
A Review of Soft Robotic Actuations for Minimally Invasive Implantable Devices and Sensors
ⓒ The Korean Sensors Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Soft robotic actuations are emerging as a transformative paradigm for minimally invasive implantable devices and sensors. Unlike mechanically passive, rigid systems that often require open surgery, these systems enable compact insertion followed by programmable in situ deployment through diverse actuation mechanisms, including fluidic unfolding and eversion, magnetically guided deformation, thermally triggered shape-memory polymers, and electrically driven elastomeric actuators. In this review, we systematically categorize recent advances in soft robotic actuation strategies for implantable bioelectronics and analyze how these mechanisms facilitate minimally invasive delivery, adaptive conformal interfacing, and multifunctional operation across various organ systems. We further discuss complementary materials and structural innovations—such as bioresorbable platforms, tissue-adhesive interfaces, and mechanically optimized architectures—that enhance chronic stability and biocompatibility. Beyond summarizing recent demonstrations, this manuscript identifies two critical gaps hindering clinical translation: the lack of systematic evaluation of actuator-induced mechanical effects on biological tissues and the limited integration of intrinsic self- and environmental sensing for closed-loop operation. By outlining these challenges and emerging research directions, this review aims to provide a conceptual framework for the rational design and future development of next-generation minimally invasive soft robotic implants.
Keywords:
Soft bioelectronics, Implantable devices, Soft robotics, Minimally invasive surgery1. INTRODUCTION
Implantable bioelectronic devices hold substantial potential to transform diverse domains of science and technology, with applications spanning medical treatments for epilepsy, Parkinson’s disease, and severe injuries to the spine or cochlea [1,2], as well as wearable robotic systems such as prosthetics and exoskeletons designed to augment human capabilities. However, these devices typically require invasive surgical procedures for implantation, which are associated with significant risks, including infection, surgical trauma, aesthetic complications, and prolonged hospitalization and rehabilitation before resuming daily activities [3,4]. Such issues arise because most conventional implantable electronics currently available are mechanically passive systems, whose deployment depends entirely on open surgical approaches. Even with the adoption of advanced surgical robotic systems, limitations persist due to the physical dimensions of the end effectors and the additional operating space to position the implants onto delicate target tissues.
To mitigate these challenges, extensive research efforts have focused on electrode platforms based on flexible, stretchable, and injectable materials. Flexible thin-film electrodes fabricated on elastomeric substrates have enabled improved mechanical compliance with soft tissues [5], reducing micromotion-induced damage while maintaining stable electrical performance. Beyond planar flexible platforms, injectable bioelectronic systems based on hydrogels or elastomeric composites have recently emerged as a promising alternative, allowing syringe-based delivery of neural electrodes that form conductive interfaces in situ [6]. Such soft electrodes minimize surgical footprints, conform intimately to complex tissue geometries, and exhibit mechanical properties closely matched to neural tissues, thereby reducing foreign-body responses and improving chronic stability [7]. Despite these advantages, most injectable and flexible electrode systems remain mechanically passive, lacking the ability to actively position, deploy over large areas, or adapt their geometry in response to anatomical constraints.
In recent years, research has increasingly explored the integration of soft robotic actuation with sensing technologies to overcome the inherent mechanical passiveness of conventional implantable electronics. By adopting robot-like capabilities, these systems can navigate through small incisions to reach targeted sites and deploy large-area electrode arrays or various transducers [8]. These systems can navigate through narrow incisions or catheters in a compact, folded configuration and subsequently expand, unfold, or morph into their functional form via diverse actuation methods, including fluidic inflation, magnetic guidance, thermal triggers, and shape-memory polymers. Supporting such transformations, recent advances in geometry-driven, stretchable electronic designs have enabled the robust integration of soft electronics and multimodal sensors onto highly deformable substrates [9]. These approaches ensure mechanical compliance while preserving reliable sensing and stimulation performance under dynamic strain. By embedding stretchable sensors in combination with such soft robotic actuators, users can precisely position the implantable devices to the targeting biological tissues or organs, facilitating on-demand neural stimulation, localized drug delivery, physiological signal monitoring, and closed-loop therapeutic feedback. This integration represents a critical step toward next-generation implantable bio interfaces.
In this review, we want to introduce a new paradigm of implantable soft bioelectronics called ‘soft robotic implants’, which is a class of bioelectronics that incorporates soft robotic actuations and sensing capabilities. We aim to provide a general overview of this emerging research topic, especially focused on useful soft robotic actuations and functionalities that have a high potential to be incorporated with other soft bioelectronic interfaces for various organs and applications. The following are our discussions about actuation mechanisms that have been utilized in the state-of-the-art soft robotic implants recently demonstrated in several emerging papers, followed by add-on functionalities that would be essential for further developments of the future multi-functional soft robotic implants.
2. ACTUATION TYPES FOR SOFT ROBOTIC IMPLANTS
Actuation is a core feature that distinguishes soft robotic implants from conventional bioelectronic devices. These actuation mechanisms not only enable the transformation of device shapes but also facilitate minimally invasive delivery and precise deployment. In this section, we highlight several representative actuation strategies – fluidic, magnetic, and shape-memory polymer systems – employed in different organ systems.
2.1 Fluidic Actuation
Fluidic soft actuators utilize pressurized media – typically air, water, or biocompatible fluids – to generate shape transformation and motion. The implant is delivered in a compact form and expands to its functional configuration inside the body. This mechanism enables minimally invasive surgeries and precise organ targeting.
The most common strategy for a deployment system is an origami-inspired folding mechanism, in which we can reduce its two-dimensional device size via a traditional Japanese art-inspired folding pattern. A landmark work by Coles et al. demonstrated an origami-inspired fluidic actuator that deploys a large-area ECoG array through radial expansion once inside the skull (Fig. 1(a)) [10]. This approach enables the integration of stretchable electronics and sensors onto folded soft substrates, ensuring effective cortical interfacing across wide areas. Origami techniques are particularly advantageous due to their programmable kinematics, compact initial volume, and ability to support multimodal device integration such as embedded electrodes, strain sensors [11], and microfluidic channels.
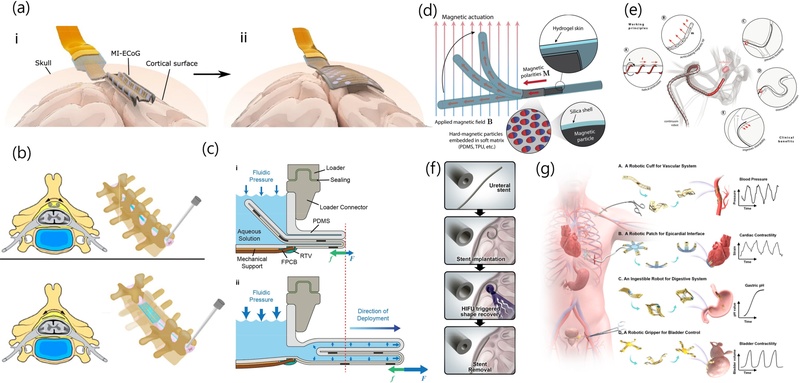
Representative soft robotic actuation strategies; (a) Origami-inspired fluidic actuation for minimally invasive large-area ECoG placement. Adapted from Ref. [10]. (b) Shape-actuating implant for minimally invasive spinal cord stimulation. Adapted from Ref. [12]. (c) Fluidic actuator deploying an electrocorticography (ECoG) array from a compact form. Adapted from Ref. [32]. (d) Ferromagnetic soft continuum robot with programmable magnetic deformation. Adapted from Ref. [16]. (e) Helical magnetic robot for navigation through complex vasculature. Adapted from Ref. [17]. (f) Ultrasound-triggered shape recovery of implanted shape-memory devices. Adapted from Ref. [25]. (g) Skin-inspired sensory robots for physiological monitoring and control across vascular, cardiac, digestive, and bladder systems. Adapted from Ref. [8].
