. Robblee, K.A. Secora, L.M. Alhilali, K.L. Knievel, Spontaneous intracranial hypotension, Pract. Neurol. (2020) 41-52.
-
J. van Egmond, L.H. Booij, The role of pleural pressure in inducing pneumothorax and other adverse effects of positive pressure ventilation, J. Thorac. Dis. 16 (2024) 8103–8109.
[https://doi.org/10.21037/jtd-24-497]

-
S.H. Loring, G.P. Topulos, R.D. Hubmayr, Transpulmonary Pressure: The Importance of Precise Definitions and Limiting Assumptions, Am. J. Respir. Crit. Care Med. 194 (2016) 1452–1457.
[https://doi.org/10.1164/rccm.201512-2448CP]

-
J. Ragland, K. Lee, Critical Care Management and Monitoring of Intracranial Pressure, J. Neurocrit. Care 9 (2016) 105–112.
[https://doi.org/10.18700/jnc.160101]

- S.-B. Ko, Intracranial Pressure and Hemodynamics, J. Neurocrit. Care 4 (2011) 35–41.
-
M. Jozwiak, J.L. Teboul, Heart-Lungs interactions:the basics and clinical implications, Ann. Intensive Care 14 (2024) 122.
[https://doi.org/10.1186/s13613-024-01356-5]

-
K. Kwon, J.U. Kim, S.M. Won, J. Zhao, R. Avila, H. Wang, et al., A battery-less wireless implant for the continuous monitoring of vascular pressure, flow rate and temperature, Nat. Biomed. Eng. 7 (2023) 1215–1228.
[https://doi.org/10.1038/s41551-023-01022-4]

- G. Mancia, R. Kreutz, M. Brunström, M. Burnier, G. Grassi, A. Januszewicz, et al., 2023 ESH Guidelines for the management of arterial hypertension, J. Hypertens. 41 (2023) 1874–2071.
-
W.-J. Kim, Y.-S. Cho, H.-J. Kang, S.-Y. Choi, Development of miniature weight sensor using piezoresistive pressure sensor, J. Sens. Sci. Technol. 14 (2005) 237–243.
[https://doi.org/10.5369/JSST.2005.14.4.237]

-
B.-L. Lee, J.-W. Jung, K.-J. Chun, A Cantilever Type Contact Force Sensor Array for Blood Pressure Measurement, J. Sens. Sci. Technol. 21 (2012) 121–126.
[https://doi.org/10.5369/JSST.2012.21.2.121]

-
J.I. Bucan, T. Braut, A. Krsek, V. Sotosek, L. Baticic, Updates in Gastroesophageal Reflux Disease Management: From Proton Pump Inhibitors to Dietary and Lifestyle Modifications, Gastrointest. Disord. 7 (2025) 33.
[https://doi.org/10.3390/gidisord7020033]

-
L. Bertin, V. Savarino, E. Marabotto, M. Ghisaa, N. de Bortoli, E.V. Savarino, Pathophysiology of Gastroesophageal Reflux Disease, Digestion (2025) 1–17.
[https://doi.org/10.1159/000547023]

- E.K. Mitten, P. Portincasa, G. Baffy, Portal Hypertension in Nonalcoholic Fatty Liver Disease: Challenges and Paradigms, J. Clin. Transl. Hepatol. 11 (2023) 1201–1211.
-
X.-Y. Xie, A. Benmassaound, Advances in the diagnosis and management of clinically significant portal hypertension in cirrhosis: A narrative review, World J. Hepatol. 17 (2025) 104761.
[https://doi.org/10.4254/wjh.v17.i6.104761]

-
Wound/Pressure Ulcer/Burn Guidelines Drafting Committee (Pressure Ulcer Group), H. Fujiwara, R. Irisawa, M. Otsuka, T. Kako, T. Kaji, et al., Wound, Pressure Ulcer, and Burn Guidelines (2023)-2:Guidelines for the Diagnosis and Treatment of Pressure Ulcers, Third Edition, J. Dermatol. 52 (2025) e744–e794.
[https://doi.org/10.1111/1346-8138.17758]

-
T.-W. Cheng, Y.-N. Lin, S.-S. Lee, Y.-R. Kuo, The niche of dermal graft to reconstruct a complex pressure injury wound in sacral region: A case report, Medicine 102 (2023) e36617.
[https://doi.org/10.1097/MD.0000000000036617]