Beyond the origami mechanism, an unfurling motion that can be seen in a rolling paper has also been demonstrated by the fluidic actuation. Woodington et al. developed a shape-actuating electronic implant that combines thin, flexible electronics with integrated fluidic channels as a spinal cord stimulator (SCS) (Fig. 1(b)) [12]. The device can be rolled into a compact cylindrical shape to fit within a standard percutaneous needle and then unfurled in situ into a paddle-shaped configuration. This approach merges the clinical benefits of paddle-type devices with the minimal invasiveness of needle-type implants. It paves the way for fluid-driven sensory and stimulation implants that offer a large functional footprint while reducing surgical burden and enabling deployment under local anesthesia.
Although folding- and unfurling-based mechanisms have demonstrated promising applications in minimally invasive deployment, they are fundamentally constrained by the requirement that the entire folded device must be inserted through an incision prior to expansion. As the surface area of the final interface increases, the folded device inevitably becomes thicker, raising the risk of tissue damage during insertion even before deployment occurs. To overcome this limitation, Song et al. introduced a bio-inspired fluidic actuation strategy known as ‘eversion’, which mimics the growth pattern of tree roots [13]. In this approach, the folded substrate is stored within a loader positioned outside the incision, and only the everting tip is advanced through the opening to progressively deploy the surface area (Fig. 1(c)). This configuration ensures that the insertion depth remains constant, in contrast to origami- or unfurling-based mechanisms, where the compression depth scales with the final device size. The critical advantage of this design has been validated through an electrocorticography (ECoG) electrode array, which achieved successful in-vivo recording of somatosensory evoked potentials (SSEP) in a minipig during acute surgery via a small craniotomy.
In addition to the application for central nervous systems like the brain or the spinal cord, fluidic systems have also been applied to various other organ systems. In the gastrointestinal (GI) tract, soft actuations have enabled applications such as site-specific drug delivery or localized physiological sensing. Integration of the fluidic actuations with various physiological sensing systems allows navigating through the highly deformable and wet GI environment for the above application. [14,15].
In summary, fluidic actuation enables fast and precise mechanical transformations with relatively high force density, offering a simple yet powerful means of achieving minimally invasive functionality.
2.2 Magnetic Actuation
Magnetic actuation enables wireless control of soft implants by applying external magnetic fields to magnetically responsive materials embedded within the device. Magnetic actuation combined with position-sensing mechanisms, such as fluoroscopy, CT, or magnetometers, allows high controllability of the soft magnetic robot motions with high precision.
Thread-shaped ferromagnetic soft continuum robots developed by Kim et al. have shown significant potential of such magnetic actuation for minimally invasive soft bioelectronics. These robots are fabricated from elastomeric matrices embedded with hard-magnetic microparticles, enabling programmable magnetization profiles along their bodies [16]. This design allows complex, continuous deformation in response to uniform magnetic fields, producing a wide range of 3D shapes and motions—including bending, twisting, and gripping—without requiring the integration of multiple actuators (Fig. 1(d)). These capabilities make ferromagnetic soft continuum robots particularly suitable for minimally invasive interventions in deep or confined anatomical regions.
In line with the development of thread-like soft robots mentioned above, Dreyfus et al. Further advanced this technique by enabling the forward and backward motions of the continuum robots [17]. The proposed thread robot consists of a rod-shaped inner core surrounded by a helical structure; the inner rod rotates in response to an applied magnetic field, while the helical structure enables forward and backward movement depending on the direction of the magnetic field rotation. Such reversible and tunable motion has demonstrated its potential to open doors to myriad applications that require precise navigation through intricated vasculatures in the brain, especially to treat aneurysms (Fig. 1(e)).
Beyond the thread-shaped soft continuum robots, soft magnetic microrobots such as the milli-spinner—a hollow cylindrical robot with helical fins—navigate tubular environments at speeds exceeding 0.2 m/s, powered solely by rotating magnetic fields. This platform offers on-the-fly drug delivery and mechanical interaction while preserving blood flow through its internal channel design [18]. Another remarkable application is in targeted kidney stone dissolution: researchers have developed thin, flexible soft robots embedded with urease and micromagnets that are guided magnetically through the urinary tract. Once positioned near the kidney stone, they locally release the enzyme to dissolve the stone, providing a minimally invasive alternative to traditional surgical methods [19].
Magnetic actuation allows untethered deployment and motion for soft robotic implants, which provides critical advantages over other actuation mechanisms for chronic in-vivo validation, especially enabling constraint-free movement of target animals [20]. Also, this feature eliminates the need for internal power sources and batteries for deployment motions [21], which are especially beneficial for highly confined anatomical spaces such as the cerebral vasculature or spinal canal. Importantly, the biocompatibility of magnetically loaded elastomers has been well-demonstrated, ensuring minimal immune response [22]. Eventually, these open venues for multi-functional and responsive implants that are both adaptive and safe for long-term use.
2.3 Thermal actuation
For implantable systems, thermal actuations are particularly attractive as a soft actuator, because they enable wireless, untethered, and programmable shape changes, reducing or even eliminating the need for bulky mechanical components or onboard power supplies [23]. In some cases, actuation can be triggered directly by physiological conditions—such as body temperature—allowing operation without batteries or complex controllers, thereby simplifying peripheral hardware for actuation [24].
Among them, here we mainly focus on discussing the use of SMPs, capable of triggering their pre-programmed shape morphing capabilities with respect to multiple external stimuli, such as lights and heat, including active and passive heating with human body temperature. In Fig. 1(f), the use of high-intensity focused ultrasound (HIFU) activates the recovery of SMP-based implants deep inside the brain, as demonstrated by Zhu et al. [25]. This non-contact thermal trigger facilitates precise shape morphing even in anatomically constrained or sensitive tissue regions. Since HIFU can be applied externally and focused on specific internal targets, it enables remote control of the implant without the need for physical access.
Leveraging the advantage of SMP-based shape morphing mechanisms mentioned above, Bae, J. Y et al. introduced a biodegradable and self-deployable electronic tent electrode that leverages the minimal actuator design enabled by a passive heating system for SMPs for ECoG [26]. The device is designed for syringe-based delivery in a highly compact form and unfolds into a three-dimensional tent shape once inside the cranial cavity. This enables large-area conformal contact with the brain surface while maintaining a minimally invasive footprint. Over time, the device naturally degrades, eliminating the need for retrieval surgery – a milestone for transient biomedical implants.
Light-based actuation of SMPs has also been explored to achieve precise, localized shape transformation. By incorporating photothermal fillers such as gold nanorods, carbon nanotubes, or graphene into SMP matrices, these systems can convert incident light into heat, triggering shape recovery with high spatial and temporal control [27,28]. This approach enables wireless, targeted actuation through optical fibers or minimally invasive endoscopes. Moreover, the ability to modulate light intensity and wavelength provides additional control over actuation speed and recovery profile, expanding the functional versatility of SMP-based implants.
Wubin Bai and colleagues further advanced the SMP-based shape-morphing concept by integrating skin-inspired sensor arrays with an active heating system for stimuli-responsive 3D SMP microarchitectures (Fig. 1(g)), enabling fully flexible, wireless electronic implants that combine mechanical adaptation with functional sensing and data acquisition [8]. These systems are built using mechanically guided assembly, allowing for programmable geometry and modular integration of electronics.
Additionally, SMP-based structures offer inherent advantages for implantable applications. Because the actuation originates from the deformation of the material itself, SMP systems require minimal peripheral hardware, eliminating the need for complex mechanical linkages or large onboard components. Eventually, these structures are compatible with bioelectric signal sensing, advancing toward fully functional and biocompatible soft robotic interfaces.
2.4 Electrical Actuation
Electrically-responsive materials undergo mechanical transformation in response to applied external electrical fields or current. These materials include dielectric elastomer actuators (DEAs) and liquid crystalline elastomers (LCEs).
In DEAs, a soft dielectric elastomer layer is sandwiched between compliant electrodes. Upon voltage application, electrostatic attraction between the electrodes generates Maxwell stress, leading to thickness compression and in-plane expansion of the elastomer membrane. Such electrostatically driven large-strain deformation has been widely explored in soft robotic systems, offering high compliance and mechanical adaptability suitable for minimally invasive deployment and biointegrated applications [29].
Beyond direct membrane deformation, DEAs have also been integrated with enclosed fluidic chambers to enable electrically controlled pneumatic or hydraulic pressure generation. In such configurations, voltage-induced membrane displacement modulates chamber volume, thereby producing controllable fluidic pressure output for soft robotic manipulation and implantable actuation. Recent studies have demonstrated that coupling electrostatic elastomer actuation with fluid-mediated force transmission can enhance output force while preserving structural softness, including frameless fluid-electrode architectures that enable translucent and mechanically compliant soft robotic platforms [30].
LCEs combine the ordered characteristics of liquid crystal materials with elastic polymer networks. Under electrical stimulation, these materials undergo reversible shape change due to electrically induced structural reconfiguration within the polymer matrix. This mechanism enables programmable deformation and tunable mechanical behavior in soft structures. Recent advances have demonstrated electrically addressable LCE architectures capable of spatially controlled and reversible three-dimensional morphing, highlighting their potential for soft robotic systems that require compact, lightweight, and minimally invasive actuation strategies. Such material-level programmability offers opportunities for integrated implantable devices and sensor platforms where adaptive mechanical conformity and electronic control are essential [31].
3. FUNCTIONALITIES
In addition to actuation mechanisms for minimally invasive surgical implantation, traditional challenges remain in the field of soft bioelectronics for developing multifunctional, chronically stable implantable devices. These challenges include achieving long-term capabilities for sensing or stimulating biological tissues, eliminating the need for surgical retrieval, and ensuring intimate, reliable electrode–tissue interfaces. While these functionalities have been extensively explored in prior works on soft bioelectronics, they have yet to be integrated with shape-morphing actuation and sensing mechanisms—an opportunity that holds great promise for advancing the next generation of multifunctional soft robotic implants.
3.1 Long-term biocompatibility
The primary source of biocompatibility challenges in bioelectronic systems lies in the host immune system’s tendency to recognize and reject implanted artificial structures, a response known as the foreign-body reaction. Numerous strategies have been explored to mitigate this immune response and promote long-term integration. In this context, prior work can be broadly categorized into two complementary approaches: materials development, aimed at reducing immunogenicity at the device–tissue interface, and structural optimization, designed to enhance mechanical and geometric compatibility with surrounding biological tissues.
Hydrogel is regarded as one of the most promising materials for long-term implantation. It features high-water content, tunable mechanical properties, and biochemical versatility. Their high hydration level enables mechanical compliance closely matching that of ultra-soft tissues such as brain and spinal cord, thereby reducing strain mismatch at the device–tissue interface and mitigating micromotion-induced injury [33]. The capability of hydrogel replicates key features of the extracellular matrix(ECM), which promote cell adhesion and neural integration [34]. These attributes, combined with their compatibility with microfabrication and 3D printing, make hydrogels a versatile platform for implantable systems that maintain stable electrical performance and biocompatibility over extended periods.
Zwitterionic materials present a further advantage by resisting nonspecific protein adsorption and inhibiting the adhesion of immune cells like macrophages (Fig. 2(a)). Yang et al. demonstrated that zwitterionic hydrogels exhibited strong resistance to foreign-body reaction in a mouse model [35]. Combined with robotics, Cabanach et al. developed zwitterionic 3D-printed non-immunogenic stealth microrobots that maintained stealth functionality to macrophages in vivo without triggering immune recognition [36]. These microrobots highlight the potential of zwitterionic coatings not only for static implants but also for mobile or shape-morphing biomedical devices, underscoring their versatility in chronic implantation scenarios. Collectively, these materials combine mechanical softness with surface chemistry that actively suppresses fibrotic encapsulation, extending the functional lifetime of implants.
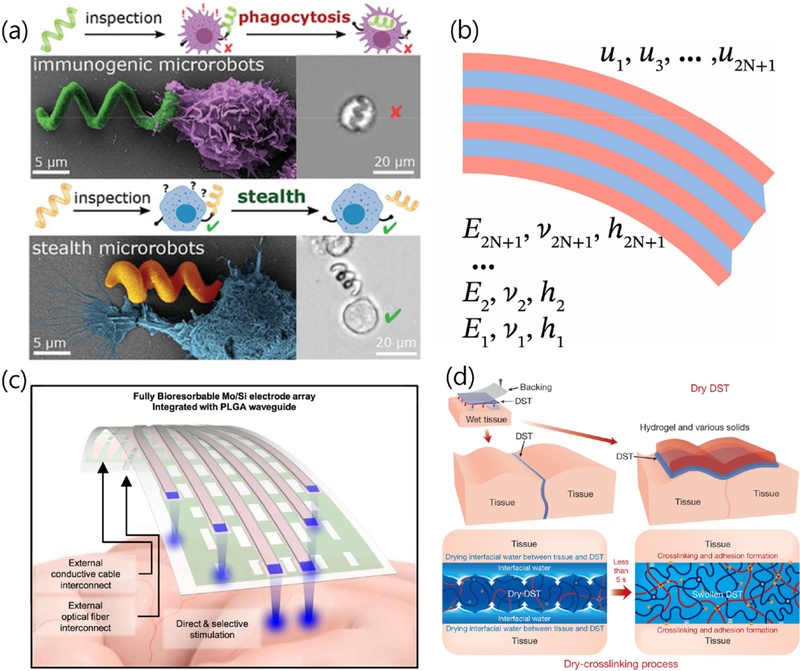
Key functionalities of soft robotic actuators in implantable systems; (a) Immunogenic vs. stealth microrobots. Adapted from Ref. [36]. (b) shear-lag design visualization. Adapted from Ref. [37]. (c) Fully bioresorbable electrode array. Adapted from Ref. [43]. (d) Double-sided dry tape (DST) adhesion. Adapted from Ref. [45].
Structural optimization is equally important as material selection in ensuring the long-term stability of implantable bioelectronic systems. Beyond biocompatible material interfaces, the mechanical design of the device plays a pivotal role in minimizing chronic tissue irritation and maintaining functional performance over time. One representative approach is the shear-lag–inspired composite design, which strategically segments the device structure in thickness to reduce its overall bending stiffness without compromising its functional integrity.
The shear-lag model (Fig. 2(b)) describes how load is transferred between stiff and compliant regions via interfacial shear stresses. In the context of bioelectronics, this principle has been applied to laminated films with alternating stiff and soft layers to accommodate shear motions between stiff layers upon bending [37]. For a given overall thickness, shear-lag–inspired designs can significantly reduce bending stiffness, allowing the implant to more closely match the mechanical modulus of surrounding biological tissues. This compliance facilitates dynamic adaptation to tissue deformations, preserving intimate, conformal contact at the electrode–tissue interface during movement. By reducing mechanical mismatch, such designs help maintain signal quality during chronic operation and mitigate the immune responses associated with micromotion-induced irritation.
In addition to shear-lag-inspired design, the use of bio-inspired 3D micro/nanostructures offers advanced functionalities such as selective permeability and mechanical robustness. Recent reviews highlight the use of soft elastomer-based microstructures, inspired by natural surfaces, in creating flexible sensors, dry/wet adhesives, and energy harvesters [38]. These strategies not only improve device-tissue interaction but also provide scalable and low-cost solutions for large-area soft implant deployment.
3.2 Bioresorbability
Bioresorbable electronics offer a transformative approach for temporary implantable devices by enabling complete in vivo degradation after fulfilling their functional purpose, thereby eliminating the need for surgical retrieval. Such devices are fabricated from bioresorbable materials—including transient metals [39], bioresorbable polymers [40], and naturally derived substrates [41]—that dissolve into biocompatible byproducts via hydrolysis, enzymatic degradation, or metabolic absorption. The degradation profile can be precisely tuned through copolymer composition, degree of crosslinking, and protective encapsulation layers, allowing device lifetime to be synchronized with clinical requirements [42].
Clinically relevant systems have been demonstrated, such as a fully bioresorbable, flexible hybrid opto-electronic neural implant capable of simultaneous electrophysiological recording and optogenetic stimulation in the brain (Fig. 2(c)) [43]. Another case is a wireless, dual-mode bioresorbable stimulator for peripheral nerve regeneration that operated reliably over therapeutic timeframes before complete resorption [44]. These examples underline the potential of bioresorbable platforms for short-term neuromodulation, post-operative monitoring, and pediatric applications, where avoiding repeat surgeries is of particular benefit.