-
M. Choi, S. Lee, S.W. Kim, J.Y. Lee, Y. Kim, J. Kim, et al, Battery-Free, Wireless Multi-Sensing Platform for Comprehensive Management of Pressure Injury and Hygiene, Adv. Funct. Mater. 35 (2025) e06774.
[https://doi.org/10.1002/adfm.202506774]

-
S. Yoo, Z. Lv, N. Fadell, J.-Y. Yoo, S. Oh, K.-H. Ha, et al., A wireless, skin-integrated system for continuous pressure distribution monitoring to prevent ulcers across various healthcare environments, npj Flex. Electron. 10 (2026) 1.
[https://doi.org/10.1038/s41528-025-00501-9]

-
S.G. Seo, S. Kim, S. Yoo, S. Oh, H. Luan, Z. Lv, et al., Minimally Invasive, Bioadaptive Multimodal Sensor Probe with Safe Deployment for Real-Time Acute Compartment Syndrome Diagnosis, Adv. Sci. 12 (2025) e06942.
[https://doi.org/10.1002/advs.202506942]

-
X. Li, X. Huang, L. Yang, S. Jung, J. Wang, H. Zhao, Implantable physical sensors for in vivo organ monitoring, Med-X 3 (2025) 1.
[https://doi.org/10.1007/s44258-024-00047-x]

-
X. Peng, K. Dong, C. Ye, Y. Jiang, S. Zhai, R. Cheng, et al., A breathable, biodegradable, antibacterial, and self-powered electronic skin based on all-nanofiber triboelectric nanogenerators, Sci. Adv. 6 (2020) eaba9624.
[https://doi.org/10.1126/sciadv.aba9624]

-
Y. Park, H. Luan, K. Kwon, T. S. Chung, S. Oh, J.-Y. Yoo, et al., Soft, full Wheatstone bridge 3D pressure sensors for cardiovascular monitoring, npj Flex. Electron. 8 (2024) 6.
[https://doi.org/10.1038/s41528-024-00294-3]

-
S. Vogels, E.D. Ritchie, D. de Vries, G.-J. Kleinrensink, M.H. Verhofstad, R. Hoencamp, Applicability of devices available for the measurement of intracompartmental pressures: a cadaver study, J. Exp. Orthop. 9 (2022) 98.
[https://doi.org/10.1186/s40634-022-00529-0]

-
C. Yang, X. Huang, X. Li, C. Yang, T. Zhang, Q. Wu, et al., Wearable and implantable intraocular pressure biosensors: Recent progress and future prospects, Adv. Sci. 8 (2021) 2002971.
[https://doi.org/10.1002/advs.202002971]

-
S. Lakshmanan, S.B. Suseela, R. Sankararajan, Reduced graphene oxide/polyaniline hydrogel-based piezo pressure sensor for biomedical applications, J. Mater. Sci. Mater. Electron. 36 (2025) 1766.
[https://doi.org/10.1007/s10854-025-15819-3]

-
J. Wang, G. Xia, L. Xia, Y. Chen, Q. Li, H. Zeng, et al., HCNT/AgNPs/PVA/PAM hydrogel-based flexible pressure sensor for physiological monitoring, J. Mater. Sci. Mater. Electron. 35 (2024) 1931.
[https://doi.org/10.1007/s10854-024-13689-9]

-
B.-S. Park, S.-M. Im, H. Lee, Y. T. Lee, C. Nam, S. Hong, et al., Visual and tactile perception techniques for braille recognition, Micro Nanosyst. Lett. 11 (2023) 23.
[https://doi.org/10.1186/s40486-023-00191-w]

-
M.-H. Jun, Y.J. Jeon, Y.-M. Kim, Interference Effects on the Thickness of a Pulse Pressure Sensor Array Coated with Silicone, J. Sens. Sci. Technol. 25 (2016) 35–40.
[https://doi.org/10.5369/JSST.2016.25.1.35]

-
M.-H. Jun, Y.J. Jeon, Y.-M. Kim, Signal Change and Compensation of Pulse Pressure Sensor Array Due to Wrist Surface Temperature, J. Sens. Sci. Technol. 26 (2017) 141–147.
[https://doi.org/10.5369/JSST.2017.26.2.141]

-
B.-S. Park, S. Lee, Y.-H. Song, P. Yeon, M.-G. Kim, Flexible multilayered skin health sensing platform for eyewear applications, Micro Nanosyst. Lett. 13 (2025) 6.
[https://doi.org/10.1186/s40486-025-00226-4]