As materials and their structural design strategies advance, future bioresorbable systems are expected to integrate multimodal sensing, targeted actuation, and closed-loop therapeutic control into single, transient platforms—maximizing clinical impact while minimizing long-term tissue burden.
3.3 Contact reliability
Reliable contact stability is one of the major challenges in soft bioelectronics due to the arbitrary 3D shapes of organ systems and various physiological motions from respiration, heartbeat, peristalsis, and muscle contraction. Such motions can lead to micro gaps at the device–tissue interface, causing signal drift, reduced stimulation accuracy, and premature device failure. Thus, adhesion strategies ensuring stable, conformal contact are of traditional interest for high-fidelity signal transduction over prolonged implantation.
Permanent wet-tissue adhesion has been achieved using double-sided dry tapes that rapidly bond hydrated tissues to device surfaces without external stimuli (Fig. 2(d)) [45]. This method provides robust mechanical coupling even under challenging surgical conditions. For applications requiring temporary adhesion, bioresorbable adhesives present an effective solution. Yang et al. developed a photocurable, bioresorbable adhesive that forms a mechanically stable, electrically conductive interface between flexible bioelectronic devices and soft tissues, which gradually degrades after the intended operational period [7]. Bioresorbable adhesive interfaces for biomedical applications further emphasize tunable degradation rates, strong wet adhesion, and compatibility with diverse device substrates [46].
Collectively, these advances provide a strong foundation for contact-reliable bioelectronic systems capable of maintaining signal integrity over extended timeframes, while allowing non-invasive removal or complete bio resorption—an essential requirement for next-generation multifunctional soft robotic implants.
4. DISCUSSION & CONCLUSION
4.1 Implications of actuator-induced mechanical deformation on neural tissues
The integration of soft actuators into implantable bioelectronic systems inevitably introduces mechanical deformation to surrounding tissues, raising fundamental questions regarding mechanobiological safety, particularly in the brain and spinal cord. While recent soft robotic implant studies have demonstrated promising in vivo feasibility, the majority of these works infer safety indirectly through acute implantation outcomes or post hoc histological analysis rather than through systematic mechanical characterization.
From a broader biomechanical perspective, prior neurotrauma studies clearly demonstrate that neural injury is governed not only by deformation magnitude but also by deformation rate and mode. For example, three-dimensional in vitro compression models have shown that neuronal injury strongly depends on both strain amplitude and strain rate, indicating that dynamic loading conditions critically influence neural damage thresholds [47]. Similarly, in vivo spinal cord injury models have revealed a strong correlation between maximum principal strain and tissue damage across contusion and dislocation scenarios [48]. These findings establish that neural tissues exhibit quantifiable strain-based injury criteria, even in the absence of implantable devices.
Despite this foundational knowledge, such mechanobiological principles have not yet been systematically incorporated into the design or evaluation of soft robotic implants. Existing implant studies—including fluidically deployable spinal cord stimulation systems, deployable ECoG platforms, and shape-memory–based implants triggered by external stimuli—suggest that actuator-induced deformation can be tolerated under carefully constrained conditions [25,26]. However, these observations remain qualitative and application-specific, and do not yet translate into generalizable design guidelines.
Taken together, these considerations highlight a critical gap between established neural injury biomechanics and the emerging field of soft robotic implants. Bridging this gap will require future studies that explicitly link actuator deformation modes, strain levels, and actuation dynamics to tissue-level mechanical and biological responses. Such efforts are essential not only for improving safety but also for enabling rational design and reliable clinical translation of soft robotic implant technologies.
4.2 Sensing as a key enabler for soft robotic implants
The paradigm of soft robotic implants proposed in this review envisions soft robotic platforms that integrate actuation and sensing to enable minimally invasive implantation of bioflexible electronic devices. To realize this vision, proprioception, achieved through self-sensing and environmental sensing, is essential for reliable deployment, safe actuation, and adaptive interaction with biological tissues.
Despite its importance, sensing integration in soft robotic implants remains extremely limited. To date, explicit self-sensing has been demonstrated only in rare cases, such as the on-board strain sensor used to detect deployment status in a soft robotic ECoG system. In most other reported platforms, sensing is either restricted to environmental measurements, such as neural signal recording after deployment, or inferred indirectly through external imaging modalities, including fluoroscopy, MRI, or ultrasound. As a result, actuation and sensing are largely decoupled, and most systems operate in an open-loop manner.
These observations indicate that while sensing is already present at the system level, its integration as an intrinsic feedback mechanism remains largely unexplored. Rather than a limitation, this gap highlights a significant opportunity for future research. The development of robust self-sensing and multimodal environmental sensing will be critical for enabling closed-loop control, improving safety, and advancing soft robotic implants toward intelligent and clinically translatable systems.
The rapid advancement of implantable soft robotic systems underscores their transformative potential in next-generation bioelectronics. Across fluidic, thermal, optical, and magnetic actuation modalities, recent studies in functional integration of these soft robotic actuators with bioelectrical interfaces have demonstrated minimally invasive surgical implantations to diverse target organs enabled by programmable deployment. Each actuation strategy carries distinct strengths and limitations: fluidic actuation provides large force density and rapid deployment but is constrained by tethered fluidic hardware peripherals; magnetically responsive systems offer precise spatiotemporal control but require a bulky external magnetic field generator and sophisticated control system; shape memory polymers enable compact delivery with remote stimulation triggers, though their slow recovery speed and long-term durability remain challenges. A key insight from these diverse approaches is that no single mechanism is universally optimal, and hybrid or multi-modal actuation strategies may provide the best fit to a specific clinical practice.
Equally important are the materials and structural design considerations that dictate chronic stability. Biocompatibility challenges arise from the foreign-body response, necessitating surface modifications such as zwitterionic coatings to reduce immune rejection. Structural optimization, exemplified by shear-lag inspired composites, provides a powerful means to match mechanical compliance with tissue and maintain conformal electrode contact during dynamic motion. Bioresorbable systems further expand the design space by eliminating the need for retrieval surgeries, an essential milestone toward transient biomedical implants. Together, these strategies highlight the necessity of integrating both material science and mechanical engineering principles in soft implant design.
In conclusion, soft robotic implants represent a compelling frontier at the intersection of materials science, biomedical engineering, and robotics. By uniting advances in soft robotic actuation, biomaterials, and structural optimization, these systems hold promise for overcoming long-standing barriers in bioelectronics, including chronic immune responses and mechanical mismatch between devices and tissues. Continued interdisciplinary research will be essential to realize their potential in enabling minimally invasive, adaptive, and multifunctional implants for next-generation healthcare.
Acknowledgments
This research was supported by the National Research Foundation of Korea (NRF), funded by the Ministry of Science and ICT (MSIT), through the Basic Science Research Program for Outstanding Early-Career Researchers (Seed Research) (Grant No. RS-2025-21342968).
REFERENCES
-
Y. Hanein, J. Goding, Guest Editorial: Implantable bioelectronics, APL Bioeng. 8 (2024) 020401.
[https://doi.org/10.1063/5.0209537]

-
C.A. Angeli, M. Boakye, R.A. Morton, J. Vogt, K. Benton, Y. Chen, et al., Recovery of Over-Ground Walking after Chronic Motor Complete Spinal Cord Injury, N. Engl. J. Med. 379 (2018) 1244–1250.
[https://doi.org/10.1056/NEJMoa1803588]

-
M.P. Engle, B.P. Vinh, N. Harun, D. Koyyalagunta, Infectious complications related to intrathecal drug delivery system and spinal cord stimulator system implantations at a comprehensive cancer pain center, Pain Physician 16 (2013) 251–257.
[https://doi.org/10.36076/ppj.2013/16/251]

-
J.E. Poole, M.J. Gleva, T. Mela, M.K. Chung, D.Z. Uslan, R. Borge, et al., Complication Rates Associated With Pacemaker or Implantable Cardioverter-Defibrillator Generator Replacements and Upgrade Procedures, Circulation 122 (2010) 1553–1561.
[https://doi.org/10.1161/CIRCULATIONAHA.110.976076]

-
F. Vitale, S.R. Summerson, B. Aazhang, C. Kemere, M. Pasquali, Neural Stimulation and Recording with Bidirectional, Soft Carbon Nanotube Fiber Microelectrodes, ACS Nano 9 (2015) 4465–4474.
[https://doi.org/10.1021/acsnano.5b01060]