-
J. Kim, S. Yoo, C. Liu, S.S. Kwak, J.R. Walter, S. Xu, et al., Skin-interfaced wireless biosensors for perinatal and paediatric health, Nat. Rev. Bioeng. 1 (2023) 631–647.
[https://doi.org/10.1038/s44222-023-00090-0]

-
D. Cho, R. Li, H. Jeong, S. Li, C. Wu, A. Tzavelis, et al., Bitter Flavored, Soft Composites for Wearables Designed to Reduce Risks of Choking in Infants, Adv. Mater. 33 (2021) 2103857.
[https://doi.org/10.1002/adma.202103857]

-
S.S. Kwak, S. Yoo, R. Avila, H.U. Chung, H. Jeong, C. Liu, et al., Skin-Integrated Devices with Soft, Holey Architectures for Wireless Physiological Monitoring, With Applications in the Neonatal Intensive Care Unit, Adv. Mater. 33 (2021) 2103974.
[https://doi.org/10.1002/adma.202103974]

-
K.R. Jinkins, S. Li, H. Arafa, H. Jeong, Y.J. Lee, C. Wu, et al., Thermally switchable, crystallizable oil and silicone composite adhesives for skin-interfaced wearable devices, Sci. Adv. 8 (2022) eabo0537.
[https://doi.org/10.1126/sciadv.abo0537]

-
C. Liu, J.-T. Kim, D.S. Yang, D. Cho, S. Yoo, S. R. Madhvapathy, et al., Multifunctional Materials Strategies for Enhanced Safety of Wireless, Skin-Interfaced Bioelectronic Devices, Adv. Funct. Mater. 33 (2023) 2302256.
[https://doi.org/10.1002/adfm.202302256]

-
S. Yoo, T. Yang, M. Park, H. Jeong, Y.J. Lee, D. Cho, et al., Responsive materials and mechanisms as thermal safety systems for skin-interfaced electronic devices, Nat. Commun. 14 (2023) 1024.
[https://doi.org/10.1038/s41467-023-36690-y]

-
T.F. Ni, J.-L. Wang, C.-K. Chen, D.F. Shih, J. Wang, Can a prolonged healing pressure injury be benefited by using an AI mattress? A case study, BMC Geriatr. 24 (2024) 307.
[https://doi.org/10.1186/s12877-024-04900-x]

-
M. Yang, J. Liu, D. Liu, J. Jiao, N. Cui, S. Liu, et al., A fully self-healing piezoelectric nanogenerator for self-powered pressure sensing electronic skin, Research 2021 (2021) 9793458.
[https://doi.org/10.34133/2021/9793458]

-
D. Oh, J. Seo, H. G. Kim, C. Ryu, S.-W. Bang, S. Park, et al., Multi-height micropyramids based pressure sensor with tunable sensing properties for robotics and step tracking applications, Micro Nanosyst. Lett. 10 (2022) 7.
[https://doi.org/10.1186/s40486-022-00149-4]

-
J. Shang, X. Ma, P. prototype, C. Huang, Z. Lao, J. Wang, et al., A flexible catheter-based sensor array for upper airway soft tissues pressure monitoring, Nat. Commun. 16 (2025) 287.
[https://doi.org/10.1038/s41467-024-55088-y]


Gyurin Moon is an undergraduate in the School of Biomedical Engineering at Inje University. Her research interests are cognitive neuroscience and targeted drug delivery. She participated in a major study abroad program in the United States and actively served as a teaching assistant for the Basic Math course taught by Prof. Seonggwang Yoo at the University.

Youngkyu Hwang is an Assistant Professor in the Graduate School of Management of Technology at Hoseo University. He received his B.S. in life science from Kyunghee University, South Korea, in 2007. He then earned his doctorate in materials science and engineering from Gwangju Institute of Science and Technology, South Korea, in 2018. He continued his postdoctoral training in materials science and engineering at Nanyang Technological University in 2019. He joined Sungkyunkwan University as a postdoctoral fellow in 2020, and his latest research is focused on the fundamental study and applications of biomaterials for flexible and wearable electronics.

Seonggwang Yoo is an Assistant Professor in the College of Biomedical Science & Health at Inje University. He received his B.S. degree in Physics and his Ph.D. degree in Materials Science and Engineering from Gwangju Institute of Science and Technology (GIST) in 2014 and 2020, respectively. He then moved to Northwestern University as a postdoctoral scholar in the Querrey Simpson Institute for Bioelectronics. His research interests include soft materials and mechanics for flexible electronics and bioelectronics with various biomedical applications.