-
J. Liu, T.-M. Fu, Z. Cheng, G. Hong, T. Zhou, L. Jin, et al., Syringe-injectable electronics, Nat. Nanotechnol. 10 (2015) 629–636.
[https://doi.org/10.1038/nnano.2015.115]

-
Q. Yang, T. Wei, R.T. Yin, M. Wu, Y. Xu, J. Koo, et al., Photocurable bioresorbable adhesives as functional interfaces between flexible bioelectronic devices and soft biological tissues, Nat. Mater. 20 (2021) 1559–1570.
[https://doi.org/10.1038/s41563-021-01051-x]

-
L. Zhang, S. Xing, H. Yin, H. Weisbecker, H.T. Tran, Z. Guo, et al., Skin-inspired, sensory robots for electronic implants, Nat. Commun. 15 (2024) 4777.
[https://doi.org/10.1038/s41467-024-48903-z]

-
J.H. Kim, S.E. Lee, J.H. Park, H.J. Jin, J.S. Choe, J. Ko, et al., Next-Generation Sensors with Three-Dimensional Micro-/Nano-Structures, J. Sens. Sci. Technol. 33 (2024) 419–428.
[https://doi.org/10.46670/JSST.2024.33.6.419]

-
L. Coles, D. Ventrella, A. Carnicer-Lombarte, A. Elmi, J.G. Troughton, M. Mariello, et al., Origami-inspired soft fluidic actuation for minimally invasive large-area electrocorticography, Nat. Commun. 15 (2024) 6290.
[https://doi.org/10.1038/s41467-024-50597-2]

-
S. Lee, K. Pak, J.C. Yang, S. Park, Development of Stretchable Electronics Using Geometric Strategies and Applications, J. Sens. Sci. Technol. 32 (2023) 370–377.
[https://doi.org/10.46670/JSST.2023.32.6.370]

-
B.J. Woodington, V.F. Curto, Y.-L. Yu, H. Martínez-Domínguez, L. Coles, G.G. Malliaras, et al., Electronics with shape actuation for minimally invasive spinal cord stimulation, Sci. Adv. 7 (2021) eabg7833.
[https://doi.org/10.1126/sciadv.abg7833]

-
E.W. Hawkes, L.H. Blumenschein, J.D. Greer, A.M. Okamura, A soft robot that navigates its environment through growth, Sci. Robot. 2 (2017) eaan3028.
[https://doi.org/10.1126/scirobotics.aan3028]

-
V. Consumi, L. Lindenroth, J. Merlin, D. Stoyanov, A. Stilli, Design and Evaluation of the SoftSCREEN Capsule for Colonoscopy, IEEE Robot. Autom. Lett. 8 (2023) 1659–1666.
[https://doi.org/10.1109/LRA.2023.3241811]

-
A. Giri, C. Girerd, J. Cervera-Torralba, M.T. Tolley, T.K. Morimoto, InchIGRAB: An Inchworm-Inspired Guided Retraction and Bending Device for Vine Robots During Colonoscopy, IEEE/ASME Trans. Mechatron. 30 (2025) 6870–6881.
[https://doi.org/10.1109/TMECH.2025.3535876]

-
Y. Kim, G.A. Parada, S. Liu, X. Zhao, Ferromagnetic soft continuum robots, Sci. Robot. 4 (2019) eaax7329.
[https://doi.org/10.1126/scirobotics.aax7329]

-
R. Dreyfus, Q. Boehler, S. Lyttle, P. Gruber, J. Lussi, C. Chautems, et al., Dexterous helical magnetic robot for improved endovascular access, Sci. Robot. 9 (2024) eadh0298.
[https://doi.org/10.1126/scirobotics.adh0298]

- S. Wu, S. Leanza, L. Lu, Y. Chang, Q. Li, D. Stone, et al., Magnetic Milli-spinner for Robotic Endovascular Surgery, ArXiv., arXiv:2410.21112 (2024).
-
A. Khabbazian, L. Kwong, A. Lewis, E. Liu, N. Abdelrazec, A.C. Bakenecker, et al., Kidney Stone Dissolution By Tetherless, Enzyme-Loaded, Soft Magnetic Miniature Robots, Adv. Healthcare Mater. (2025) e2403423.
[https://doi.org/10.1002/adhm.202403423]

-
S. Yin, D.R. Yao, Y. Song, W. Heng, X. Ma, H. Han, et al., Wearable and Implantable Soft Robots, Chem. Rev. 124 (2024) 11585–11636.
[https://doi.org/10.1021/acs.chemrev.4c00513]

-
A.C. Bakenecker, A. von Gladiss, H. Schwenke, A. Behrends, T. Friedrich, K. Lüdtke-Buzug, et al., Navigation of a magnetic micro-robot through a cerebral aneurysm phantom with magnetic particle imaging, Sci. Rep. 11 (2021) 14082.
[https://doi.org/10.1038/s41598-021-93323-4]

-
J. Wan, Z. Nie, J. Xu, Z. Zhang, S. Yao, Z. Xiang, et al., Millimeter-scale magnetic implants paired with a fully integrated wearable device for wireless biophysical and biochemical sensing, Sci. Adv. 10 (2024) eadm9314.
[https://doi.org/10.1126/sciadv.adm9314]

-
J. Hu, Y. Zhu, H. Huang, J. Lu, Recent advances in shape–memory polymers: Structure, mechanism, functionality, modeling and applications, Prog. Polym. Sci. 37 (2012) 1720–1763.
[https://doi.org/10.1016/j.progpolymsci.2012.06.001]

-
Q. Zhao, W. Zou, Y. Luo, T. Xie, Shape memory polymer network with thermally distinct elasticity and plasticity, Sci. Adv. 2 (2016) e1501297.
[https://doi.org/10.1126/sciadv.1501297]

-
Y. Zhu, K. Deng, J. Zhou, C. Lai, Z. Ma, H. Zhang, et al., Shape-recovery of implanted shape-memory devices remotely triggered via image-guided ultrasound heating, Nat, Commun. 15 (2024) 1123.
[https://doi.org/10.1038/s41467-024-45437-2]

-
J.Y. Bae, G.S. Hwang, Y.S. Kim, J. Jeon, M. Chae, J.W. Kim, et al., A biodegradable and self-deployable electronic tent electrode for brain cortex interfacing, Nat. Electron. 7 (2024) 815–828.
[https://doi.org/10.1038/s41928-024-01216-x]

-
X. Qian, Y. Zhao, Y. Alsaid, X. Wang, M. Hua, T. Galy, et al., Artificial phototropism for omnidirectional tracking and harvesting of light, Nat. Nanotechnol. 14 (2019) 1048–1055.
[https://doi.org/10.1038/s41565-019-0562-3]

-
H. Zeng, P. Wasylczyk, C. Parmeggiani, D. Martella, M. Burresi, D.S. Wiersma, Light-Fueled Microscopic Walkers, Adv. Mater. 27 (2015) 3883–3887.
[https://doi.org/10.1002/adma.201501446]

-
M.H.O.R. Molla, J. Chen, C. Xu, Advancing soft robotics: recent progress in dielectric elastomer and fluid actuators, npj Robot. 4 (2026) 17.
[https://doi.org/10.1038/s44182-026-00074-3]

-
C. Christianson, N.N. Goldberg, D.D. Deheyn, S. Cai, M.T. Tolley, Translucent soft robots driven by frameless fluid electrode dielectric elastomer actuators, Sci. Robot. 3 (2018) eaat1893.
[https://doi.org/10.1126/scirobotics.aat1893]

-
S. Kim, G. Dong, A. Mangan, D. Agrawal, S. Cai, T.K. Morimoto, LCE-integrated soft skin for millimeter-scale steerable soft everting robots, Sci. Adv. 11 (2025) eadw8636.
[https://doi.org/10.1126/sciadv.adw8636]

-
S. Song, F. Fallegger, A. Trouillet, K. Kim, S.P. Lacour, Deployment of an electrocorticography system with a soft robotic actuator, Sci. Robot. 8 (2023) eadd1002.
[https://doi.org/10.1126/scirobotics.add1002]

-
C. Jiao, J. Liu, S. Yan, Z. Xu, Z. Hou, W. Xu, Hydrogel-based soft bioelectronic interfaces and their applications, J. Mater. Chem. C 13 (2025) 2620–2645.
[https://doi.org/10.1039/D4TC04150J]

-
P. Lu, D. Ruan, M. Huang, M. Tian, K. Zhu, Z. Gan, et al., Harnessing the potential of hydrogels for advanced therapeutic applications: current achievements and future directions, Signal Transduct. Target. Ther. 9 (2024) 166.
[https://doi.org/10.1038/s41392-024-01852-x]

-
L. Zhang, Z. Cao, T. Bai, L. Carr, J.-R. Ella-Menye, C. Irvin, et al., Zwitterionic hydrogels implanted in mice resist the foreign-body reaction, Nat. Biotechnol. 31 (2013) 553–556.
[https://doi.org/10.1038/nbt.2580]

-
P. Cabanach, A. Pena-Francesch, D. Sheehan, U. Bozuyuk, O. Yasa, S. Borros, et al., Zwitterionic 3D-Printed Non-Immunogenic Stealth Microrobots, Adv. Mater. 32 (2020) 2003013.
[https://doi.org/10.1002/adma.202003013]

-
Z. Wang, X. Lin, J. Liu, N. Lu, Shear-lag model of laminated films with alternating stiff and soft layers wrinkling on soft substrates, J. Mech. Phys. Solids 202 (2025) 106172.
[https://doi.org/10.1016/j.jmps.2025.106172]

-
Y. Jung, I. Park, Bio-Inspired Micro/Nanostructures for Functional Applications: A Mini-Review, J. Sens. Sci. Technol. 32 (2023) 31–38.
[https://doi.org/10.46670/JSST.2023.32.1.31]

-
S.-W. Hwang, H. Tao, D.-H. Kim, H. Cheng, J.-K. Song, E. Rill, et al., A Physically Transient Form of Silicon Electronics, Science 337 (2012) 1640–1644.
[https://doi.org/10.1126/science.1226325]

-
S.-K. Kang, R.K.J. Murphy, S.-W. Hwang, S.M. Lee, D.V. Harburg, N.A. Krueger, et al., Bioresorbable silicon electronic sensors for the brain, Nature 530 (2016) 71–76.
[https://doi.org/10.1038/nature16492]

-
H. Tao, S.-W. Hwang, B. Marelli, B. An, J.E. Moreau, M. Yang, et al., Silk-based resorbable electronic devices for remotely controlled therapy and in vivo infection abatement, Proc. Natl. Acad. Sci. U.S.A. 111 (2014) 17385–17389.
[https://doi.org/10.1073/pnas.1407743111]

-
M. Irimia-Vladu, “Green” electronics: biodegradable and biocompatible materials and devices for sustainable future, Chem. Soc. Rev. 43 (2014) 588–610.
[https://doi.org/10.1039/C3CS60235D]

-
M. Cho, J.-K. Han, J. Suh, J.J. Kim, J.R. Ryu, I.S. Min, et al., Fully bioresorbable hybrid opto-electronic neural implant system for simultaneous electrophysiological recording and optogenetic stimulation, Nat. Commun. 15 (2024) 2000.
[https://doi.org/10.1038/s41467-024-45803-0]

-
H.-Y. Ahn, J.B. Walters, R. Avila, S. Oh, S.G. Seo, J.U. Kim, et al., Bioresorbable, wireless dual stimulator for peripheral nerve regeneration, Nat. Commun. 16 (2025) 4752.
[https://doi.org/10.1038/s41467-025-59835-7]

-
H. Yuk, C.E. Varela, C.S. Nabzdyk, X. Mao, R.F. Padera, E.T. Roche, et al., Dry double-sided tape for adhesion of wet tissues and devices, Nature 575 (2019) 169–174.
[https://doi.org/10.1038/s41586-019-1710-5]

-
Y.S. Lee, S. Jeon, D.W. Kim, C. Pang, Biodegradable Adhesive Interfaces for Bioelectronic and Biomedical Applications, J. Sens. Sci. Technol. 34 (2025) 224–230.
[https://doi.org/10.46670/JSST.2025.34.3.224]

-
E. Bar-Kochba, M.T. Scimone, J.B. Estrada, C. Franck, Strain and rate-dependent neuronal injury in a 3D in vitro compression model of traumatic brain injury, Sci. Rep. 6 (2016) 30550.
[https://doi.org/10.1038/srep30550]

-
C.M. Russell, A.M. Choo, W. Tetzlaff, T.-E. Chung, T.R. Oxland, Maximum Principal Strain Correlates with Spinal Cord Tissue Damage in Contusion and Dislocation Injuries in the Rat Cervical Spine, J. Neurotrauma 29 (2012) 1574–1585.
[https://doi.org/10.1089/neu.2011.2225]


Jeonghyeon Im is a graduate student at the Daegu Gyeongbuk Institute of Science and Technology (DGIST), Daegu, South Korea. His research interests focus on soft robotics for brain–computer interface applications and bio-signal processing. He is currently engaged in research on both hardware and software platforms for bio-signal acquisition and analysis, including neural spike signals and soft actuation.

Sukho Song is an Assistant Professor at DGIST (Daegu Gyeongbuk Institute of Science and Technology) in Daegu, South Korea. His research interests lie at the intersection of design and manufacturing of multi-scale soft robots, combining microscale features with larger-scale soft robotic architectures to achieve new functionalities. His diverse research experience at several world-renowned research institutions, including Carnegie Mellon University in the United States, Seoul National University in South Korea, EPFL and EMPA in Switzerland, and the Max-Planck Institute in Germany, has led him to multidisciplinary research projects. These include the development of gecko-inspired adhesive grippers, electronics-free soft robots, and minimally invasive soft robotic brain implants, which have been published in prestigious journals such as Advanced Materials, PNAS, Advanced Science, and Science Robotics.

